
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Marco Contardi | + 1126 word(s) | 1126 | 2021-07-05 05:17:01 | | | |
| 2 | Peter Tang | Meta information modification | 1126 | 2021-07-06 09:39:08 | | |
Video Upload Options
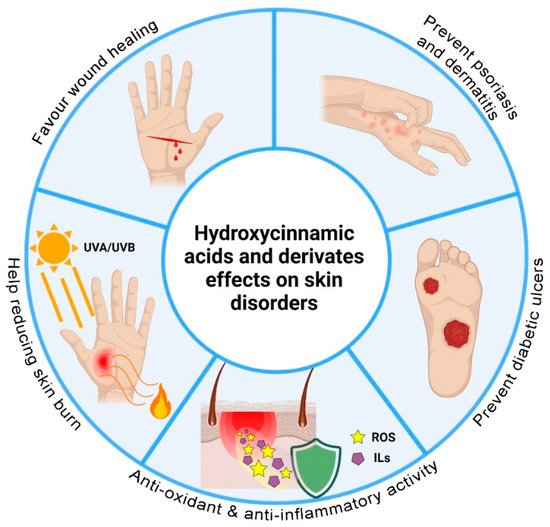
Alterations of skin homeostasis are widely diffused in our everyday life both due to accidental injuries, such as wounds and burns, and physiological conditions, such as late-stage diabetes, dermatitis, or psoriasis. These events are locally characterized by an intense inflammatory response, a high generation of harmful free radicals, or an impairment in the immune response regulation, which can profoundly change the skin tissue’ repair process, vulnerability, and functionality. Moreover, diabetes diffusion, antibiotic resistance, and abuse of aggressive soaps and disinfectants following the COVID-19 emergency could be causes for the future spreading of skin disorders. In the last years, hydroxycinnamic acids and derivatives have been investigated and applied in several research fields for their anti-oxidant, anti-inflammatory, and anti-bacterial activities.
1. Introduction
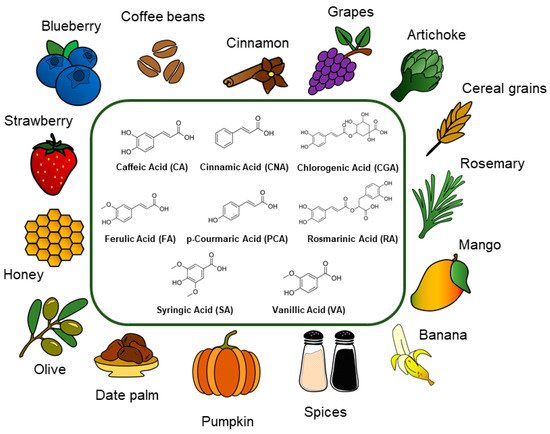
2. Hydroxycinnamic Acids and Derivatives: General Uses and Current Applications
3. Hydroxycinnamic Acids and Derivatives: Activities on Skin Disorders
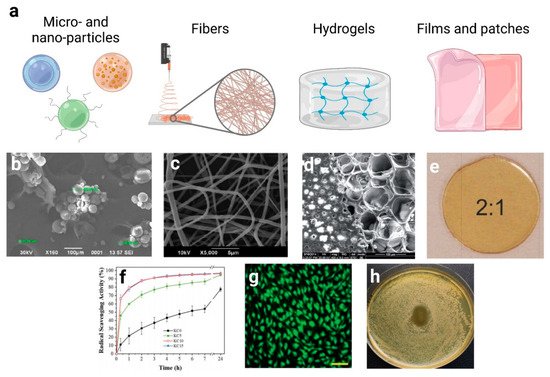
4. Hydroxycinnamic Acids and Derivatives: Advanced Formulations
References
- Suarato, G.; Bertorelli, R.; Athanassiou, A. Borrowing from Nature: Biopolymers and biocomposites as smart wound care materials. Front. Bioeng. Biotechnol. 2018, 6, 137.
