
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Christian Bailly | + 3123 word(s) | 3123 | 2021-06-21 07:50:09 | | | |
| 2 | Vivi Li | Meta information modification | 3123 | 2021-06-30 04:42:26 | | |
Video Upload Options
The interaction of programmed cell death ligand-1 (PDL1) with its receptor PD1 inhibits T-cell responses. Blockade of this interaction with monoclonal antibodies leads to major antitumor effects. However, not all cancer patients respond well to anti-(PD-1/PD-L1) immunotherapy. The PD-L1 protein is expressed at the cell plasma membrane (mPD-L1), at the surface of exosomes (exoPD-L1), in cell nuclei (nPD-L1) and as a soluble circulating protein (sPD-L1). The aim of our analysis was to highlight the multiple variants of sPD-L1 generated either by the proteolytic cleavage of m/exoPD-L1 or by the alternative splicing of PD-L1 pre-mRNA. The objective was also to underline the presence and role of circulating sPD-L1 isoforms in multiple cancer indications and many other diseases (including chronic inflammatory and viral diseases), and under non-pathological conditions (pregnancy). sPD-L1 often represents a general marker of an inflammatory status. The pool of sPD-L1 proteins is an integral part of the highly dynamic PD-1/PD-L1 signaling pathway.
1. The PD-1/PD-L1 Checkpoint
1.1. Membrane PD-L1
1.2. Exosomal PD-L1
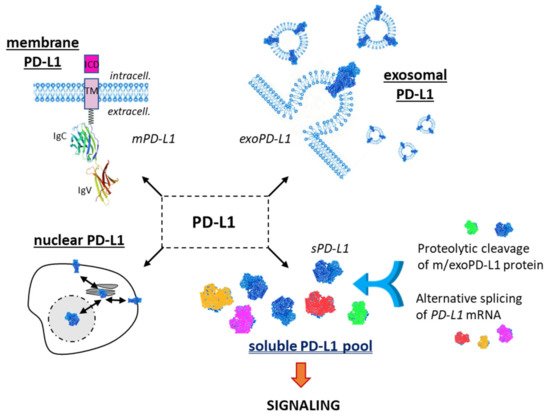
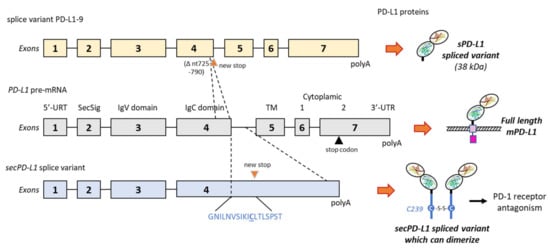
1.3. Soluble PD-L1
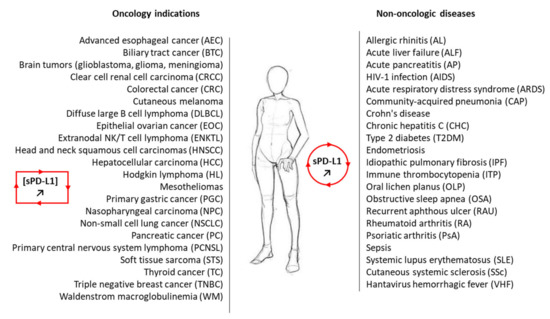
2. Significance of Soluble PD-L1 in Cancer
2.1. sPD-L1 as a Cancer Biomarker
2.2. Functionality of sPD-L1 in Cancer
References
- Ghosh, C.; Luong, G.; Sun, Y. A snapshot of the PD-1/PD-L1 pathway. J. Cancer 2021, 12, 2735–2746.
- Zam, W.; Ali, L. Immune checkpoint inhibitors in the treatment of cancer. Curr. Clin. Pharmacol. 2021.
- Persico, P.; Lorenzi, E.; Dipasquale, A.; Pessina, F.; Navarria, P.; Politi, L.S.; Santoro, A.; Simonelli, M. Checkpoint inhibitors as high-grade gliomas treatment: State of the art and future perspectives. J. Clin. Med. 2021, 10, 1367.
- Jimbu, L.; Mesaros, O.; Popescu, C.; Neaga, A.; Berceanu, I.; Dima, D.; Gaman, M.; Zdrenghea, M. Is there a place for PD-1-PD-L blockade in acute myeloid leukemia? Pharmaceuticals 2021, 14, 288.
- Bailly, C.; Thuru, X.; Quesnel, B. Combined cytotoxic chemotherapy and immunotherapy of cancer: Modern times. NAR Cancer 2020, 2, zcaa002.
- Chen, Y.; Liu, C.; Zhu, S.; Liang, X.; Zhang, Q.; Luo, X.; Yuan, L.; Song, L. PD-1/PD-L1 immune checkpoint blockade-based combinational treatment: Immunotherapeutic amplification strategies against colorectal cancer. Int. Immunopharmacol. 2021, 96, 107607.
