
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Sachin Rustgi | + 2783 word(s) | 2783 | 2021-07-07 06:19:01 | | | |
| 2 | Peter Tang | Meta information modification | 2783 | 2021-07-13 07:41:44 | | |
Video Upload Options
Celiac disease, wheat sensitivity, and allergy represent three different reactions, which may occur in genetically predisposed individuals on the ingestion of wheat and derived products with various manifestations. Improvements in the disease diagnostics and understanding of disease etiology unveiled that these disorders are widespread around the globe affecting about 7% of the population. The only known treatment so far is a life-long gluten-free diet, which is almost impossible to follow because of the contamination of allegedly “gluten-free” products. Accidental contamination of inherently gluten-free products could take place at any level from field to shelf because of the ubiquity of these proteins/grains. Gluten contamination of allegedly “gluten-free” products is a constant threat to celiac patients and a major health concern. Several detection procedures have been proposed to determine the level of contamination in products for celiac patients.
1. Introduction
|
Company |
Neogen Corp. |
R-Biopharm AG |
R-Biopharm AG |
Inmunología y Genética Aplicada SA |
Romer Labs |
Tepnel Biosystem |
Morinaga Inc. |
|
Product |
Veratox |
RIDA- SCREEN |
Ridascreen® Gliadin Competitive |
INgezim Gluten |
AgraQuant® Gluten G12 |
Gluten assay |
Wheat protein |
|
Antibody |
2 mAb |
R5 mAb |
R5 mAb |
R5 mAb |
G12 mAb |
Skerritt mAb |
Wheat pAb |
|
ELISA type |
Sandwich |
Sandwich |
Competitive |
Sandwich |
Sandwich |
Sandwich |
Sandwich |
|
Time |
30 min |
1.5 h |
40 min |
60 min |
60 min |
30 min |
2.5 h |
|
Target |
gliadin |
ω, α/β- & γ-gliadins and LMWg |
ω, α/β- & γ-gliadins and LMWg |
ω, α/β- & γ-gliadins and LMWg |
α gliadins |
ω gliadins and HMWg |
Wheat proteins |
|
Antigen |
|||||||
|
LOD (mg/kg) |
n/a |
3 |
1.36 |
3 |
2 |
1 |
0.3 |
|
LOQ (mg/kg) |
10 |
5 |
5 |
10 |
4 |
10 |
3.12 |
2. Celiac Disease Prevalence
3. Hidden Gluten or Gluten Contamination
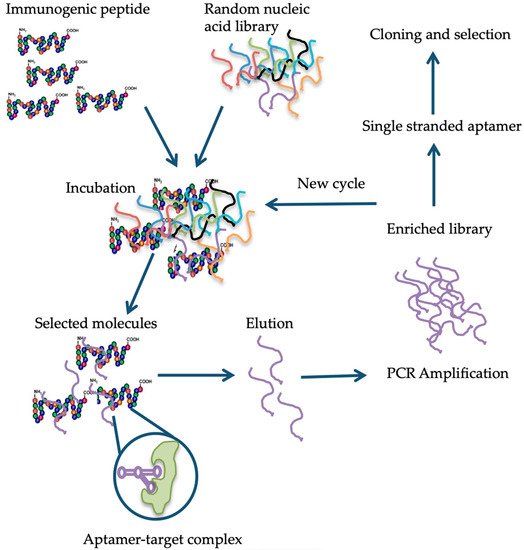
4. Available Detection Methods
|
Properties |
Aptamers |
Antibodies |
Reference |
|---|---|---|---|
|
Affinity |
Very high target affinity, dissociation constants from micro to picomolar range. |
Lower target affinity except for some monoclonal antibodies. |
[89] |
|
Immunogenic effect |
Independent of immunogenic effect, due to their in vitro production. |
Immune response can fail when the target molecule, has a structure similar to an endogenous protein. |
[90] |
|
Specificity |
High binding specificity, e.g., the Anti-theophyllin aptamer displayed 10,000-fold discrimination against caffeine (Theophyllin differs from caffeine by a single methyl group). |
Depends on target type. |
[91] |
|
Production |
In vitro. |
In vivo. Use of animals or cell lines. |
[92] |
|
Consistency |
Chemical synthesis, extreme accuracy, and reproducibility. Little or no batch-to-batch variation. |
May have in vivo variations. Restricted to environmental conditions. |
[90] |
|
Properties |
Can be optimized on demand for increasing binding affinity and specificity. |
Properties cannot be changed on demand. |
|
|
Stability |
Undergo denaturation, but reversible within minutes. |
Irreversible denaturation. Stable under physiological conditions |
[90] |
|
Range of targets |
Combinatorial library can be produced against any type of target, even toxic targets. |
Restricted to molecules that produce immunogenic effect. |
[93] |
|
Shelf-life |
Stable to long-term storage at ambient temperature. |
Limited shelf-life. |
[92] |
|
Functionalization |
Labeling does not affect affinity. |
Attachment of molecules can cause loss in affinity. |
References
- Domenico, L.; Donald, K. One and Two Dimensional (two pH) Polyacrilamide Gel Electrophoresis in a Single Gel: Separation of Wheat Proteins. Cereal Chem. 1985, 62, 314–319.