- Raghav, A.; Khan, Z.A.; Labala, R.K.; Ahmad, J.; Noor, S.; Mishra, B.K. Financial burden of diabetic foot ulcers to world: A progressive topic to discuss always. Ther. Adv. Endocrinol. Metab. 2018, 9, 29–31.
- Thomsen, S.F.; Skov, L.; Dodge, R.; Hedegaard, M.S.; Kjellberg, J. Socioeconomic costs and health inequalities from psoriasis: A cohort study. Dermatology 2019, 235, 372–379.
- Drucker, A.M.; Wang, A.R.; Li, W.-Q.; Sevetson, E.; Block, J.K.; Qureshi, A.A. The burden of atopic dermatitis: Summary of a report for the National Eczema Association. J. Investig. Dermatol. 2017, 137, 26–30.
- Chandra, A.; Ray, A.; Senapati, S.; Chatterjee, R. Genetic and epigenetic basis of psoriasis pathogenesis. Mol. Immunol. 2015, 64, 313–323.
- Frykberg, R.G.; Banks, J. Challenges in the treatment of chronic wounds. Adv. Wound Care 2015, 4, 560–582.
- Kaddoura, I.; Abu-Sittah, G.; Ibrahim, A.; Karamanoukian, R.; Papazian, N. Burn injury: Review of pathophysiology and therapeutic modalities in major burns. Ann. Burn. Fire Disasters 2017, 30, 95.
- Hall, T.J.; Villapún, V.M.; Addison, O.; Webber, M.A.; Lowther, M.; Louth, S.E.; Mountcastle, S.E.; Brunet, M.Y.; Cox, S.C. A call for action to the biomaterial community to tackle antimicrobial resistance. Biomater. Sci. 2020, 8, 4951–4974.
- Ejtahed, H.-S.; Hasani-Ranjbar, S.; Siadat, S.D.; Larijani, B. The most important challenges ahead of microbiome pattern in the post era of the COVID-19 pandemic. J. Diabetes Metab. Disord. 2020, 19, 2031–2033.
- Proksch, E. pH in nature, humans and skin. J. Dermatol. 2018, 45, 1044–1052.
- Da Silva, N.T.; Quintana, H.T.; Bortolin, J.A.; Ribeiro, D.A.; de Oliveira, F. Burn injury induces skeletal muscle degeneration, inflammatory host response, and oxidative stress in wistar rats. J. Burn. Care Res. 2015, 36, 428–433.
- Dunnill, C.; Patton, T.; Brennan, J.; Barrett, J.; Dryden, M.; Cooke, J.; Leaper, D.; Georgopoulos, N.T. Reactive oxygen species (ROS) and wound healing: The functional role of ROS and emerging ROS-modulating technologies for augmentation of the healing process. Int. Wound J. 2017, 14, 89–96.
- Todke, P.; Shah, V.H. Psoriasis: Implication to disease and therapeutic strategies, with an emphasis on drug delivery approaches. Int. J. Dermatol. 2018, 57, 1387–1402.
- Hon, K.L.; Leung, A.K.; Barankin, B. Barrier repair therapy in atopic dermatitis: An overview. Am. J. Clin. Dermatol. 2013, 14, 389–399.
- Nobili, S.; Lippi, D.; Witort, E.; Donnini, M.; Bausi, L.; Mini, E.; Capaccioli, S. Natural compounds for cancer treatment and prevention. Pharmacol. Res. 2009, 59, 365–378.
- Tundis, R.; Loizzo, M.; Bonesi, M.; Menichini, F. Potential role of natural compounds against skin aging. Curr. Med. Chem. 2015, 22, 1515–1538.
- Marrelli, M.; Menichini, G.; Provenzano, E.; Conforti, F. Applications of natural compounds in the photodynamic therapy of skin cancer. Curr. Med. Chem. 2014, 21, 1371–1390.
- Sychrová, A.; Koláriková, I.; Žemlička, M.; Šmejkal, K. Natural compounds with dual antimicrobial and anti-inflammatory effects. Phytochem. Rev. 2020, 19, 1471–1502.