- Sato, K.; Uehara, T.; Nakajima, T.; Iwaya, M.; Miyagawa, Y.; Watanabe, T.; Ota, H. Inverse correlation between PD-L1 expression and LGR5 expression in tumor budding of stage II/III colorectal cancer. Ann. Diagn. Pathol. 2021, 52, 151739.
- Seliger, B. Basis of PD1/PD-L1 Therapies. J. Clin. Med. 2019, 8, 2168.
- Qin, W.; Hu, L.; Zhang, X.; Jiang, S.; Li, J.; Zhang, Z.; Wang, X. The Diverse Function of PD-1/PD-L Pathway Beyond Cancer. Front. Immunol. 2019, 10, 2298.
- Meggyes, M.; Nagy, D.U.; Szereday, L. Investigation of the PD-1 and PD-L1 immune checkpoint molecules throughout healthy human pregnancy and in nonpregnant women. J. Clin. Med. 2020, 9, 2536.
- Dall’Olio, F.G.; Gelsomino, F.; Conci, N.; Marcolin, L.; De Giglio, A.; Grilli, G.; Sperandi, F.; Fontana, F.; Terracciano, M.; Fragomeno, B.; et al. PD-L1 expression in circulating tumor cells as a promising prognostic biomarker in advanced non-small-cell lung cancer treated with immune checkpoint inhibitors. Clin. Lung Cancer 2021.
- Ntzifa, A.; Strati, A.; Kallergi, G.; Kotsakis, A.; Georgoulias, V.; Lianidou, E. Gene expression in circulating tumor cells reveals a dynamic role of EMT and PD-L1 during osimertinib treatment in NSCLC patients. Sci. Rep. 2021, 11, 2313.
- Zhang, L.; Zhang, X.; Liu, Y.; Zhang, T.; Wang, Z.; Gu, M.; Li, Y.; Wang, D.D.; Li, W.; Lin, P.P. PD-L1+ aneuploid circulating tumor endothelial cells (CTECs) exhibit resistance to the checkpoint blockade immunotherapy in advanced NSCLC patients. Cancer Lett. 2020, 469, 355–366.
- Wang, W.; Zhang, T. Expression and analysis of PD-L1 in peripheral blood circulating tumor cells of lung cancer. Future Oncol. 2021, 17, 1625–1635.
- Kloten, V.; Lampignano, R.; Krahn, T.; Schlange, T. Circulating Tumor Cell PD-L1 expression as biomarker for therapeutic efficacy of immune checkpoint inhibition in NSCLC. Cells 2019, 8, 809.
- Zhang, L.; Yu, D. Exosomes in cancer development, metastasis, and immunity. Biochim. Biophys. Acta Rev. Cancer 2019, 1871, 455–468.
- Chen, G.; Huang, A.C.; Zhang, W.; Zhang, G.; Wu, M.; Xu, W.; Yu, Z.; Yang, J.; Wang, B.; Sun, H.; et al. Exosomal PD-L1 contributes to immunosuppression and is associated with anti-PD-1 response. Nature 2018, 560, 382–386.
- Su, D.; Tsai, H.I.; Xu, Z.; Yan, F.; Wu, Y.; Xiao, Y.; Liu, X.; Wu, Y.; Parvanian, S.; Zhu, W.; et al. Exosomal PD-L1 functions as an immunosuppressant to promote wound healing. J. Extracell Vesicles 2019, 9, 1709262.
- Xie, F.; Xu, M.; Lu, J.; Mao, L.; Wang, S. The role of exosomal PD-L1 in tumor progression and immunotherapy. Mol. Cancer 2019, 18, 146.
- Lawler, S.E.; Nowicki, M.O.; Ricklefs, F.L.; Chiocca, E.A. Immune escape mediated by exosomal PD-L1 in cancer. Adv. Biosyst. 2020, 4, e2000017.
- Tang, Y.; Zhang, P.; Wang, Y.; Wang, J.; Su, M.; Wang, Y.; Zhou, L.; Zhou, J.; Xiong, W.; Zeng, Z.; et al. The biogenesis, biology, and clinical significance of exosomal PD-L1 in cancer. Front. Immunol. 2020, 11, 604.
- Zhou, K.; Guo, S.; Li, F.; Sun, Q.; Liang, G. Exosomal PD-L1: New insights into tumor immune escape mechanisms and therapeutic strategies. Front. Cell. Dev. Biol. 2020, 8, 569219.
- Morrissey, S.M.; Yan, J. Exosomal PD-L1: Roles in tumor progression and Immunotherapy. Trends Cancer 2020, 6, 550–558.
