
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Tomohiko Fukuda | + 2457 word(s) | 2457 | 2021-07-26 10:00:03 | | | |
| 2 | Amina Yu | Meta information modification | 2457 | 2021-07-26 11:00:31 | | | | |
| 3 | Amina Yu | Meta information modification | 2457 | 2021-07-26 11:01:34 | | |
Video Upload Options
The effects of bone morphogenetic proteins (BMPs), members of the transforming growth factor-β (TGF-β) family, in endometrial cancer (EC) have yet to be determined. In this study, we analyzed the TCGA and MSK-IMPACT datasets and investigated the effects of BMP2 and of TWSG1, a BMP antagonist, on Ishikawa EC cells. Frequent ACVR1 mutations and high mRNA expressions of BMP ligands and receptors were observed in EC patients of the TCGA and MSK-IMPACT datasets. Ishikawa cells secreted higher amounts of BMP2 compared with ovarian cancer cell lines. Exogenous BMP2 stimulation enhanced EC cell sphere formation via c-KIT induction. BMP2 also induced EMT of EC cells, and promoted migration by induction of SLUG. The BMP receptor kinase inhibitor LDN193189 augmented the growth inhibitory effects of carboplatin. Analyses of mRNAs of several BMP antagonists revealed that TWSG1 mRNA was abundantly expressed in Ishikawa cells. TWSG1 suppressed BMP7-induced, but not BMP2-induced, EC cell sphere formation and migration. Our results suggest that BMP signaling promotes EC tumorigenesis, and that TWSG1 antagonizes BMP7 in EC. BMP signaling inhibitors, in combination with chemotherapy, might be useful in the treatment of EC patients.
1. Introduction
2. Tumor Promoting Effect of BMP Signaling in Endometrial Cancer
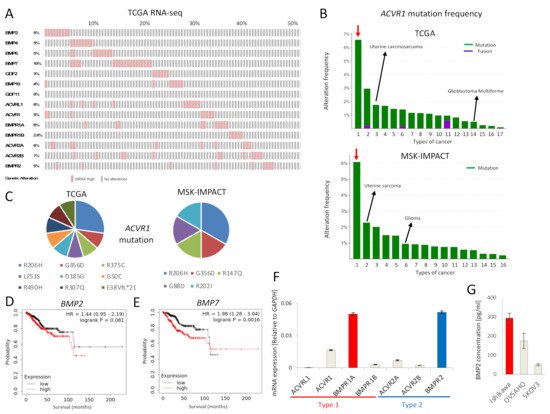
2.1. BMP Signaling Is Activated in EC
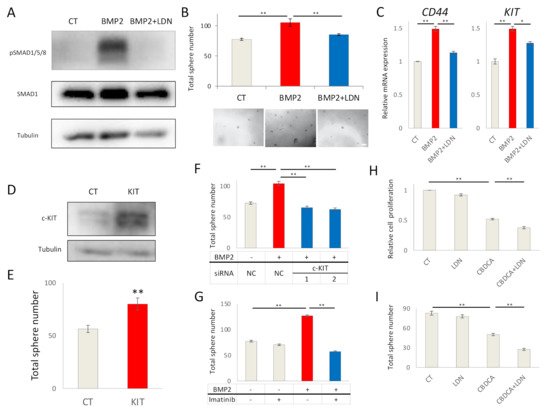
2.2. BMP2 Promotes EC Cell Stemness by c-KIT Induction
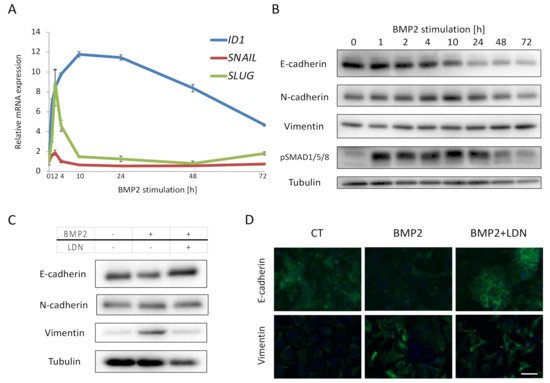
2.3. BMP2 Induces EMT of EC Cells
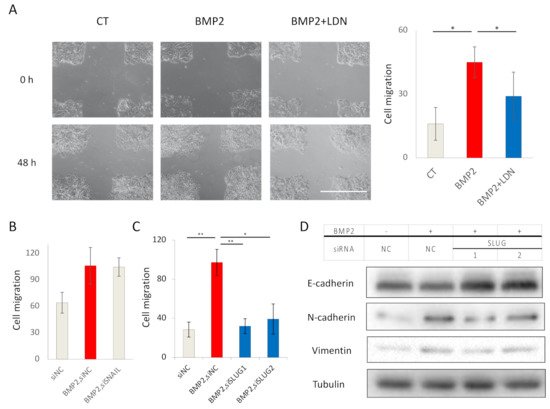
2.4. BMP2 Enhances EC Cell Migration via SLUG Induction
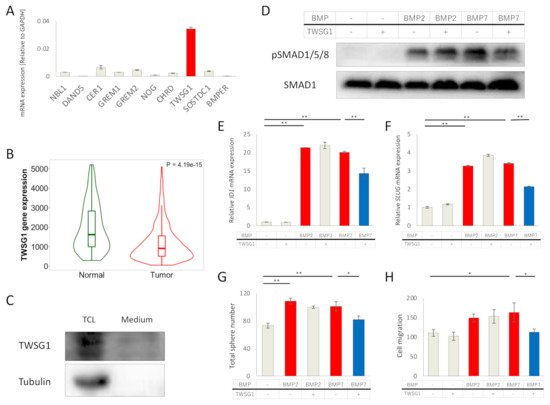
2.5. TWSG1 Antagonizes BMP7 in EC Cells
References
- Bray, F.; Ferlay, J.; Soerjomataram, I.; Siegel, R.L.; Torre, L.A.; Jemal, A. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J. Clin. 2018, 68, 394–424.
- Johnson, A.L.; Medina, H.N.; Schlumbrecht, M.P.; Reis, I.; Kobetz, E.N.; Pinheiro, P.S. The role of histology on endometrial cancer survival disparities in diverse Florida. PLoS ONE 2020, 15, e0236402.
- Gonzalez Bosquet, J.; Terstriep, S.A.; Cliby, W.A.; Brown-Jones, M.; Kaur, J.S.; Podratz, K.C.; Keeney, G.L. The impact of multi-modal therapy on survival for uterine carcinosarcomas. Gynecol. Oncol. 2010, 116, 419–423.
- de Jong, R.A.; Nijman, H.W.; Wijbrandi, T.F.; Reyners, A.K.; Boezen, H.M.; Hollema, H. Molecular markers and clinical behavior of uterine carcinosarcomas: Focus on the epithelial tumor component. Mod. Pathol. 2011, 24, 1368–1379.
- Han, H.J.; Jain, P.; Resnick, A.C. Shared ACVR1 mutations in FOP and DIPG: Opportunities and challenges in extending biological and clinical implications across rare diseases. Bone 2018, 109, 91–100.