- Kasarda, D.D. Can an increase in celiac disease be attributed to an increase in the gluten content of wheat as a consequence of wheat breeding? J. Agric. Food Chem. 2013, 61, 1155–1159.
- Arranz-Otaegui, A.; Carretero, L.G.; Ramsey, M.N.; Fuller, D.Q.; Richter, T. Archaeobotanical evidence reveals the origins of bread 14,400 years ago in northeastern Jordan. Proc. Natl. Acad. Sci. USA 2018, 115, 7925–7930.
- Elli, L.; Branchi, F.; Tomba, C.; Villalta, D.; Norsa, L.; Ferretti, F.; Roncoroni, L.; Bardella, M.T. Diagnosis of gluten related disorders: Celiac disease, wheat allergy and non-celiac gluten sensitivity. World J. Gastroenterol. WJG 2015, 21, 7110.
- Kilian, B.; Martin, W.; Salamini, F. Genetic diversity, evolution and domestication of wheat and barley in the Fertile Crescent. In Evolution in Action; Springer: Berlin, Germany, 2010; pp. 137–166.
- Turner, G.D.; Dunne, M.R.; Ryan, A.W. Celiac Disease: Background and Historical Context. In Celiac Disease; Humana Press: New York, NY, USA, 2015; pp. 3–14.
- Simpson, R. Gluten-Free Rome: Celiac disease in the bioarchaeological record. COMPASS 2017, 1, 13–24.
- Van den Broeck, H.C.; de Jong, H.C.; Salentijn, E.M.; Dekking, L.; Bosch, D.; Hamer, R.J.; Gilissen, L.J.; van der Meer, I.M.; Smulders, M.J. Presence of celiac disease epitopes in modern and old hexaploid wheat varieties: Wheat breeding may have contributed to increased prevalence of celiac disease. TAG. Theor. Appl. Genet. 2010, 121, 1527–1539.
- Sapone, A.; Lammers, K.M.; Casolaro, V.; Cammarota, M.; Giuliano, M.T.; De Rosa, M.; Stefanile, R.; Mazzarella, G.; Tolone, C.; Russo, M.I. Divergence of gut permeability and mucosal immune gene expression in two gluten-associated conditions: Celiac disease and gluten sensitivity. BMC Med. 2011, 9, 23.
- Freeman, H.J. The Neolithic revolution and subsequent emergence of the celiac affection. Int. J. Celiac. Dis. 2013, 1, 19–22.
- Sapone, A.; Bai, J.C.; Ciacci, C.; Dolinsek, J.; Green, P.H.; Hadjivassiliou, M.; Kaukinen, K.; Rostami, K.; Sanders, D.S.; Schumann, M. Spectrum of gluten-related disorders: Consensus on new nomenclature and classification. BMC Med. 2012, 10, 13.