- Liang, B.; Hu, X.; Ding, Y.; Liu, M. Tumor-derived exosomes in the PD-1/PD-L1 axis: Significant regulators as well as promising clinical targets. J. Cell Physiol. 2021, 236, 4138–4151.
- Yin, Z.; Yu, M.; Ma, T.; Zhang, C.; Huang, S.; Karimzadeh, M.R.; Momtazi-Borojeni, A.A.; Chen, S. Mechanisms underlying low-clinical responses to PD-1/PD-L1 blocking antibodies in immunotherapy of cancer: A key role of exosomal PD-L1. J. Immunother. Cancer 2021, 9, e001698.
- Liu, S.; Zhu, Y.; Zhang, C.; Meng, X.; Sun, B.; Zhang, G.; Fan, Y.; Kang, X. The clinical significance of soluble programmed cell death-ligand 1 (sPD-L1) in patients with gliomas. Front. Oncol. 2020, 10, 9.
- Jovanovic, D.; Roksandic Milenkovic, M.; Kotur Stevuljevic, J.; Markovic, J.; Ceriman, V.; Kontic, M.; Skodric Trifunovic, V. Membrane PD-L1 expression and soluble PD-L1 plasma levels in idiopathic pulmonary fibrosis-a pilot study. J. Thorac. Dis. 2018, 10, 6660–6669.
- Aguirre, J.E.; Beswick, E.J.; Grim, C.; Uribe, G.; Tafoya, M.; Chacon Palma, G.; Samedi, V.; McKee, R.; Villeger, R.; Fofanov, Y.; et al. Matrix metalloproteinases cleave membrane-bound PD-L1 on CD90+ (myo-)fibroblasts in Crohn’s disease and regulate Th1/Th17 cell responses. Int. Immunol. 2020, 32, 57–68.
- Okuyama, M.; Mezawa, H.; Kawai, T.; Urashima, M. Elevated Soluble PD-L1 in pregnant women’s serum suppresses the immune reaction. Front. Immunol. 2019, 10, 86.
- Khan, M.; Zhao, Z.; Arooj, S.; Fu, Y.; Liao, G. Soluble PD-1: Predictive, prognostic, and therapeutic value for cancer immunotherapy. Front. Immunol. 2020, 11, 587460.
- Wuethrich, A.; Rajkumar, A.R.; Shanmugasundaram, K.B.; Reza, K.K.; Dey, S.; Howard, C.B.; Sina, A.A.; Trau, M. Single droplet detection of immune checkpoints on a multiplexed electrohydrodynamic biosensor. Analyst 2019, 144, 6914–6921.
- Goto, M.; Chamoto, K.; Higuchi, K.; Yamashita, S.; Noda, K.; Iino, T.; Miura, M.; Yamasaki, T.; Ogawa, O.; Sonobe, M.; et al. Analytical performance of a new automated chemiluminescent magnetic immunoassays for soluble PD-1, PD-L1, and CTLA-4 in human plasma. Sci. Rep. 2019, 9, 10144.
- Reza, K.K.; Sina, A.A.; Wuethrich, A.; Grewal, Y.S.; Howard, C.B.; Korbie, D.; Trau, M. A SERS microfluidic platform for targeting multiple soluble immune checkpoints. Biosens. Bioelectron. 2019, 126, 178–186.
- Luo, B.; Wang, Y.; Lu, H.; Wu, S.; Lu, Y.; Shi, S.; Li, L.; Jiang, S.; Zhao, M. Label-free and specific detection of soluble programmed death ligand-1 using a localized surface plasmon resonance biosensor based on excessively tilted fiber gratings. Biomed. Opt. Express. 2019, 10, 5136–5148.
- Abu Hejleh, T.; Furqan, M.; Ballas, Z.; Clamon, G. The clinical significance of soluble PD-1 and PD-L1 in lung cancer. Crit. Rev. Oncol. Hematol. 2019, 143, 148–152.
- Theodoraki, M.N.; Yerneni, S.S.; Hoffmann, T.K.; Gooding, W.E.; Whiteside, T.L. Clinical significance of PD-L1(+) exosomes in plasma of head and neck cancer patients. Clin. Cancer Res. 2018, 24, 896–905.
- Yang, Q.; Chen, M.; Gu, J.; Niu, K.; Zhao, X.; Zheng, L.; Xu, Z.; Yu, Y.; Li, F.; Meng, L.; et al. Novel biomarkers of dynamic blood PD-L1 expression for immune checkpoint inhibitors in advanced non-small-cell lung cancer patients. Front. Immunol. 2021, 12, 665133.
