The molecular basis of antimicrobial resistance in
Aeromonas spp
., has been widely studied, but their importance in the hospital area as a cause of outbreaks is not fully established. Likewise, reports on resistance are varied regarding the origin of isolation and the type of antimicrobials tested in vitro
[7]. The
Aeromonas resistance profile has not changed significantly; until now, the mechanism of action of inducible chromosomal β-lactamases and carbapenemase expression has been suggested as being the main resistance mechanism for
Aeromonas species. Three classes of β-lactamases are recognized in
Aeromonas; one of class C cephalosporinase, one of class D penicillinase, and one of class B metallo-β-lactamase (MBL)
[9][10].
Colistin Resistance in Aeromonas
Since the report in 2016, where it was shown that colistin resistance can be encoded by the
mcr genes detected within a plasmid, it was determined that these genes are not only in bacterial genophores but can also be present in mobile genetic elements as plasmids
[2]. From this report, attention was paid to the search for and detection of these genes in different bacterial genera, mainly those of medical importance, but also isolated from other sources, such as the environment or animals.In
K. pneumoniae,
P. aeruginosa, or others, such as
Aeromonas genus,
mcr gene variants have been detected, and the reports are increasing
[13][14].
Colistin resistance in
Aeromonas has been reported in several regions of the world, mainly in Europe and Asia. Resistance to this antibiotic has been reported in Latin America in other genera, but not in
Aeromonas. The detection of colistin resistance is more common in the clinical area; however, colistin-resistant strains of
Aeromonas have been isolated from other origins that have been detected, from which investigations and reports have emerged in the world. The extensive use of antibiotics in aquaculture and in human treatment has led to an increase in the resistance of this genus to antimicrobial drugs
[11][12].
The species
A. dhakensis, A. hydrophila, A. caviae, and
A. veronii are considered the main causes of human infections that can cause infection in wounds, diarrheal syndromes, and other clinical presentations
[9]. Commonly, the isolates do not present resistance to antimicrobials; however, MDR isolates have still appeared, and in recent years the report of
Aeromonas isolates from clinical samples and from various sources with resistance to colistin has increased
[15]. This resistance has been investigated in
Aeromonas spp., isolates, by means of disk diffusion test and by minimum inhibitory concentration (MIC), showing the MIC method to be more effective. Induction of colistin resistance in the strains showed an 85% increase after overnight incubation in a tube with
Müller–
Hinton broth and a 50 µL colistin disk. This result allowed the establishment of a phenotypic marker in the
Aeromonas isolates
[16].
In an
A. veronii isolate from chicken meat, two adjacent genes with colistin resistance markers, called
mcr-3.3 and
mcr-3-like, were detected in the genophore. The result had 95.2 and 84.19% identity in the nucleotide sequence, when compared to the
mcr-3 gene of an
E. coli of porcine origin
[17].
The evidence of the
mcr genes in
Aeromonas was demonstrated by a group of scientists who analyzed a total of 6497 strains that were collected in 13 provinces of China between 2016 and 2017. In these samples, the presence of the
mcr-3 genes was detected by PCR. The
mcr-3 gene was detected in 49 strains only, of which eight strains corresponded to the genus
Aeromonas, two
A. hydrophila strains, one with
mcr-3.8 variant, and one with
mcr-3.9 variant, one
A. caviae with
mcr-3.1 variant, and one
A. media with
mcr-3.6 variant. Of the four remaining strains, one each were of
A. veronii, A. media, and
A. caviae, and one was
Aeromonas spp., with
mcr-3 without variant. All the strains were grouped into a subclade, after the phylogenetic analysis of the sequences of the
mcr-3 genes detected in the strains
[18].
