
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Federica Collino | + 2410 word(s) | 2410 | 2021-06-25 08:11:24 | | | |
| 2 | Lily Guo | Meta information modification | 2410 | 2021-07-19 06:00:51 | | |
Video Upload Options
Intercellular communication governs multicellular interactions in complex organisms. A variety of mechanisms exist through which cells can communicate, e.g., cell-cell contact, the release of paracrine/autocrine soluble molecules, or the transfer of extracellular vesicles (EVs). EVs are membrane-surrounded structures released by almost all cell types, acting both nearby and distant from their tissue/organ of origin. In the kidney, EVs are potent intercellular messengers released by all urinary system cells and are involved in cell crosstalk, contributing to physiology and pathogenesis. Moreover, urine is a reservoir of EVs coming from the circulation after crossing the glomerular filtration barrier—or originating in the kidney. Thus, urine represents an alternative source for biomarkers in kidney-related diseases, potentially replacing standard diagnostic techniques, including kidney biopsy.
1. Introduction
2. Extracellular Vesicles
| Exosomes | Microvesicles | |
|---|---|---|
| Size (nm) | 20–150 | 100–1000 |
| Origin | MVB fusion with plasma membrane | Outward blebbing of plasma membrane |
| Release process | Constitutive/Cellular Activation | Constitutive/Cellular Activation |
| Pathways | Tetraspanins-dependent | Cytoskeleton reorganization- and Ca2+-dependent |
| Composition | ||
| Proteins | CD9, CD63, CD81, Alix, TSG101, Rabs, annexins, MHC-II, CD86, signaling-, oncogenic-, integrin-, adhesion molecules, flotillins, Hsp70 and Hsp90 |
Annexins, flotillins, Alix, TSG101, CD40, ARF-6, selectins, phosphatidylserine, Rho family members, MMP2, ADAM, GAPDH, pyruvate kinase, proteins localized to centrosome, nucleolus, cytoplasm and mitochondria |
| Nucleic Acids | mRNAs, miRNAs, lncRNAs, mtDNA, gDNA, nDNA, dsDNA |
mRNAs, miRNAs, gDNA |
| Isolation Method | UC, DG-UC, filtration, immune- affinity, PEG precipitation, SEC |
UC, DG-UC, filtration, SEC |
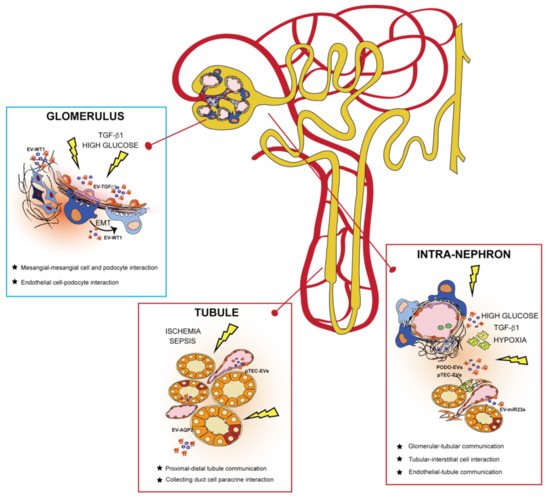
3. EVs in the Kidney: Mechanism of Cell-to-Cell Communication
4. Urinary EVs as Biomarkers for Personalized Medicine
5. uEVs as a Liquid Biopsy in Kidney-related Diseases
| Disease | No. of Patient | Target | uEV Isolation | Detection Method | Main Results | Ref. |
|---|---|---|---|---|---|---|
| 25 patients and 5 HD | WT1 | Differential UC | WB | -WT1 was increased FSGS patients compared with healthy volunteers or SSNS patients. -Urinary exosomal WT1 was decreased in patients in remission for either FSGS or SSNS or following steroid treatment in six SSNS subjects. |
H. Zhou, 2013 [36] |
|
| Nephrotic syndrome (NS) | 40 | WT1 | Differential UC | WB | -WT1 was detected in 25 patients (62.5%). -No significant correlation between WT1 amount and steroid responsiveness or renal pathological condition was found. |
H. Lee, 2012 [37] |
| 13 | miR-193a | -ExoQuick exosome precipitation (System Biosciences) | -qRT-PCR -ROC analysis -WB |
-miR-193a was higher in children with primary FSGS than those in children with MCNS. | Z. Huang, 2017 [38] |
|
| 129 NS children and 126 age-/sex-matched HD. | miR-194-5p, miR-146b-5p, miR-378a-3p, miR-23b-3p and miR-30a-5p | UC | -High-throughput Illumina sequencing -qRT-PCR |
-These 5 miRNAs were increased in NS and markedly reduced during the clinical remission period. -The concentrations of miR-194-5p and miR-23b-3p were positively correlated with the urine protein content and were markedly higher in the high urine protein group than in the low urine protein group. |
T. Chen, 2019 [39] |
|
| Glomerulonephritis (GN) | 12 IgAN, 12 TBMN patients and 6 HD | ANPEP, VASN, A1AT and CP | Differential UC | Label-free LC-MS/MS | -These four proteins are biomarkers to distinguish between early IgAN from TBMN. | P. G. Moon, 2011 [40] |