- Tu, X.; Qin, B.; Zhang, Y.; Zhang, C.; Kahila, M.; Nowsheen, S.; Yin, P.; Yuan, J.; Pei, H.; Li, H.; et al. PD-L1 (B7-H1) Competes with the RNA exosome to regulate the DNA damage response and can be targeted to sensitize to radiation or chemotherapy. Mol. Cell 2019, 74, 1215–1226.
- Yu, J.; Qin, B.; Moyer, A.M.; Nowsheen, S.; Tu, X.; Dong, H.; Boughey, J.C.; Goetz, M.P.; Weinshilboum, R.; Lou, Z.; et al. Regulation of sister chromatid cohesion by nuclear PD-L1. Cell Res. 2020, 30, 590–601.
- Du, W.; Zhu, J.; Zeng, Y.; Liu, T.; Zhang, Y.; Cai, T.; Fu, Y.; Zhang, W.; Zhang, R.; Liu, Z.; et al. KPNB1-mediated nuclear translocation of PD-L1 promotes non-small cell lung cancer cell proliferation via the Gas6/MerTK signaling pathway. Cell Death Differ. 2021, 28, 1284–1300.
- Yoshida, J.; Ishikawa, T.; Doi, T.; Ota, T.; Yasuda, T.; Okayama, T.; Sakamoto, N.; Inoue, K.; Dohi, O.; Yoshida, N.; et al. Clinical significance of soluble forms of immune checkpoint molecules in advanced esophageal cancer. Med. Oncol. 2019, 36, 60.
- Kim, J.W.; Lee, K.H.; Kim, J.W.; Suh, K.J.; Nam, A.R.; Bang, J.H.; Jin, M.H.; Oh, K.S.; Kim, J.M.; Kim, T.Y.; et al. The prognostic role of soluble transforming growth factor-beta and its correlation with soluble programmed death-ligand 1 in biliary tract cancer. Liver Int. 2021, 41, 388–395.
- Ha, H.; Nam, A.R.; Bang, J.H.; Park, J.E.; Kim, T.Y.; Lee, K.H.; Han, S.W.; Im, S.A.; Kim, T.Y.; Bang, Y.J.; et al. Soluble programmed death-ligand 1 (sPDL1) and neutrophil-to-lymphocyte ratio (NLR) predicts survival in advanced biliary tract cancer patients treated with palliative chemotherapy. Oncotarget 2016, 7, 76604–76612.
- Mair, M.J.; Pajenda, S.; Ilhan-Mutlu, A.; Steindl, A.; Kiesel, B.; Widhalm, G.; Dieckmann, K.; Feldmann, K.; Hainfellner, J.; Marosi, C.; et al. Soluble PD-L1 is associated with local and systemic inflammation markers in primary and secondary brain tumours. ESMO Open 2020, 5, e000863.
- Larrinaga, G.; Solano-Iturri, J.D.; Errarte, P.; Unda, M.; Loizaga-Iriarte, A.; Pérez-Fernández, A.; Echevarría, E.; Asumendi, A.; Manini, C.; Angulo, J.C.; et al. Soluble PD-L1 is an independent prognostic factor in clear cell renal cell carcinoma. Cancers 2021, 13, 667.
- Montemagno, C.; Hagege, A.; Borchiellini, D.; Thamphya, B.; Rastoin, O.; Ambrosetti, D.; Iovanna, J.; Rioux-Leclercq, N.; Porta, C.; Negrier, S.; et al. Soluble forms of PD-L1 and PD-1 as prognostic and predictive markers of sunitinib efficacy in patients with metastatic clear cell renal cell carcinoma. Oncoimmunology 2020, 9, 1846901.
- Omura, Y.; Toiyama, Y.; Okugawa, Y.; Yin, C.; Shigemori, T.; Kusunoki, K.; Kusunoki, Y.; Ide, S.; Shimura, T.; Fujikawa, H.; et al. Prognostic impacts of tumoral expression and serum levels of PD-L1 and CTLA-4 in colorectal cancer patients. Cancer Immunol. Immunother. 2020, 69, 2533–2546.
- Zhou, J.; Mahoney, K.M.; Giobbie-Hurder, A.; Zhao, F.; Lee, S.; Liao, X.; Rodig, S.; Li, J.; Wu, X.; Butterfield, L.H.; et al. Soluble PD-L1 as a biomarker in malignant melanoma treated with checkpoint blockade. Cancer Immunol. Res. 2017, 5, 480–492.
- Fei, Y.; Yu, J.; Li, Y.; Li, L.; Zhou, S.; Zhang, T.; Li, L.; Qiu, L.; Meng, B.; Pan, Y.; et al. Plasma soluble PD-L1 and STAT3 predict the prognosis in diffuse large B cell lymphoma patients. J. Cancer 2020, 11, 7001–7008.