- Wen, S.; Wen, N.; Pang, J.; Langen, G.; Brew-Appiah, R.A.; Mejias, J.H.; Osorio, C.; Yang, M.; Gemini, R.; Moehs, C.P.; et al. Structural genes of wheat and barley 5-methylcytosine DNA glycosylases and their potential applications for human health. Proc. Natl. Acad. Sci. USA 2012, 109, 20543–20548.
- Shewry, P.R.; Sayanova, O.; Tatham, A.S.; Tamas, L.; Turner, M.; Richard, G.; Hickman, D.; Fido, R.; Halford, N.G.; Greenfield, J.; et al. Structure, Assembly and Targeting of Wheat Storage Proteins. J. Plant Physiol. 1995, 145, 620–625.
- Osorio, C.; Wen, N.; Gemini, R.; Zemetra, R.; von Wettstein, D.; Rustgi, S. Targeted modification of wheat grain protein to reduce the content of celiac causing epitopes. Funct. Integr. Genom. 2012, 12, 417–438.
- Lebwohl, B.; Ludvigsson, J.F.; Green, P.H. Celiac disease and non-celiac gluten sensitivity. BMJ 2015, 351, h4347.
- Biesiekierski, J.R. What is gluten? J. Gastroenterol. Hepatol. 2017, 32, 78–81.
- Shan, L.; Molberg, O.; Parrot, I.; Hausch, F.; Filiz, F.; Gray, G.M.; Sollid, L.M.; Khosla, C. Structural basis for gluten intolerance in celiac sprue. Science 2002, 297, 2275–2279.
- Shewry, P.R.; Tatham, A.S. Disulphide bonds in wheat gluten proteins. J. Cereal Sci. 1997, 25, 207–227.
- Sollid, L.M. Coeliac disease: Dissecting a complex inflammatory disorder. Nat. Rev. Immunol. 2002, 2, 647–655.
- Shan, L.; Qiao, S.W.; Arentz-Hansen, H.; Molberg, O.; Gray, G.M.; Sollid, L.M.; Khosla, C. Identification and analysis of multivalent proteolytically resistant peptides from gluten: Implications for Celiac Sprue. J. Proteome Res. 2005, 4, 1732–1741.
- Theethira, T.G.; Dennis, M.; Leffler, D.A. Nutritional consequences of celiac disease and the gluten-free diet. Expert Rev. Gastroenterol. Hepatol. 2014, 8, 123–129.
- Green, P.H.; Lebwohl, B.; Greywoode, R. Celiac disease. J. Allergy Clin. Immunol. 2015, 135, 1099–1106.
- Leonard, M.M.; Sapone, A.; Catassi, C.; Fasano, A. Celiac disease and nonceliac gluten sensitivity: A review. JAMA 2017, 318, 647–656.
- Guandalini, S.; Discepolo, V. Celiac disease. In Textbook of Pediatric Gastroenterology, Hepatology and Nutrition; Springer: Cham, Switzerland, 2016; pp. 453–469.
- Molberg, O.; McAdam, S.N.; Korner, R.; Quarsten, H.; Kristiansen, C.; Madsen, L.; Fugger, L.; Scott, H.; Noren, O.; Roepstorff, P.; et al. Tissue transglutaminase selectively modifies gliadin peptides that are recognized by gut-derived T cells in celiac disease. Nat. Med. 1998, 4, 713–717.
- Dieterich, W.; Ehnis, T.; Bauer, M.; Donner, P.; Volta, U.; Riecken, E.O.; Schuppan, D. Identification of tissue transglutaminase as the autoantigen of celiac disease. Nat. Med. 1997, 3, 797–801.
- Bethune, M.T.; Khosla, C. Parallels between pathogens and gluten peptides in celiac sprue. PLoS Pathog. 2008, 4, e34.
- Siegel, M.; Strnad, P.; Watts, R.E.; Choi, K.; Jabri, B.; Adler, G.; Ornary, B.; Khosla, C. Extracellular transglutaminase 2 is catalytically inactive, but is transiently activated upon tissue injury in the small intestine. Gastroenterology 2008, 134, A151.