| 55 IgAN patients and 24 HD | CCL2 mRNA | Differential UC | qRT-PCR | - CCL2 was specifically expressed in uEVs of patients with GN compared to healthy controls. - Exosomal CCL2 was correlated with tubulointerstitial inflammation and C3 deposition in GN patients. |
Y. Feng, 2018 [41] |
|
| Lupus nephritis (LN) |
13 LN patients and 8 HD | miR-26a | Differential UC | qRT-PCR | -miR-26a levels in urinary exosomes were higher compared with healthy control. | O. Ichii, 2014 [42] |
| 41 SLE patients, 27 LN and 20 HD | miR-146a | UC | qRT-PCR | -Compared to controls, urinary level of miR-146a was higher in SLE patients. | J. Perez-Hernandez, 2020 [43] |
|
| 32 s patients with biopsy-proven LN, 15 non-lupus CKD and 20 HD | miR-29-c | Differential UC | qRT-PCR | -miR-29c correlated with the degree of renal chronicity but not with renal function. -MiR-29c expression levels could predict the degree of chronicity in patients with LN. |
C. Solé, 2015 [44] |
|
| 31 patients | let-7a and miR-21 | UC | qRT-PCR | -Urinary exosome-associated miRNA, let-7a and miR-21, could be used to guide the clinical stage of LN patients and possibly plays a role in epigenetic regulation of the kidney during the disease. | P. Tangtanatakul, 2019 [45] |
|
| Ciliopathies | 12 ciliopathy patients and 12 age- and gender-matched HD | 156 differentially expressed proteins | Differential UC | -Electro-phoresis -In-depth label-free LC-MS/MS proteomics analysis |
-The most overexpressed or downregulated proteins in uEVs (VCAN, DPEP1 and FAT4) correlated with nephronophthisis-related ciliopathies. | M.F. Stokman, 2019 [46] |
| 1 ADPKD patient and multiple HD | PC1, PC2, TMEM2 |
-Differential UC - DG-UC |
Gel electro-phoresis | In patients with PKD1 mutations, levels of PC1 and PC2 were reduced. -TMEM2 was higher in individuals with PKD1 mutations. -The PC1/TMEM2 ratio correlated inversely with height-adjusted total kidney volume in the discovery cohort. -The ratio of PC1/TMEM2 or PC2/TMEM2 could be used to distinguish individuals with PKD1 mutations from controls in a confirmation cohort. |
M.C. Hogan, 2009 [47] |
|
| Diabetic nephropathy (DN) | 48 type-1 diabetes mellitus patients and 25 HD | WT1 | UC | WB | -WT1 expression in in uEVs was higher than in controls. -WT1 levels were associated with an increase in urine protein-to-creatinine ratio, albumin-to-creatinine ratio, and serum creatinine as well as a decline in eGFR. |
A. Kalani, 2013 [48] |
| 30 HD and 30 T1D | miR-424 and miR-218 | combined centrifugation | RT-qPCR | Association of urinary exosomal level of miR-424 and miR-218 with renal damage in T1D patients. | Q. Kong, 2019 [49] |
|
| 48 | uEV miRNAs | qEV size exclusion columns (Izon Science) |
NGS | Identification of a set of miRNAs with concentration changes associated with DN occurrence, microalbuminuria status, and other variables. | V. Ghai, 2018 [50] |
|
| Obstructive nephropathy | 27 patients and 20 HD | AQP2 TGFβ-1 L1CAM |
Centrifugation | Immuno-blotting | In boys with PUV, AQP2, TGFβ-1 and L1CAM correlated with eGFR. | P. Trnka, 2012 [51] |
| 3 UPJO and 3 healthy fetusis | 633 differentially expressed proteins were identified in the amniotic fluid-derived exosomes from patients with UPJO. | Exosome Extraction Kit | iTRAQ quantitative proteomic profiles | - ACE and AP-N were significantly decreased in the amniotic fluid exosomes of women with a fetus diagnosed UPJO. - They correlate with suppressed cell proliferation, elevated ROS production, and increased pro-inflammatory cytokine levels in tubular cells. |
R. Liu, 2020 [52] |
6. EVs in Clinics: Perspectives and Limitations
7. Conclusions
References
- Taal, M.; Chertow, G.; Marsden, P.; Skorecki, K.; Yu, A.; Brenner, B. Brenner and Rector’s The Kidney, 11th ed.; Elsevier: Amsterdam, The Netherlands, 2019.