- Cho, I.; Lee, H.; Yoon, S.E.; Ryu, K.J.; Ko, Y.H.; Kim, W.S.; Kim, S.J. Serum levels of soluble programmed death-ligand 1 (sPD-L1) in patients with primary central nervous system diffuse large B-cell lymphoma. BMC Cancer 2020, 20, 120.
- Buderath, P.; Schwich, E.; Jensen, C.; Horn, P.A.; Kimmig, R.; Kasimir-Bauer, S.; Rebmann, V. Soluble programmed death receptor ligands sPD-L1 and sPD-L2 as liquid biopsy markers for prognosis and platinum response in epithelial ovarian cancer. Front. Oncol. 2019, 9, 1015.
- Koukourakis, M.I.; Kontomanolis, E.; Sotiropoulou, M.; Mitrakas, A.; Dafa, E.; Pouliliou, S.; Sivridis, E.; Giatromanolaki, A. Increased soluble PD-L1 levels in the plasma of patients with epithelial ovarian cancer correlate with plasma levels of miR34a and miR200. Anticancer Res. 2018, 38, 5739–5745.
- Li, J.W.; Wei, P.; Guo, Y.; Shi, D.; Yu, B.H.; Su, Y.F.; Li, X.Q.; Zhou, X.Y. Clinical significance of circulating exosomal PD-L1 and soluble PD-L1 in extranodal NK/T-cell lymphoma, nasal-type. Am. J. Cancer Res. 2020, 10, 4498–4512.
- Feng, Y.; Jing, C.; Yu, X.; Cao, X.; Xu, C. Predicting treatment response of patients with extranodal natural killer/T-cell lymphoma based on levels of PD-L1 mRNA and soluble PD-L1. Hematol. Oncol. 2020, 38, 467–477.
- Mocan, T.; Ilies, M.; Nenu, I.; Craciun, R.; Horhat, A.; Susa, R.; Minciuna, I.; Rusu, I.; Mocan, L.P.; Seicean, A.; et al. Serum levels of soluble programmed death-ligand 1 (sPD-L1): A possible biomarker in predicting post-treatment outcomes in patients with early hepatocellular carcinoma. Int. Immunopharmacol. 2021, 94, 107467.
- Li, X.S.; Li, J.W.; Li, H.; Jiang, T. Prognostic value of programmed cell death ligand 1 (PD-L1) for hepatocellular carcinoma: A meta-analysis. Biosci. Rep. 2020, 40, BSR20200459.
- El-Gebaly, F.; Abou-Saif, S.; Elkadeem, M.; Helmy, A.; Abd-Elsalam, S.; Yousef, M.; Elkhouly, R.A.; Amer, I.F.; El-Demerdash, T. Study of serum soluble programmed death ligand 1 as a prognostic factor in hepatocellular carcinoma in Egyptian patients. Curr. Cancer Drug Targets. 2019, 19, 896–905.
- Veldman, J.; Alsada, Z.N.D.; van den Berg, A.; Plattel, W.J.; Diepstra, A.; Visser, L. Soluble PD-L1 is a promising disease biomarker but does not reflect tissue expression in classic Hodgkin lymphoma. Br. J. Haematol. 2021, 193, 506–514.
- Chiarucci, C.; Cannito, S.; Daffinà, M.G.; Amato, G.; Giacobini, G.; Cutaia, O.; Lofiego, M.F.; Fazio, C.; Giannarelli, D.; Danielli, R.; et al. Circulating levels of PD-L1 in mesothelioma patients from the NIBIT-MESO-1 study: Correlation with survival. Cancers 2020, 12, 361.
- Carosio, R.; Fontana, V.; Mastracci, L.; Ferro, P.; Grillo, F.; Banelli, B.; Canessa, P.A.; Dessanti, P.; Vigani, A.; Morabito, A.; et al. Characterization of soluble PD-L1 in pleural effusions of mesothelioma patients: Potential implications in the immune response and prognosis. J. Cancer Res. Clin. Oncol. 2021, 147, 459–468.
- Kase, K.; Kondo, S.; Wakisaka, N.; Dochi, H.; Mizokami, H.; Kobayashi, E.; Kano, M.; Komori, T.; Hirai, N.; Ueno, T.; et al. Epstein-barr virus LMP1 induces soluble PD-L1 in nasopharyngeal carcinoma. Microorganisms 2021, 9, 603.
- Yang, J.; Hu, M.; Bai, X.; Ding, X.; Xie, L.; Ma, J.; Fan, B.; Yu, J. Plasma levels of soluble programmed death ligand 1 (sPD-L1) in WHO II/III nasopharyngeal carcinoma (NPC): A preliminary study. Medicine 2019, 98, e17231.