- Sollid, L.M.; Jabri, B. Celiac disease and transglutaminase 2: A model for posttranslational modification of antigens and HLA association in the pathogenesis of autoimmune disorders. Curr. Opin. Immunol. 2011, 23, 732–738.
- Sollid, L.M.; Jabri, B. Triggers and drivers of autoimmunity: Lessons from coeliac disease. Nat. Rev. Immunol. 2013, 13, 294.
- Fasano, A.; Sapone, A.; Zevallos, V.; Schuppan, D. Nonceliac gluten sensitivity. Gastroenterology 2015, 148, 1195–1204.
- Tatham, A.S.; Shewry, P.R. Allergens to wheat and related cereals. Clin. Exp. Allergy 2008, 38, 1712–1726.
- Czaja-Bulsa, G. Non coeliac gluten sensitivity–A new disease with gluten intolerance. Clin. Nutr. 2015, 34, 189–194.
- Bascunan, K.A.; Vespa, M.C.; Araya, M. Celiac disease: Understanding the gluten-free diet. Eur. J. Nutr. 2017, 56, 449–459.
- Shepherd, S.; Gibson, P. Nutritional inadequacies of the gluten-free diet in both recently-diagnosed and long-term patients with coeliac disease. J. Hum. Nutr. Diet. 2013, 26, 349–358.
- Løvik, A.; Skodje, G.; Bratlie, J.; Brottveit, M.; Lundin, K. Diet adherence and gluten exposure in coeliac disease and self-reported non-coeliac gluten sensitivity. Clin. Nutr. 2017, 36, 275–280.
- Verma, A.; Gatti, S.; Galeazzi, T.; Monachesi, C.; Padella, L.; Del, B.G.; Annibali, R.; Lionetti, E.; Catassi, C. Detection of gluten content in the naturally gluten free and gluten free labelled commercially available food products in Italy. Dig. Liver Dis. 2016, 48, e279.
- Verma, A.; Gatti, S.; Galeazzi, T.; Monachesi, C.; Padella, L.; Baldo, G.; Annibali, R.; Lionetti, E.; Catassi, C. Gluten contamination in naturally or labeled gluten-free products marketed in Italy. Nutrients 2017, 9, 115.
- Catassi, C.; Bai, J.; Bonaz, B.; Bouma, G.; Calabrò, A.; Carroccio, A.; Castillejo, G.; Ciacci, C.; Cristofori, F.; Dolinsek, J. Non-celiac gluten sensitivity: The new frontier of gluten related disorders. Nutrients 2013, 5, 3839–3853.
- Allen, P.J. Primary Care Approaches. Gluten-Related Disorders: Celiac Disease, Gluten Allergy, Non-Celiac Gluten Sensitivity. Pediatric Nurs. 2015, 41, 3.
- Dominguez-Ortega, G.; Borrelli, O.; Meyer, R.; Dziubak, R.; De Koker, C.; Godwin, H.; Fleming, C.; Thapar, N.; Elawad, M.; Kiparissi, F. Extraintestinal manifestations in children with gastrointestinal food allergy. J. Pediatr. Gastroenterol. Nutr. 2014, 59, 210–214.
- Volta, U.; Caio, G.; Tovoli, F.; De Giorgio, R. Non-celiac gluten sensitivity: Questions still to be answered despite increasing awareness. Cell. Mol. Immunol. 2013, 10, 383.
- Codex Alimentarius Commission. Codex Standard 118-1979 (rev. 2008), Foods for special dietary use for persons intolerant to gluten. In Codex Alimentarium; FAO: Rome, Italy; WHO: Geneva, Switzerland, 2008; pp. 1–3.
- Thompson, T.; Lee, A.R.; Grace, T. Gluten contamination of grains, seeds, and flours in the United States: A pilot study. J. Am. Diet. Assoc. 2010, 110, 937–940.
- Thompson, T.; Dennis, M.; Emerson, L. Gluten-Free Labeling: Are Growth Media Containing Wheat, Barley, and Rye Falling through the Cracks? J. Acad. Nutr. Diet. 2018, 118, 2025.