- Gounden, V.; Vashisht, R.; Jialal, I. Hypoalbuminemia; StatPearls Publishing: Treasure Island, FL, USA, 2021.
- Sirolli, V.; Pieroni, L.; Di Liberato, L.; Urbani, A.; Bonomini, M. Urinary Peptidomic Biomarkers in Kidney Diseases. Int. J. Mol. Sci. 2019, 21, 96.
- Sun, I.O.; Lerman, L.O. Urinary microRNA in kidney disease: Utility and roles. Am. J. Physiol. Ren. Physiol. 2019, 316, F785–F793.
- Gowda, S.; Desai, P.B.; Kulkarni, S.S.; Hull, V.V.; Math, A.A.K.; Vernekar, S.N. Markers of renal function tests. N. Am. J. Med. Sci. 2010, 2, 170–173.
- Campion, C.G.; Sanchez-Ferras, O.; Batchu, S.N. Potential role of serum and urinary biomarkers in diagnosis and prognosis of diabetic nephropathy. Can. J. Kidney Health Dis. 2017, 4.
- Merchant, M.L.; Rood, I.M.; Deegens, J.K.J.; Klein, J.B. Isolation and characterization of urinary extracellular vesicles: Implications for biomarker discovery. Nat. Rev. Nephrol. 2017, 13, 731–749.
- Buzás, E.I.; Toth, E.A.; Sodar, B.W.; Szabo-Taylor, K.E. Molecular interactions at the surface of extracellular vesicles. Semin. Immunopathol. 2018, 40, 453–464.
- Ståhl, A.; Johansson, K.; Mossberg, M.; Kahn, R.; Karpman, D. Exosomes and microvesicles in normal physiology, pathophysiology, and renal diseases. Pediatr. Nephrol. 2019, 34, 11–30.
- Bennett, M.R.; Devarajan, P. Proteomic analysis of acute kidney injury: Biomarkers to mechanisms. Proteom. Clin. Appl. 2011, 5, 67–77.
- Fernández-Lázaro, D.; Hernández, J.L.G.; García, A.C.; Martínez, A.C.; Mielgo-Ayuso, J.; Cruz-Hernández, J.J. Liquid biopsy as novel tool in precision medicine: Origins, properties, identification and clinical perspective of cancer’s biomarkers. Diagnostics 2020, 10, 215.
- Erdbrügger, U.; Le, T.H. Extracellular Vesicles in Renal Diseases: More than Novel Biomarkers? J. Am. Soc. Nephrol. 2016, 27, 12–26.
- Théry, C.; Witwer, K.W.; Aikawa, E.; Alcaraz, M.J.; Anderson, J.D.; Andriantsitohaina, R.; Antoniou, A.; Arab, T.; Archer, F.; Atkin-Smith, G.K.; et al. Minimal information for studies of extracellular vesicles 2018 (MISEV2018): A position statement of the International Society for Extracellular Vesicles and update of the MISEV2014 guidelines. J. Extracell. Vesicles 2018, 7, 1535750.
- Medeiros, T.; Myette, R.L.; Almeida, J.R.; Silva, A.A.; Burger, D. Extracellular vesicles: Cell-derived biomarkers of glomerular and tubular injury. Cell. Physiol. Biochem. 2020, 54, 88–109.
- Skokos, D.; Le Panse, S.; Villa, I.; Rousselle, J.C.; Peronet, R.; David, B.; Namane, A.; Mécheri, S. Mast cell-dependent B and T lymphocyte activation is mediated by the secretion of immunologically active exosomes. J. Immunol. 2001, 166, 868–876.