Another group of researchers found four
Aeromonas isolates with the presence of the
mcr-3 gene through PCR, while the
mcr-1 or
mcr-2 genes were not detected. Each of the four isolates with
mcr-3 genes presented a different variant each; these presented identities in the amino acid chain were of 95 to 98% compared to the original protein MCR-3. These variants of the protein were designated as MCR-3.6 obtained from the
A. allosaccharophila strain isolated from
Leuciscus idus, MCR-3.7 for the protein detected in the
A. media strain isolated from
Meleagris gallopavo, MCR-3.8 for that detected in the
A. jandaei strain isolated from a
Cyprinus carpio carp, and MCR-3.9 for the protein of the
A. hydrophila strain of
Cyprinus carpio. The isolate with the
mcr-3.9 gene also contained an additional
mcr-3.8 gene in the MIC test, with colistin showing an MIC ≥ 128 mg/L higher compared to the other isolates
[19].
The reports include a new variant of the
mcr-3 gene in
A. caviae, also detected in
Proteus mirabilis and
E. coli that were isolated from a domestic duck. These strains were obtained from sewage samples from free-range ducks, which were raised near a river in the suburban area of Qingdao, Shangdon Province, in China. The presence of the
mcr-3 gene was demonstrated in 1 of 15 samples processed in this study. The result was obtained by detection of the
mcr gene directly in the sample; the positive sample was seeded in a CHROMagar plate from Biomerieux
®, France, to which 2 mg/L of colistin was added. Based on the above, three positive strains were detected for
mcr-3 gene.
A. caviae 17AC,
P. mirabilis 17PM, and
E. coli 17EC strains were identified by MALDI-TOF (Matrix-Assisted Laser Desorption/Ionization-Time of Flight) technology, and by
16S rRNA gene sequencing
[19]. In another study, the prevalence was determined, complemented by a genetic analysis of the
mcr-3 gene in
Aeromonas species. These isolates were obtained from human rectal exudates, meat for human consumption, and environmental water samples
[20].
The variant
mcr-5 gene of colistin resistance was detected in an
A. hydrophila strain isolated from a fecal sample from a backyard pig. In this case, the
mcr-5 gene was detected in a plasmid with 7915 base pairs (bp) named pI064-2. Additionally, they analyzed the possibility of transforming the
A. hydrophila strains susceptible to colistin into a resistant strain
[21].
Various mechanisms of resistance to colistin, in addition to the mechanism mediated by
mcr genes, have been described in some bacteria, including
P. aeruginosa, A. baumannii, members of the
Enterobacteriaceae family, such as
E. coli, Salmonella spp
., and
K. pneumoniae they have an acquired resistance against colistin. However, the possibility of the appearance of strains resistant to this antibiotic should be monitored, due to the presence of mutations, new mechanisms, or adaptations
[22].
The protein generated by the
mcr genes confers resistance to polymyxins; this protein, called MCR, from the inner membrane, adds a molecule of phosphoethanolamine (PEA) to lipid A of lipopolysaccharide (Kdo2-PEA-Lipid A), synthesized by the binding of uridine diphosphate (UDP) and N-acetyl glucosamine (GlcNac) mediated by Lpx proteins (C, D, H, B, K, L, M). The product binds to 2-keto deoxioctanate acid (Kdo2), which is transported by the MsbA protein to the inner membrane, where the PEA molecule is added. The Lpt protein complex (ABCFD/DE) transports the modified LPS that generates resistance to colistin, since the negative charge with which it interacts was modified (Figure 1)
[23][24][25].