- Falcomer, A.L.; Araújo, L.S.; Farage, P.; Monteiro, J.S.; Nakano, E.Y.; Zandonadi, R.P. Gluten contamination in food services and industry: A systematic review. Crit. Rev. Food Sci. Nutr. 2018, 1–15.
- Farage, P.; de Medeiros Nóbrega, Y.K.; Pratesi, R.; Gandolfi, L.; Assunção, P.; Zandonadi, R.P. Gluten contamination in gluten-free bakery products: A risk for coeliac disease patients. Public Health Nutr. 2017, 20, 413–416.
- Don, C.; Halbmayr-Jech, E.; Rogers, A.; Koehler, P. AACCI Approved Methods Technical Committee report: Collaborative study on the immunochemical quantitation of intact gluten in rice flour and rice-based products using G12 sandwich ELISA. Cereal Foods World 2014, 59, 187–193.
- Allred, L.K.; Kupper, C.; Iverson, G.; Perry, T.B.; Smith, S.; Stephen, R. Definition of the “Purity Protocol” for Producing Gluten-Free Oats. Cereal Chem. 2017, 94, 377–379.
- Van Eckert, R.; Pfannhauser, W.; Riedl, O. Beitrag zur Qualitätssicherung bei der Herstellung von glutenfreien Lebensmitteln. Ernährung. Nutrition 1992, 16, 511–512.
- Fritschy, F.; Windemann, H.; Baumgartner, E. Determination of wheat gliadins in food with ELISA. Zeitschrift fur Lebensmittel-Untersuchung und-Forschung 1985, 181, 379–385.
- Janssen, F.; Hägele, G.; de Baaij, J. Gluten-free products, the Dutch experience. In Coeliac Disease; Springer: Dordrecht, The Netherlands, 1991; pp. 95–100.
- Olexova, L.; Dovičovičová, L.; Švec, M.; Siekel, P.; Kuchta, T. Detection of gluten-containing cereals in flours and “gluten-free” bakery products by polymerase chain reaction. Food Control 2006, 17, 234–237.
- Hernando, A.; Mujico, J.R.; Mena, M.C.; Lombardía, M.; Mendez, E. Measurement of wheat gluten and barley hordeins in contaminated oats from Europe, the United States and Canada by Sandwich R5 ELISA. Eur. J. Gastroenterol. Hepatol. 2008, 20, 545–554.
- Geng, T.; Westphal, C.; Yeung, J. Detection of Gluten by Commercial Test Kits: Effects of Food Matrices and Extraction Procedures; American Chemical Society Symposium Series: Washington, DC, USA, 2008; Volume 1001, pp. 462–475.
- Kelly, C.P.; Bai, J.C.; Liu, E.; Leffler, D.A. Advances in diagnosis and management of celiac disease. Gastroenterology 2015, 148, 1175–1186.
- Rostami, K.; Kerckhaert, J.; Tiemessen, R.; Von Blomberg, B.M.E.; Meijer, J.W.; Mulder, C.J. Sensitivity of antiendomysium and antigliadin antibodies in untreated celiac disease: Disappointing in clinical practice. Am. J. Gastroenterol. 1999, 94, 888.
- Catassi, C.; Fasano, A. Celiac disease. In Gluten-Free Cereal Products and Beverages; Academic Press: Cambridge, MA, USA, 2008; p. 1-I.
- Singh, P.; Arora, A.; Strand, T.A.; Leffler, D.A.; Catassi, C.; Green, P.H.; Kelly, C.P.; Ahuja, V.; Makharia, G.K. Global prevalence of celiac disease: Systematic review and meta-analysis. Clin. Gastroenterol. Hepatol. 2018, 16, 823–836.e2.
- Tanveer, M.; Ahmed, A. Non-Celiac Gluten Sensitivity: A Systematic Review. J. Coll. Physicians Surg. Pak. 2019, 29, 51–57.