- Sapet, C.; Simoncini, S.; Loriod, B.; Puthier, D.; Sampol, J.; Nguyen, C.; Dignat-George, F.; Anfosso, F. Thrombin-induced endothelial microparticle generation: Identification of a novel pathway involving ROCK-II activation by caspase-2. Blood 2006, 108, 1868–1876.
- Combes, V.; Simon, A.C.; Grau, G.E.; Arnoux, D.; Camoin, L.; Sabatier, F.; Mutin, M.; Sanmarco, M.; Sampol, J.; Dignat-George, F. In vitro generation of endothelial microparticles and possible prothrombotic activity in patients with lupus anticoagulant. J. Clin. Invest. 1999, 104, 93–102.
- Park, J.E.; Tan, H.S.; Datta, A.; Lai, R.C.; Zhang, H.; Meng, W.; Lim, S.K.; Sze, S.K. Hypoxic tumor cell modulates its microenvironment to enhance angiogenic and metastatic potential by secretion of proteins and exosomes. Mol. Cell. Proteom. 2010, 9, 1085–1099.
- Tati, R.; Kristoffersson, A.-C.; Ståhl, A.-L.; Rebetz, J.; Wang, L.; Licht, C.; Motto, D.; Karpman, D. Complement activation associated with ADAMTS13 deficiency in human and murine thrombotic microangiopathy. J. Immunol. 2013, 191, 2184–2193.
- Collino, F.; Lopes, J.A.; Corrêa, S.; Abdelhay, E.; Takiya, C.M.; Wendt, C.H.C.; de Miranda, K.R.; Vieyra, A.; Lindoso, R.S. Adipose-Derived Mesenchymal Stromal Cells Under Hypoxia: Changes in Extracellular Vesicles Secretion and Improvement of Renal Recovery after Ischemic Injury. Cell. Physiol. Biochem. 2019, 52, 1463–1483.
- Hyland, M.; Mennan, C.; Wilson, E.; Clayton, A.; Kehoe, O. Pro-Inflammatory Priming of Umbilical Cord Mesenchymal Stromal Cells Alters the Protein Cargo of Their Extracellular Vesicles. Cells 2020, 9, 726.
- Borges, F.T.; Melo, S.A.; Ozdemir, B.C.; Kato, N.; Revuelta, I.; Miller, C.A.; Gattone, V.H.; LeBleu, V.S.; Kalluri, R. TGF-β1-containing exosomes from injured epithelial cells activate fibroblasts to initiate tissue regenerative responses and fibrosis. J. Am. Soc. Nephrol. 2013, 24, 385–392.
- Parolini, I.; Federici, C.; Raggi, C.; Lugini, L.; Palleschi, S.; De Milito, A.; Coscia, C.; Iessi, E.; Logozzi, M.; Molinari, A.; et al. Microenvironmental pH is a key factor for exosome traffic in tumor cells. J. Biol. Chem. 2009, 284, 34211–34222.
- Trino, S.; Lamorte, D.; Caivano, A.; De Luca, L.; Sgambato, A.; Laurenzana, I. Clinical relevance of extracellular vesicles in hematological neoplasms: From liquid biopsy to cell biopsy. Leukemia 2021, 35, 661–678.
- Brenner, A.W.; Su, G.H.; Momen-Heravi, F. Isolation of Extracellular Vesicles for Cancer Diagnosis and Functional Studies. Methods Mol. Biol. 2019, 1882, 229–237.
- Colombo, M.; Raposo, G.; Théry, C. Biogenesis, secretion, and intercellular interactions of exosomes and other extracellular vesicles. Annu. Rev. Cell Dev. Biol. 2014, 30, 255–289.
- Margolis, L.; Sadovsky, Y. The biology of extracellular vesicles: The known unknowns. PLOS Biol. 2019, 17, e3000363.
- Latifkar, A.; Hur, Y.H.; Sanchez, J.C.; Cerione, R.A.; Antonyak, M.A. New insights into extracellular vesicle biogenesis and function. J. Cell Sci. 2019, 132.
- Alvarez, M.L.; Khosroheidari, M.; Kanchi Ravi, R.; di Stefano, J.K. Comparison of protein, microRNA, and mRNA yields using different methods of urinary exosome isolation for the discovery of kidney disease biomarkers. Kidney Int. 2012, 82, 1024–1032.
- Pisitkun, T.; Shen, R.-F.; Knepper, M.A. Identification and proteomic profiling of exosomes in human urine. Proc. Natl. Acad. Sci. USA 2004, 101, 13368–13373.