- Kushlinskii, N.E.; Gershtein, E.S.; Chang, V.L.; Korotkova, E.A.; Alferov, A.A.; Kontorshchikov, M.M.; Sokolov, N.Y.; Karamysheva, E.I.; Ognerubov, N.A.; Stilidi, I.S.; et al. Prognostic significance of soluble forms of immune checkpoint PD-1/PDL1 receptor and ligand in blood plasma of gastric cancer patients. Klin. Lab. Diagn. 2021, 66, 139–146.
- Ando, K.; Hamada, K.; Watanabe, M.; Ohkuma, R.; Shida, M.; Onoue, R.; Kubota, Y.; Matsui, H.; Ishiguro, T.; Hirasawa, Y.; et al. Plasma levels of soluble PD-L1 correlate with tumor regression in patients with lung and gastric cancer treated with immune checkpoint inhibitors. Anticancer Res. 2019, 39, 5195–5201.
- Shigemori, T.; Toiyama, Y.; Okugawa, Y.; Yamamoto, A.; Yin, C.; Narumi, A.; Ichikawa, T.; Ide, S.; Shimura, T.; Fujikawa, H.; et al. Soluble PD-L1 expression in circulation as a predictive marker for recurrence and prognosis in gastric cancer: Direct comparison of the clinical burden between tissue and serum PD-L1 expression. Ann. Surg. Oncol. 2019, 26, 876–883.
- Murakami, S.; Shibaki, R.; Matsumoto, Y.; Yoshida, T.; Goto, Y.; Kanda, S.; Horinouchi, H.; Fujiwara, Y.; Yamamoto, N.; Ohe, Y.; et al. Association between serum level soluble programmed cell death ligand 1 and prognosis in patients with non-small cell lung cancer treated with anti-PD-1 antibody. Thorac. Cancer 2020, 11, 3585–3595.
- Jia, Y.; Li, X.; Zhao, C.; Ren, S.; Su, C.; Gao, G.; Li, W.; Zhou, F.; Li, J.; Zhou, C. Soluble PD-L1 as a Predictor of the Response to EGFR-TKIs in non-small cell lung cancer patients with EGFR mutations. Front. Oncol. 2020, 10, 1455.
- Wu, W.; Xia, X.; Cheng, C.; Niu, L.; Wu, J.; Qian, Y. Serum soluble PD-L1, PD-L2, and B7-H5 as potential diagnostic biomarkers of human pancreatic cancer. Clin. Lab. 2021, 67, 6.
- Asanuma, K.; Nakamura, T.; Hayashi, A.; Okamoto, T.; Iino, T.; Asanuma, Y.; Hagi, T.; Kita, K.; Nakamura, K.; Sudo, A. Soluble programmed death-ligand 1 rather than PD-L1 on tumor cells effectively predicts metastasis and prognosis in soft tissue sarcomas. Sci. Rep. 2020, 10, 9077.
- Kushlinskii, N.E.; Alferov, A.A.; Boulytcheva, I.V.; Timofeev, Y.S.; Korotkova, E.A.; Khvan, O.T.; Kuzmin, Y.B.; Kuznetsov, I.N.; Bondarev, A.V.; Shchupak, M.Y.; et al. Comparative analysis of the levels of soluble forms of receptor and ligand of the immunity control point PD-1/PD-L1 in the blood serum of patients with typical bone osteosarcoma and chondrosarcoma. Klin. Lab. Diagn. 2020, 65, 669–675.
- Aghajani, M.J.; Roberts, T.L.; Yang, T.; McCafferty, C.E.; Caixeiro, N.J.; DeSouza, P.; Niles, N. Elevated levels of soluble PD-L1 are associated with reduced recurrence in papillary thyroid cancer. Endocr. Connect. 2019, 8, 1040–1051.
- Yazdanpanah, P.; Alavianmehr, A.; Ghaderi, A.; Monabati, A.; Montazer, M.; Tahmasbi, K.; Farjadian, S. PD-L1 expression in tumor lesions and soluble PD-L1 serum levels in patients with breast cancer: TNBC versus TPBC. Breast Dis. 2021, 40, 43–50.
- Jalali, S.; Price-Troska, T.; Paludo, J.; Villasboas, J.; Kim, H.J.; Yang, Z.Z.; Novak, A.J.; Ansell, S.M. Soluble PD-1 ligands regulate T-cell function in Waldenstrom macroglobulinemia. Blood Adv. 2018, 2, 1985–1997.
- Zhu, X.; Lang, J. Soluble PD-1 and PD-L1: Predictive and prognostic significance in cancer. Oncotarget 2017, 8, 97671–97682.
- Wei, W.; Xu, B.; Wang, Y.; Wu, C.; Jiang, J.; Wu, C. Prognostic significance of circulating soluble programmed death ligand-1 in patients with solid tumors: A meta-analysis. Medicine 2018, 97, e9617.