- Rubio-Tapia, A.; Ludvigsson, J.F.; Brantner, T.L.; Murray, J.A.; Everhart, J.E. The prevalence of celiac disease in the United States. Am. J. Gastroenterol. 2012, 107, 1538.
- Barada, K.; Bitar, A.; Mokadem, M.A.-R.; Hashash, J.G.; Green, P. Celiac disease in Middle Eastern and North African countries: A new burden? World J. Gastroenterol. WJG 2010, 16, 1449.
- Peña, A.S.; Rodrigo, L. Epidemiology of celiac disease and non-celiac gluten-related disorders. In Advances in the Understanding of Gluten Related Pathology and the Evolution of Gluten-Free Foods; OmniaScience: Barcelona, Spain, 2015; pp. 27–73.
- Malekzadeh, R.; Sachdev, A.; Ali, A.F. Coeliac disease in developing countries: Middle East, India and North Africa. Best Pract. Res. Clin. Gastroenterol. 2005, 19, 351–358.
- Ramakrishna, B.; Makharia, G.K.; Chetri, K.; Dutta, S.; Mathur, P.; Ahuja, V.; Amarchand, R.; Balamurugan, R.; Chowdhury, S.D.; Daniel, D. Prevalence of adult celiac disease in India: Regional variations and associations. Am. J. Gastroenterol. 2016, 111, 115.
- Makharia, G.K.; Verma, A.K.; Amarchand, R.; Bhatnagar, S.; Das, P.; Goswami, A.; Bhatia, V.; Ahuja, V.; Datta Gupta, S.; Anand, K. Prevalence of celiac disease in the northern part of India: A community based study. J. Gastroenterol. Hepatol. 2011, 26, 894–900.
- Rajpoot, P.; Makharia, G. Problems and challenges to adaptation of gluten free diet by Indian patients with celiac disease. Nutrients 2013, 5, 4869–4879.
- Wu, J.; Xia, B.; von Blomberg, B.; Zhao, C.; Yang, X.; Crusius, J.; Peña, A. Coeliac disease: Emerging in China? Gut 2010, 59, 418–419.
- Yuan, J.; Zhou, C.; Gao, J.; Li, J.; Yu, F.; Lu, J.; Li, X.; Wang, X.; Tong, P.; Wu, Z. Prevalence of celiac disease autoimmunity among adolescents and young adults in China. Clin. Gastroenterol. Hepatol. 2017, 15, 1572–1579.e1.
- Reese, I.; Schäfer, C.; Kleine-Tebbe, J.; Ahrens, B.; Bachmann, O.; Ballmer-Weber, B.; Beyer, K.; Bischoff, S.C.; Blümchen, K.; Dölle, S.; et al. Non-celiac gluten/wheat sensitivity (NCGS)—A currently undefined disorder without validated diagnostic criteria and of unknown prevalence. Allergo J. Int. 2018, 27, 147–151.
- Allred, L.K.; Kupper, C.; Quinn, C. The Use of Visual Examination for Determining the Presence of Gluten-Containing Grains in Gluten Free Oats and Other Grains, Seeds, Beans, Pulses, and Legumes. J. AOAC Int. 2018, 101, 36–44.
- Bustamante, M.; Fernández-Gil, M.; Churruca, I.; Miranda, J.; Lasa, A.; Navarro, V.; Simón, E. Evolution of gluten content in cereal-based gluten-free products: An overview from 1998 to 2016. Nutrients 2017, 9, 21.
- Sharma, G.M.; Pereira, M.; Williams, K.M. Gluten detection in foods available in the United States–A market survey. Food Chem. 2015, 169, 120–126.
- Miranda, J.; Simón, E. Gluten Content Change Over the Two Last Decades. In Nutritional and Analytical Approaches of Gluten-Free Diet in Celiac Disease; Springer: Cham, Switzerland, 2017; pp. 47–57.
- Moreno, M.D.; Rodríguez-Herrera, A.; Sousa, C.; Comino, I. Biomarkers to monitor gluten-free diet compliance in celiac patients. Nutrients 2017, 9, 46.