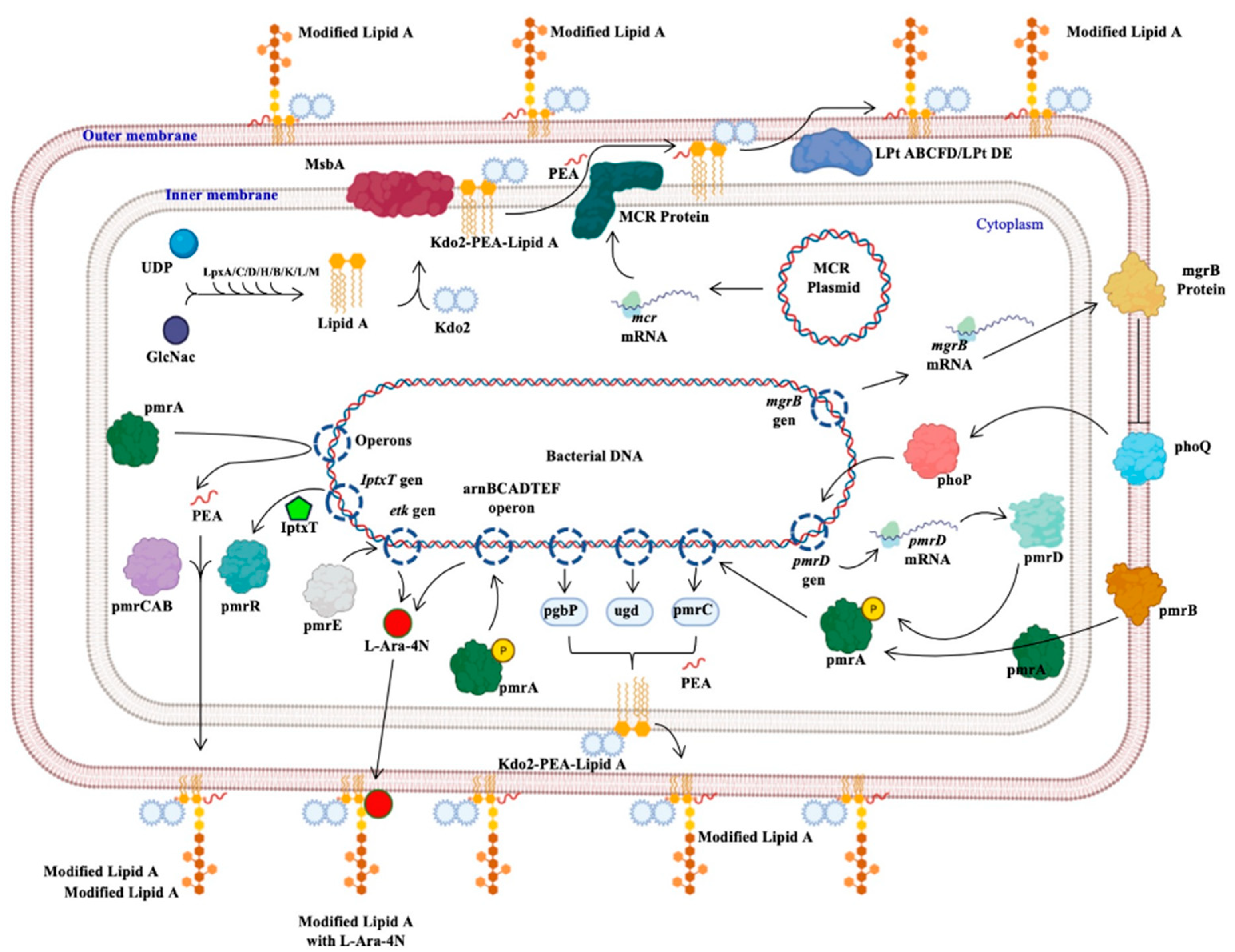 Figure 1.
Figure 1. Bacterial mechanisms of resistance to colistin. The different mechanisms of resistance to colistin described in Gram-negative bacilli and other bacteria are presented: the mechanism mediated by the
mcr genes, those regulated by the phoP and phoQ proteins, others regulated by the pmr proteins or by the
iptxT and
etk genes, and operons that encode activation proteins of pmr proteins
[22][23][24][25]. Generated from this work.

 Figure 1. Bacterial mechanisms of resistance to colistin. The different mechanisms of resistance to colistin described in Gram-negative bacilli and other bacteria are presented: the mechanism mediated by the mcr genes, those regulated by the phoP and phoQ proteins, others regulated by the pmr proteins or by the iptxT and etk genes, and operons that encode activation proteins of pmr proteins [22][23][24][25]. Generated from this work.
Figure 1. Bacterial mechanisms of resistance to colistin. The different mechanisms of resistance to colistin described in Gram-negative bacilli and other bacteria are presented: the mechanism mediated by the mcr genes, those regulated by the phoP and phoQ proteins, others regulated by the pmr proteins or by the iptxT and etk genes, and operons that encode activation proteins of pmr proteins [22][23][24][25]. Generated from this work.