- Masaoutis, C.; Al Besher, S.; Koutroulis, I.; Theocharis, S. Exosomes in Nephropathies: A Rich Source of Novel Biomarkers. Dis. Markers 2020, 2020.
- Gonzales, P.A.; Pisitkun, T.; Hoffert, J.D.; Tchapyjnikov, D.; Star, R.A.; Kleta, R.; Wang, N.S.; Knepper, M.A. Large-scale proteomics and phosphoproteomics of urinary exosomes. J. Am. Soc. Nephrol. 2009, 20, 363–379.
- Abbasian, N.S.; Salajegheh, A.; Gaspar, H.; Brett, P.O. Improving early OSV design robustness by applying ‘Multivariate Big Data Analytics’ on a ship’s life cycle. J. Ind. Inf. Integr. 2018, 10, 29–38.
- Gheinani, A.H.; Vögeli, M.; Baumgartner, U.; Vassella, E.; Draeger, A.; Burkhard, F.C.; Monastyrskaya, K. Improved isolation strategies to increase the yield and purity of human urinary exosomes for biomarker discovery. Sci. Rep. 2018, 8, 3945.
- Svenningsen, P.; Sabaratnam, R.; Jensen, B.L. Urinary extracellular vesicles: Origin, role as intercellular messengers and biomarkers; efficient sorting and potential treatment options. Acta Physiol. 2020, 228, e13346.
- Zhou, H.; Kajiyama, H.; Tsuji, T.; Hu, X.; Leelahavanichkul, A.; Vento, S.; Frank, R.; Kopp, J.B.; Trachtman, H.; Star, R.A.; et al. Urinary exosomal Wilms’ tumor-1 as a potential biomarker for podocyte injury. Am. J. Physiol. Ren. Physiol. 2013, 305, F553–F559.
- Lee, H.K.; Han, K.H.; Lee, S.E.; Kim, S.H.; Kang, H.G.; Cheong, H. Il Urinary exosomal WT1 in childhood nephrotic syndrome. Pediatr. Nephrol. 2012, 27, 317–320.
- Huang, Z.; Zhang, Y.; Zhou, J.; Zhang, Y. Urinary exosomal MIR-193a can be a potential biomarker for the diagnosis of primary focal segmental glomerulosclerosis in children. Biomed. Res. Int. 2017, 2017.
- Chen, T.; Wang, C.; Yu, H.; Ding, M.; Zhang, C.; Lu, X.; Zhang, C.Y.; Zhang, C. Increased urinary exosomal microRNAs in children with idiopathic nephrotic syndrome. EBioMedicine 2019, 39, 552–561.
- Moon, P.G.; Lee, J.E.; You, S.; Kim, T.K.; Cho, J.H.; Kim, I.S.; Kwon, T.H.; Kim, C.D.; Park, S.H.; Hwang, D.; et al. Proteomic analysis of urinary exosomes from patients of early IgA nephropathy and thin basement membrane nephropathy. Proteomics 2011, 11, 2459–2475.
- Feng, Y.; Lv, L.-L.; Wu, W.-J.; Li, Z.-L.; Chen, J.; Ni, H.-F.; Zhou, L.-T.; Tang, T.-T.; Wang, F.-M.; Wang, B.; et al. Urinary Exosomes and Exosomal CCL2 mRNA as Biomarkers of Active Histologic Injury in IgA Nephropathy. Am. J. Pathol. 2018, 188, 2542–2552.
- Ichii, O.; Otsuka-Kanazawa, S.; Horino, T.; Kimura, J.; Nakamura, T.; Matsumoto, M.; Toi, M.; Kon, Y. Decreased miR-26a expression correlates with the progression of podocyte injury in autoimmune glomerulonephritis. PLoS ONE 2014, 9, 1–12.
- Perez-Hernandez, J.; Martinez-Arroyo, O.; Ortega, A.; Galera, M.; Solis-Salguero, M.A.; Chaves, F.J.; Redon, J.; Forner, M.J.; Cortes, R. Urinary exosomal miR-146a as a marker of albuminuria, activity changes and disease flares in lupus nephritis. J. Nephrol. 2020.
- Sole, C.; Cortes-Hernandez, J.; Felip, M.L.; Vidal, M.; Ordi-Ros, J. MIR-29c in urinary exosomes as predictor of early renal fibrosis in lupus nephritis. Nephrol. Dial. Transplant. 2015, 30, 1488–1496.
