
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Alistair Gunn | + 2205 word(s) | 2205 | 2021-07-09 05:49:07 | | | |
| 2 | Lily Guo | Meta information modification | 2205 | 2021-07-22 07:36:50 | | |
Video Upload Options
Seizures are common in newborn infants with hypoxic-ischemic encephalopathy and are highly associated with adverse neurodevelopmental outcomes. The impact of seizure activity on the developing brain and the most effective way to manage these seizures remain surprisingly poorly understood, particularly in the era of therapeutic hypothermia. Critically, the extent to which seizures exacerbate brain injury or merely reflect the underlying evolution of injury is unclear. Current anticonvulsants, such as phenobarbital and phenytoin have poor efficacy and preclinical studies suggest that most anticonvulsants are associated with adverse effects on the developing brain. Levetiracetam seems to have less potential neurotoxic effects than other anticonvulsants but may not be more effective. Given that therapeutic hypothermia itself has significant anticonvulsant effects, randomized controlled trials of anticonvulsants combined with therapeutic hypothermia, are required to properly determine the safety and efficacy of these drugs. Small clinical studies suggest that prophylactic phenobarbital administration may improve neurodevelopmental outcomes compared to delayed administration; however, larger high-quality studies are required to confirm this. In conclusion, there is a distinct lack of high-quality evidence for whether and to what extent neonatal seizures exacerbate brain damage after hypoxia-ischemia and how best to manage them in the era of therapeutic hypothermia.
1. Neonatal Seizures after Hypoxic-Ischemic Encephalopathy
2. The Biology of Neonatal Seizures
3. Do Seizures Exacerbate Brain Damage after HIE?
4. Should We Try to Prevent Seizures?
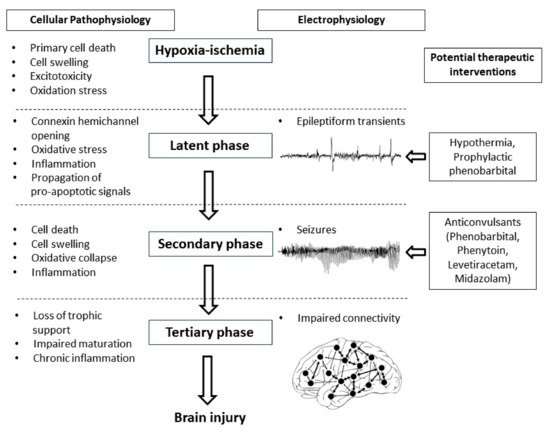
5. Conclusions
References
- Jacobs, S.E.; Berg, M.; Hunt, R.; Tarnow-Mordi, W.O.; Inder, T.E.; Davis, P.G. Cooling for newborns with hypoxic ischaemic encephalopathy. Cochrane Database Syst. Rev. 2013, 1, CD003311.
- Edwards, A.D.; Brocklehurst, P.; Gunn, A.J.; Halliday, H.; Juszczak, E.; Levene, M.; Strohm, B.; Thoresen, M.; Whitelaw, A.; Azzopardi, D. Neurological outcomes at 18 months of age after moderate hypothermia for perinatal hypoxic ischaemic encephalopathy: Synthesis and meta-analysis of trial data. BMJ 2010, 340, c363.
- Davidson, J.O.; Wassink, G.; van den Heuij, L.G.; Bennet, L.; Gunn, A.J. Therapeutic hypothermia for neonatal hypoxic-ischemic encephalopathy—Where to from here? Front. Neurol. 2015, 6, 198.
- Kharoshankaya, L.; Stevenson, N.J.; Livingstone, V.; Murray, D.M.; Murphy, B.P.; Ahearne, C.E.; Boylan, G.B. Seizure burden and neurodevelopmental outcome in neonates with hypoxic-ischemic encephalopathy. Dev. Med. Child Neurol. 2016, 58, 1242–1248.
- Lin, Y.K.; Hwang-Bo, S.; Seo, Y.M.; Youn, Y.A. Clinical seizures and unfavorable brain MRI patterns in neonates with hypoxic ischemic encephalopathy. Medicine 2021, 100, e25118.
- Basti, C.; Maranella, E.; Cimini, N.; Catalucci, A.; Ciccarelli, S.; Del Torto, M.; Di Luca, L.; Di Natale, C.; Mareri, A.; Nardi, V.; et al. Seizure burden and neurodevelopmental outcome in newborns with hypoxic-ischemic encephalopathy treated with therapeutic hypothermia: A single center observational study. Seizure 2020, 83, 154–159.
- Davidson, J.O.; Bennet, L.; Gunn, A.J. Evaluating anti-epileptic drugs in the era of therapeutic hypothermia. Pediatr. Res. 2019, 85, 931–933.
- Glass, H.C.; Shellhaas, R.A.; Wusthoff, C.J.; Chang, T.; Abend, N.S.; Chu, C.J.; Cilio, M.R.; Glidden, D.V.; Bonifacio, S.L.; Massey, S.; et al. Contemporary Profile of Seizures in Neonates: A Prospective Cohort Study. J. Pediatr. 2016, 174, 98–103.e1.
- Khazipov, R.; Khalilov, I.; Tyzio, R.; Morozova, E.; Ben-Ari, Y.; Holmes, G.L. Developmental changes in GABAergic actions and seizure susceptibility in the rat hippocampus. Eur. J. Neurosci. 2004, 19, 590–600.
- Payne, J.A.; Rivera, C.; Voipio, J.; Kaila, K. Cation-chloride co-transporters in neuronal communication, development and trauma. Trends Neurosci. 2003, 26, 199–206.