- Ding, X.C.; Wang, L.L.; Zhu, Y.F.; Li, Y.D.; Nie, S.L.; Yang, J.; Liang, H.; Weichselbaum, R.R.; Yu, J.M.; Hu, M. The change of soluble programmed cell death-ligand 1 in glioma patients receiving radiotherapy and its impact on clinical outcomes. Front. Immunol. 2020, 11, 580335.
- Atanackovic, D.; Luetkens, T. Biomarkers for checkpoint inhibition in hematologic malignancies. Semin. Cancer Biol. 2018, 52, 198–206.
- Mortensen, J.B.; Monrad, I.; Enemark, M.B.; Ludvigsen, M.; Kamper, P.; Bjerre, M.; d’Amore, F. Soluble programmed cell death protein 1 (sPD-1) and the soluble programmed cell death ligands 1 and 2 (sPD-L1 and sPD-L2) in lymphoid malignancies. Eur. J. Haematol. 2021.
- Mitsuhashi, A.; Okuma, Y. Perspective on immune oncology with liquid biopsy, peripheral blood mononuclear cells, and microbiome with non-invasive biomarkers in cancer patients. Clin. Transl. Oncol. 2018, 20, 966–974.
- Kambayashi, Y.; Fujimura, T.; Hidaka, T.; Aiba, S. Biomarkers for predicting efficacies of anti-PD1 antibodies. Front. Med. 2019, 6, 174.
- Costantini, A.; Takam Kamga, P.; Dumenil, C.; Chinet, T.; Emile, J.F.; Giroux Leprieur, E. Plasma biomarkers and immune checkpoint inhibitors in non-small cell lung cancer: New tools for better patient selection? Cancers 2019, 11, 1269.
- Carretero-González, A.; Lora, D.; Martín Sobrino, I.; Sáez Sanz, I.; Bourlon, M.T.; Anido Herranz, U.; Martínez Chanzá, N.; Castellano, D.; de Velasco, G. The Value of PD-L1 expression as predictive biomarker in metastatic renal cell carcinoma patients: A meta-analysis of randomized clinical trials. Cancers 2020, 12, 1945.
- Mildner, F.; Sopper, S.; Amann, A.; Pircher, A.; Pall, G.; Köck, S.; Naismith, E.; Wolf, D.; Gamerith, G. Systematic review: Soluble immunological biomarkers in advanced non-small-cell lung cancer (NSCLC). Crit. Rev. Oncol. Hematol. 2020, 153, 102948.
- Duchemann, B.; Remon, J.; Naigeon, M.; Mezquita, L.; Ferrara, R.; Cassard, L.; Jouniaux, J.M.; Boselli, L.; Grivel, J.; Auclin, E.; et al. Integrating circulating biomarkers in the immune checkpoint inhibitor treatment in lung cancer. Cancers 2020, 12, 3625.
- Cunha Pereira, T.; Rodrigues-Santos, P.; Almeida, J.S.; Rêgo Salgueiro, F.; Monteiro, A.R.; Macedo, F.; Soares, R.F.; Domingues, I.; Jacinto, P.; Sousa, G. Immunotherapy and predictive immunologic profile: The tip of the iceberg. Med. Oncol. 2021, 38, 51.
- Sui, X.; Jiang, L.; Teng, H.; Mi, L.; Li, B.; Shi, A.; Yu, R.; Li, D.; Dong, X.; Yang, D.; et al. Prediction of clinical outcome in locally advanced non-small cell lung cancer patients treated with chemoradiotherapy by plasma markers. Front. Oncol. 2021, 10, 625911.
- Castello, A.; Rossi, S.; Toschi, L.; Mansi, L.; Lopci, E. Soluble PD-L1 in NSCLC patients treated with checkpoint inhibitors and its correlation with metabolic parameters. Cancers 2020, 12, 1373.
- Mazzaschi, G.; Minari, R.; Zecca, A.; Cavazzoni, A.; Ferri, V.; Mori, C.; Squadrilli, A.; Bordi, P.; Buti, S.; Bersanelli, M.; et al. Soluble PD-L1 and Circulating CD8+PD-1+ and NK cells enclose a prognostic and predictive immune effector score in immunotherapy treated NSCLC patients. Lung Cancer 2020, 148, 1–11.
- Qu, Y.; Wang, H.; Liu, H.; Sun, X.; Li, J.; Yu, H. Molecular mechanism of expression changes of immunological indexes of PD-1/sPD-L1 after radiotherapy in nonsmall cell lung cancer. Biomed. Res. Int. 2021, 2021, 8811751.