- Scherf, K.A.; Poms, R.E. Recent developments in analytical methods for tracing gluten. J. Cereal Sci. 2016, 67, 112–122.
- Codex Alimentarius Commission. Codex Standard 234-1999 (amended 2011), Recommended methods of analysis and sampling. Gluten-free foods: Enzyme-linked immunoassay R5 Mendez (ELISA) method. In Codex Alimentarious; FAO: Rome, Italy; WHO: Geneva, Switzerland, 2019; pp. 1–80.
- Ellis, H.; Doyle, A.; Wieser, H.; Sturgess, R.; Day, P.; Ciclitira, P. Measurement of gluten using a monoclonal antibody to a sequenced peptide of α-gliadin from the coeliac-activating domain I. J. Biochem. Biophys. Methods 1994, 28, 77–82.
- Rumbo, M.; Chirdo, F.G.; Fossati, C.A.; Añón, M.C. Analysis of the effects of heat treatment on gliadin immunochemical quantification using a panel of anti-prolamin antibodies. J. Agric. Food Chem. 2001, 49, 5719–5726.
- Doña, V.; Fossati, C.; Chirdo, F. Interference of denaturing and reducing agents on the antigen/antibody interaction. Impact on the performance of quantitative immunoassays in gliadin analysis. Eur. Food Res. Technol. 2008, 226, 591–602.
- Song, K.-M.; Lee, S.; Ban, C. Aptamers and their biological applications. Sensors 2012, 12, 612–631.
- Berezovski, M.V.; Musheev, M.U.; Drabovich, A.P.; Jitkova, J.V.; Krylov, S.N. Non-SELEX: Selection of aptamers without intermediate amplification of candidate oligonucleotides. Nat. Protoc. 2006, 1, 1359.
- Stoltenburg, R.; Reinemann, C.; Strehlitz, B. SELEX—a (r) evolutionary method to generate high-affinity nucleic acid ligands. Biomol. Eng. 2007, 24, 381–403.
- Zhou, J.; Battig, M.R.; Wang, Y. Aptamer-based molecular recognition for biosensor development. Anal. Bioanal. Chem. 2010, 398, 2471–2480.
- Van Dorst, B.; Mehta, J.; Bekaert, K.; Rouah-Martin, E.; De Coen, W.; Dubruel, P.; Blust, R.; Robbens, J. Recent advances in recognition elements of food and environmental biosensors: A review. Biosens. Bioelectron. 2010, 26, 1178–1194.
- Nimjee, S.M.; Rusconi, C.P.; Sullenger, B.A. Aptamers: An emerging class of therapeutics. Annu. Rev. Med. 2005, 56, 555–583.
- Banerjee, J.; Nilsen-Hamilton, M. Aptamers: Multifunctional molecules for biomedical research. J. Mol. Med. 2013, 91, 1333–1342.
- Strehlitz, B.; Nikolaus, N.; Stoltenburg, R. Protein detection with aptamer biosensors. Sensors 2008, 8, 4296–4307.
- Robertson, D.L.; Joyce, G.F. Selection in vitro of an RNA enzyme that specifically cleaves single-stranded DNA. Nature 1990, 344, 467.
- Tombelli, S.; Mascini, M. Aptamers as molecular tools for bioanalytical methods. Curr. Opin. Mol. Ther. 2009, 11, 179–188.
- Jenison, R.D.; Gill, S.C.; Pardi, A.; Polisky, B. High-resolution molecular discrimination by RNA. Science 1994, 263, 1425–1429.
- Ellington, A.D.; Szostak, J.W. In vitro selection of RNA molecules that bind specific ligands. Nature 1990, 346, 818.
- Jayasena, S.D. Aptamers: An emerging class of molecules that rival antibodies in diagnostics. Clin. Chem. 1999, 45, 1628–1650.
- Wang, J.; Meng, W.; Zheng, X.; Liu, S.; Li, G. Combination of aptamer with gold nanoparticles for electrochemical signal amplification: Application to sensitive detection of platelet-derived growth factor. Biosens. Bioelectron. 2009, 24, 1598–1602.