- Tangtanatakul, P.; Klinchanhom, S.; Sodsai, P.; Sutichet, T.; Promjeen, C.; Avihingsanon, Y.; Hirankarn, N. Down-regulation of let-7a and miR-21 in urine exosomes from lupus nephritis patients during disease flare. Asian Pac. J. Allergy Immunol. 2019, 37, 189–197.
- Stokman, M.F.; Bijnsdorp, I.V.; Schelfhorst, T.; Pham, T.V.; Piersma, S.R.; Knol, J.C.; Giles, R.H.; Bongers, E.M.H.F.; Knoers, N.V.A.M.; Lilien, M.R.; et al. Changes in the urinary extracellular vesicle proteome are associated with nephronophthisis-related ciliopathies. J. Proteom. 2019, 192, 27–36.
- Hogan, M.C.; Manganelli, L.; Woollard, J.R.; Masyuk, A.I.; Masyuk, T.V.; Tammachote, R.; Huang, B.Q.; Leontovich, A.A.; Beito, T.G.; Madden, B.J.; et al. Characterization of PKD protein-positive exosome-like vesicles. J. Am. Soc. Nephrol. 2009, 20, 278–288.
- Kalani, A.; Mohan, A.; Godbole, M.M.; Bhatia, E.; Gupta, A.; Sharma, R.K.; Tiwari, S. Wilm’s Tumor-1 Protein Levels in Urinary Exosomes from Diabetic Patients with or without Proteinuria. PLoS ONE 2013, 8.
- Kong, Q.; Guo, X.; Guo, Z.; Su, T. Urinary exosome miR-424 and miR-218 as biomarkers for type 1 diabetes in children. Clin. Lab. 2019, 65.
- Ghai, V.; Wu, X.; Bheda-Malge, A.; Argyropoulos, C.P.; Bernardo, J.F.; Orchard, T.; Galas, D.; Wang, K. Genome-wide Profiling of Urinary Extracellular Vesicle microRNAs Associated With Diabetic Nephropathy in Type 1 Diabetes. Kidney Int. Rep. 2018, 3, 555–572.
- Trnka, P.; Ivanova, L.; Hiatt, M.J.; Matsell, D.G. Article Urinary Biomarkers in Obstructive Nephropathy. Clin. J. Am. Soc. Nephrol. 2012, 7, 1567–1575.
- Liu, R.; Zhang, W.; Luo, M.; Qin, X.; Yang, F.; Wei, Q. iTRAQ-based proteomics and in vitro experiments reveals essential roles of ACE and AP-N in the renin–angiotensin system-mediated congenital ureteropelvic junction obstruction. Exp. Cell Res. 2020, 393, 112086.
- Cully, M. Exosome-based candidates move into the clinic. Nat. Rev. Drug Discov. 2020, 20, 6–7.
- De Palma, G.; Sallustio, F.; Schena, F.P. Clinical Application of Human Urinary Extracellular Vesicles in Kidney and Urologic Diseases. Int. J. Mol. Sci. 2016, 17, 1043.
- Zhou, H.; Yuen, P.S.T.; Pisitkun, T.; Gonzales, P.A.; Yasuda, H.; Dear, J.W.; Gross, P.; Knepper, M.A.; Star, R.A. Collection, storage, preservation, and normalization of human urinary exosomes for biomarker discovery. Kidney Int. 2006, 69, 1471–1476.
- Huang, G.; Lin, G.; Zhu, Y.; Duan, W.; Jin, D. Emerging technologies for profiling extracellular vesicle heterogeneity. Lab. Chip 2020, 20, 2423–2437.
- Lian, H.; He, S.; Chen, C.; Yan, X. Flow Cytometric Analysis of Nanoscale Biological Particles and Organelles. Annu. Rev. Anal. Chem. 2019, 12, 389–409.
- Welsh, J.A.; Van Der Pol, E.; Arkesteijn, G.J.A.; Bremer, M.; Brisson, A.; Coumans, F.; Dignat-George, F.; Duggan, E.; Ghiran, I.; Giebel, B.; et al. MIFlowCyt-EV: A framework for standardized reporting of extracellular vesicle flow cytometry experiments. J. Extracell. Vesicles 2020, 9, 1713526.
- Gandham, S.; Su, X.; Wood, J.; Nocera, A.L.; Alli, S.C.; Milane, L.; Zimmerman, A.; Amiji, M.; Ivanov, A.R. Technologies and Standardization in Research on Extracellular Vesicles. Trends Biotechnol. 2020, 38, 1066–1098.