- He, J.; Pan, Y.; Guo, Y.; Li, B.; Tang, Y. Study on the expression levels and clinical significance of PD-1 and PD-L1 in plasma of NSCLC Patients. J. Immunother. 2020, 43, 156–164.
- Takeuchi, M.; Doi, T.; Obayashi, K.; Hirai, A.; Yoneda, K.; Tanaka, F.; Iwai, Y. Soluble PD-L1 with PD-1-binding capacity exists in the plasma of patients with non-small cell lung cancer. Immunol. Lett. 2018, 196, 155–160.
- Qu, S.; Jiao, Z.; Lu, G.; Yao, B.; Wang, T.; Rong, W.; Xu, J.; Fan, T.; Sun, X.; Yang, R.; et al. PD-L1 lncRNA splice isoform promotes lung adenocarcinoma progression via enhancing c-Myc activity. Genome Biol. 2021, 22, 104.
- Liang, Z.; Tian, Y.; Cai, W.; Weng, Z.; Li, Y.; Zhang, H.; Bao, Y.; Li, Y. High-affinity human PD-L1 variants attenuate the suppression of T cell activation. Oncotarget 2017, 8, 88360–88375.
- Han, B.; Dong, L.; Zhou, J.; Yang, Y.; Guo, J.; Xuan, Q.; Gao, K.; Xu, Z.; Lei, W.; Wang, J.; et al. The clinical implication of soluble PD-L1 (sPD-L1) in patients with breast cancer and its biological function in regulating the function of T lymphocyte. Cancer Immunol. Immunother. 2021.
- Shi, B.; Du, X.; Wang, Q.; Chen, Y.; Zhang, X. Increased PD-1 on CD4(+)CD28(-) T cell and soluble PD-1 ligand-1 in patients with T2DM: Association with atherosclerotic macrovascular diseases. Metabolism 2013, 62, 778–785.
- Frigola, X.; Inman, B.A.; Lohse, C.M.; Krco, C.J.; Cheville, J.C.; Thompson, R.H.; Leibovich, B.; Blute, M.L.; Dong, H.; Kwon, E.D.; et al. Identification of a soluble form of B7-H1 that retains immunosuppressive activity and is associated with aggressive renal cell carcinoma. Clin. Cancer Res. 2011, 17, 1915–1923.
- Tannig, P.; Peter, A.S.; Lapuente, D.; Klessing, S.; Damm, D.; Tenbusch, M.; Überla, K.; Temchura, V. Modulation of vaccine-induced HIV-1-specific immune responses by co-electroporation of PD-L1 encoding DNA. Vaccines 2020, 8, 27.
- Zhang, A.; Sun, Y.; Wang, S.; Du, J.; Gao, X.; Yuan, Y.; Zhao, L.; Yang, Y.; Xu, L.; Lei, Y.; et al. Secretion of human soluble programmed cell death protein 1 by chimeric antigen receptor-modified T cells enhances anti-tumor efficacy. Cytotherapy 2020, 22, 734–743.
- Orme, J.J.; Jazieh, K.A.; Xie, T.; Harrington, S.; Liu, X.; Ball, M.; Madden, B.; Charlesworth, M.C.; Azam, T.U.; Lucien, F.; et al. ADAM10 and ADAM17 cleave PD-L1 to mediate PD-(L)1 inhibitor resistance. Oncoimmunology 2020, 9, 1744980.
- Gong, B.; Kiyotani, K.; Sakata, S.; Nagano, S.; Kumehara, S.; Baba, S.; Besse, B.; Yanagitani, N.; Friboulet, L.; Nishio, M.; et al. Secreted PD-L1 variants mediate resistance to PD-L1 blockade therapy in non-small cell lung cancer. J. Exp. Med. 2019, 216, 982–1000.
- Ng, K.W.; Attig, J.; Young, G.R.; Ottina, E.; Papamichos, S.I.; Kotsianidis, I.; Kassiotis, G. Soluble PD-L1 generated by endogenous retroelement exaptation is a receptor antagonist. Elife 2019, 8, e50256.
- Mahoney, K.M.; Shukla, S.A.; Patsoukis, N.; Chaudhri, A.; Browne, E.P.; Arazi, A.; Eisenhaure, T.M.; Pendergraft, W.F., 3rd; Hua, P.; Pham, H.C.; et al. A secreted PD-L1 splice variant that covalently dimerizes and mediates immunosuppression. Cancer Immunol. Immunother. 2019, 68, 421–432.
- Hassounah, N.B.; Malladi, V.S.; Huang, Y.; Freeman, S.S.; Beauchamp, E.M.; Koyama, S.; Souders, N.; Martin, S.; Dranoff, G.; Wong, K.K.; et al. Identification and characterization of an alternative cancer-derived PD-L1 splice variant. Cancer Immunol. Immunother. 2019, 68, 407–420.




