
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Carolina Carlisle | + 1806 word(s) | 1806 | 2021-05-27 11:14:15 | | | |
| 2 | Lindsay Dong | Meta information modification | 1806 | 2021-06-25 03:19:59 | | |
Video Upload Options
The endocannabinoid system (ECS) consists of endogenous cannabinoids, their receptors, and metabolic enzymes that play a critical homeostatic role in modulating polyunsaturated omega fatty acid (PUFA) signaling to maintain a balanced inflammatory and redox state.
1. Introduction
The endocannabinoid system (ECS) is a major signaling network comprised of endogenous, lipid-based, physiological ligands (endocannabinoids) that play a pro-homeostatic role in central and peripheral organs of the human body. It is believed to constitute a feedback loop for nutrient-energy metabolism, and regulates various cellular functions, immune responses, and stress adaptation pathways to support physiological homeostasis as human diets change due to evolutionary and environmental factors [1]. The physiological outcomes may range from simple functions like eating, sleeping, and relaxing, to the more complex, including neuroplasticity, metabolism, and inflammation [2]. The functional integration of the ECS system in the control of inflammation, energy metabolism, and emotional homeostasis has been directly connected to its ability to adequately perceive and respond to the paucity of these signals [3]. Chronic, excessive, or unresolved overstimulation of the ECS often results in systemic imbalance. This can trigger oxidative stress by increasing the levels of reactive oxygen species (ROS) that adversely affect protein oxidation [4], mitochondrial bioenergetics [5], cellular functions, and redox balance maintained in part by the nuclear factor erythroid 2-related factor 2 (Nrf2) [6][7].
Endocannabinoids are metabolite products of dietary essential polyunsaturated fatty acids (PUFAs) (Figure 1). PUFAs are essential in terms of their critical role in maintaining the efficient structure and integral function of the cell membranes, and they must be obtained from the dietary sources [8].
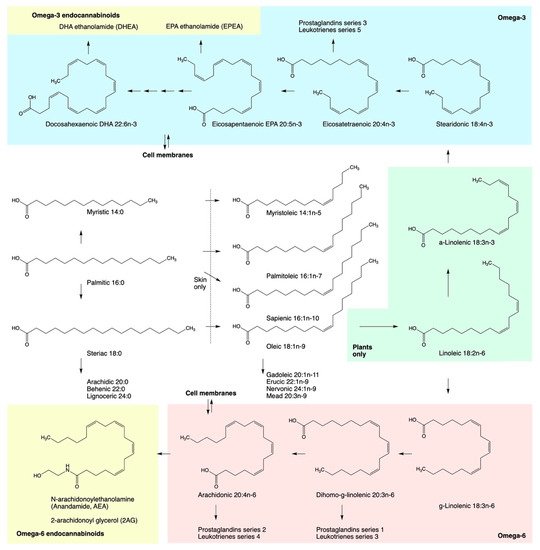
Figure 1. Pathways in the biosynthesis of polyunsaturated fatty acids including n-6 and n-3 essential fatty acids precursors, eicosanoid family metabolites, and endocannabinoids.
2. ECS Tone and Its Health Implications
Endocannabinoids activate presynaptic and astrocytic CB1 receptors distributed primarily in the central nervous system and CB2 receptors found in highest densities on the peripheral immune and hematopoietic cells [9]. The wide and heterogeneous distribution of CB1 receptors in the brain [10], and recent findings of CB2 receptors not only in microglia but also in neurons [11], at the same time suggests that strengthening of the ECS tone by mild activation of central CB1 and CB2 is closely related with an emotional (anxiety, depression, PTSD) wellbeing, while overstimulation of CB1 activity induces psychotic symptoms [12]. An alternative strategy to improve ECS tone is increasing tissue levels of endocannabinoids by inhibiting their cellular uptake and metabolism [13]. This approach was proven successful with a series of pharmacological inhibitors targeting two key enzymes of ECS metabolism-fatty acid amide hydrolase (FAAH) that degrades AEA [14], and monoacylglycerol lipase (MAGL) that degrades 2-AG [15].
3. Crosstalk between Inflammatory and ECS Signaling Mediators
Biological effects of arachidonic acid that serves as a precursor for endocannabinoids, are largely attributed to its enzymatic conversion to prostaglandins and thromboxanes by membrane-bound cyclooxygenases (COX), leukotrienes by cytoplasmic lipoxygenases (LOX), and lipoxides by cytochrome P450 epoxygenases [16]. Prostaglandins derived from arachidonic acid serve as secondary messengers of hydrophilic bioactive molecules such as glucocorticoids, non-steroidal anti-inflammatory drugs (NSAIDs), and statins to regulate blood cell aggregation, dilation of blood vessels, and vascular permeability, resulting in increased tissue edema, hyperemia, and fever to maintain the propagation of an inflammatory process. Both COX and LOX enzymes metabolize eicosanoids and endocannabinoid substrates with a similar efficacy [17]. While the oxidized endocannabinoids do not activate classical CB1/2 receptors, their cellular targets and respective biological mechanisms of action remain to be elucidated.