- Pond, B.B.; Berglund, K.; Kuner, T.; Feng, G.; Augustine, G.J.; Schwartz-Bloom, R.D. The chloride transporter Na(+)-K(+)-Cl- cotransporter isoform-1 contributes to intracellular chloride increases after in vitro ischemia. J. Neurosci. 2006, 26, 1396–1406.
- Nardou, R.; Ferrari, D.C.; Ben-Ari, Y. Mechanisms and effects of seizures in the immature brain. Semin. Fetal Neonatal Med. 2013, 18, 175–184.
- Jantzie, L.L.; Getsy, P.M.; Denson, J.L.; Firl, D.J.; Maxwell, J.R.; Rogers, D.A.; Wilson, C.G.; Robinson, S. Prenatal Hypoxia-Ischemia Induces Abnormalities in CA3 Microstructure, Potassium Chloride Co-Transporter 2 Expression and Inhibitory Tone. Front. Cell. Neurosci. 2015, 9, 347.
- Jensen, F.E. Neonatal seizures: An update on mechanisms and management. Clin. Perinatol. 2009, 36, 881–900.
- Zhou, C.; Sun, H.; Klein, P.M.; Jensen, F.E. Neonatal seizures alter NMDA glutamate receptor GluN2A and 3A subunit expression and function in hippocampal CA1 neurons. Front. Cell. Neurosci. 2015, 9, 362.
- Miller, S.P.; Weiss, J.; Barnwell, A.; Ferriero, D.M.; Latal-Hajnal, B.; Ferrer-Rogers, A.; Newton, N.; Partridge, J.C.; Glidden, D.V.; Vigneron, D.B.; et al. Seizure-associated brain injury in term newborns with perinatal asphyxia. Neurology 2002, 58, 542–548.
- Glass, H.C.; Glidden, D.; Jeremy, R.J.; Barkovich, A.J.; Ferriero, D.M.; Miller, S.P. Clinical Neonatal Seizures are Independently Associated with Outcome in Infants at Risk for Hypoxic-Ischemic Brain Injury. J. Pediatr. 2009, 155, 318–323.
- Srinivasakumar, P.; Zempel, J.; Trivedi, S.; Wallendorf, M.; Rao, R.; Smith, B.; Inder, T.; Mathur, A.M. Treating EEG Seizures in Hypoxic Ischemic Encephalopathy: A Randomized Controlled Trial. Pediatrics 2015, 136, e1302–e1309.
- Van Rooij, L.G.; Toet, M.C.; van Huffelen, A.C.; Groenendaal, F.; Laan, W.; Zecic, A.; de Haan, T.; van Straaten, I.L.; Vrancken, S.; van Wezel, G.; et al. Effect of treatment of subclinical neonatal seizures detected with aEEG: Randomized, controlled trial. Pediatrics 2010, 125, e358–e366.
- Peeples, E.S.; Rao, R.; Dizon, M.L.V.; Johnson, Y.R.; Joe, P.; Flibotte, J.; Hossain, T.; Smith, D.; Hamrick, S.; DiGeronimo, R.; et al. Predictive models of neurodevelopmental outcomes after neonatal hypoxic-ischemic encephalopathy. Pediatrics 2021, 147, e2020022962.
- Kwon, J.M.; Guillet, R.; Shankaran, S.; Laptook, A.R.; McDonald, S.A.; Ehrenkranz, R.A.; Tyson, J.E.; O’Shea, T.M.; Goldberg, R.N.; Donovan, E.F.; et al. Clinical seizures in neonatal hypoxic-ischemic encephalopathy have no independent impact on neurodevelopmental outcome: Secondary analyses of data from the neonatal research network hypothermia trial. J. Child Neurol. 2011, 26, 322–328.
- Holmes, G.L. The long-term effects of neonatal seizures. Clin. Perinatol. 2009, 36, 901–914.
- Pisani, F.; Orsini, M.; Braibanti, S.; Copioli, C.; Sisti, L.; Turco, E.C. Development of epilepsy in newborns with moderate hypoxic-ischemic encephalopathy and neonatal seizures. Brain Dev. 2009, 31, 64–68.
- Liu, X.; Jary, S.; Cowan, F.; Thoresen, M. Reduced infancy and childhood epilepsy following hypothermia-treated neonatal encephalopathy. Epilepsia 2017, 58, 1902–1911.
- Young, L.; Berg, M.; Soll, R. Prophylactic barbiturate use for the prevention of morbidity and mortality following perinatal asphyxia. Cochrane Database Syst. Rev. 2016, CD001240.
- Johne, M.; Römermann, K.; Hampel, P.; Gailus, B.; Theilmann, W.; Ala-Kurikka, T.; Kaila, K.; Löscher, W. Phenobarbital and midazolam suppress neonatal seizures in a noninvasive rat model of birth asphyxia, whereas bumetanide is ineffective. Epilepsia 2021, 62, 920–934.
- Hall, R.T.; Hall, F.K.; Daily, D.K. High-dose phenobarbital therapy in term newborn infants with severe perinatal asphyxia: A randomized, prospective study with three-year follow-up. J. Pediatr. 1998, 132, 345–348.
- Gunn, A.J.; Bennet, L.; Gunning, M.I.; Gluckman, P.D.; Gunn, T.R. Cerebral hypothermia is not neuroprotective when started after postischemic seizures in fetal sheep. Pediatr. Res. 1999, 46, 274–280.
- Barks, J.D.; Liu, Y.Q.; Shangguan, Y.; Silverstein, F.S. Phenobarbital augments hypothermic neuroprotection. Pediatr. Res. 2010, 67, 532–537.
- Krishna, S.; Hutton, A.; Aronowitz, E.; Moore, H.; Vannucci, S.J. The effects of adding prophylactic phenobarbital to therapeutic hypothermia in the term-equivalent hypoxic-ischemic rat. Pediatr. Res. 2018, 83, 506–513.