- Yong, H.; Liu, Y.; Yun, D.; Zong, S.; Jin, C.; Liu, J. Chitosan films functionalized with different hydroxycinnamic acids: Preparation, characterization and application for pork preservation. Foods 2021, 10, 536.
- Taofiq, O.; González-Paramás, A.M.; Barreiro, M.F.; Ferreira, I.C. Hydroxycinnamic acids and their derivatives: Cosmeceutical significance, challenges and future perspectives, a review. Molecules 2017, 22, 281.
- Ou, S.; Kwok, K.C. Ferulic acid: Pharmaceutical functions, preparation and applications in foods. J. Sci. Food Agric. 2004, 84, 1261–1269.
- Adefegha, S.A. Functional foods and nutraceuticals as dietary intervention in chronic diseases; novel perspectives for health promotion and disease prevention. J. Diet. Suppl. 2018, 15, 977–1009.
- Wu, D.; Zhou, J.; Creyer, M.N.; Yim, W.; Chen, Z.; Messersmith, P.B.; Jokerst, J.V. Phenolic-enabled nanotechnology: Versatile particle engineering for biomedicine. Chem. Soc. Rev. 2021, 50, 4432–4483.
- Li, Z.; Jiang, H.; Xu, C.; Gu, L. A review: Using nanoparticles to enhance absorption and bioavailability of phenolic phytochemicals. Food Hydrocoll. 2015, 43, 153–164.
- Peña-Torres, E.F.; González-Ríos, H.; Avendaño-Reyes, L.; Valenzuela-Grijalva, N.V.; Pinelli-Saavedra, A.; Muhlia-Almazán, A.; Peña-Ramos, E.A. Hydroxycinnamic acids in animal production: Pharmacokinetics, pharmacodynamics and growth promoting effects. Review. Rev. Mex. Cienc. Pecu. 2019, 10, 391–415.
- El-Seedi, H.R.; El-Said, A.M.; Khalifa, S.A.; Goransson, U.; Bohlin, L.; Borg-Karlson, A.-K.; Verpoorte, R. Biosynthesis, natural sources, dietary intake, pharmacokinetic properties, and biological activities of hydroxycinnamic acids. J. Agric. Food Chem. 2012, 60, 10877–10895.
- Coman, V.; Vodnar, D.C. Hydroxycinnamic acids and human health: Recent advances. J. Sci. Food Agric. 2020, 100, 483–499.
- Singh, A.; Srivastava, N.; Yadav, K.S.; Sinha, P.; Yadav, N.P. Preparation, optimization, characterization and bioevaluation of rosmarinic acid loaded phytovesicles for anti-inflammatory activity. J. Drug Deliv. Sci. Technol. 2020, 59, 101888.
- Kossyvaki, D.; Suarato, G.; Summa, M.; Gennari, A.; Francini, N.; Gounaki, I.; Venieri, D.; Tirelli, N.; Bertorelli, R.; Athanassiou, A. Keratin–cinnamon essential oil biocomposite fibrous patches for skin burn care. Mater. Adv. 2020, 1, 1805–1816.
- Raja, S.T.K.; Thiruselvi, T.; Aravindhan, R.; Mandal, A.B.; Gnanamani, A. In vitro and in vivo assessments of a 3-(3, 4-dihydroxyphenyl)-2-propenoic acid bioconjugated gelatin-based injectable hydrogel for biomedical applications. J. Mater. Chem. B 2015, 3, 1230–1244.
- Contardi, M.; Heredia-Guerrero, J.A.; Guzman-Puyol, S.; Summa, M.; BENITEZ, J.J.; Goldoni, L.; Caputo, G.; Cusimano, G.; Picone, P.; Di Carlo, M. Combining Dietary Phenolic Antioxidants with Polyvinylpyrrolidone: Transparent Biopolymer Films based on p-Coumaric Acid for Controlled Release. J. Mater. Chem. B 2019, 7, 1384–1396.