4. Direct Modulation of ECS by Fatty Acids
Dietary fatty acids play a direct role in generation of lipid mediators that modulate inflammation and oxidative stress. While humans are very effective at desaturating two major circulating fatty acids, palmitic acid 16:0 and stearic acid 18:0 to their palmitoleic acid 16:1(n-7) and oleic acid 18:1(n-9) monounsaturated counterparts, they lack the enzymes to introduce the second unsaturated bond to form polyunsaturated linoleic acid 18:2(n-6) (a dietary precursor of omega-6 fatty acids) and α-linolenic acid 18:3(n-3) (a dietary precursor of omega-3 fatty acids) in their bodies. Dietary α-linolenic acid can be slowly metabolized into the long-chain polyunsaturated omega-3 fatty acids EPA and DHA when ingested directly, albeit with a lower efficiency [18]. Some marine organisms, especially fatty fish and shellfish accumulate large quantities of algae-derived EPA and DHA and serve as an excellent dietary source of these fatty acids [19].
5. Hemp Oils as ECS Metabolic Modulators
Hemp oils derived from the cannabis plant (Cannabis sativa L.) are a rich source of lipid bioactive compounds, including cannabinoids, β-caryophyllene, and polyunsaturated fatty acids [20] that potentially interact with the ECS. Following the first elucidation of structure and chemical synthesis of cannabinol (CBN), THC, and CBD in 1940s-60s [21] (Figure 2a), the first THC-binding site was described as CB1 receptor twenty years later [22], while specific molecular targets of CBD remained unknown until today. The therapeutic potential of cannabinoids, both as single bioactive agents and in combination with other bioactive plant compounds, to manage and alleviate multiple human health outcomes has received an excellent review [23].
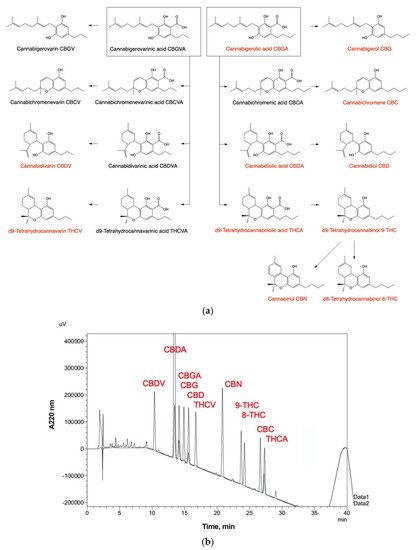
Figure 2. Major biosynthetic routes and cannabinoid metabolites found in Cannabis sativa tissues and botanical extracts. (a) The corresponding decarboxylation products are formed during collection, storage, and thermal processing of plant tissues. (b) HPLC-UV detection and full baseline resolution of 11 major cannabinoids using Shimadzu Prominence LC-2030C workflow as outlined in Table 1.
| Step | Detailed Description | Comments |
|---|---|---|
| 1a | Hemp tissues and powdered samples: Weigh 1 g of air-dried samples from sealed bags in triplicate Add 20 mL of methanol/chloroform (9:1, v:v) Agitate in an orbital shaker for 30 min at 200 rpm and 37 °C Sonicate in an ultrasonic bath for 30 min at 37 °C Centrifuge for 5 min at 3000 rpm and RT, and collect supernatant Repeat twice and combine supernatants into a single sample Evaporate to dryness and dissolve in 1 mL methanol Collect supernatant and filter through a 0.45 µm PTFE syringe filter |
Whole plant complexity, minor cannabinoids of importance, and a variable terpenoid profile may all contribute to beneficial entourage effect of hemp |
| 1b | Hemp oils and liquid formulations: Add 400 µL of isopropanol to a 2 mL Eppendorf tube Add 10 µL of liquid sample and completely dissolve Vortex the sample for 30 sec at RT Add 400 µL of methanol to the mixture Vortex the sample for 30 sec at RT Filter through a 0.45 µm PTFE syringe filter |
Hemp oil density is 0.92 (used as a conversion factor to calculate volume to weight ratio) |
| 1c | Biological fluids (urine or plasma): Add 1 mL urine (3 mL plasma) and 20 µL of DDT to a 20 mL glass vial Add 3 mL of 100 mM sodium acetate buffer (pH 4.8) and mix briefly Add 375 μL of β-glucuronidase in acetate buffer Vortex briefly and incubate at 37 °C for 16 h Add 15 mL of ice cold 1% formic acid in acetonitrile Vortex briefly and sonicate in an ultrasonic bath for 3 min at RT Centrifuge for 5 min at 10,000 rpm and RT, and collect supernatant Add 15 mL methanol to the pellet and vortex briefly Repeat once and combine supernatants into a single sample Evaporate to dryness and dissolve in 200 µL methanol |
4,4- Dichlorodiphenyltrichloro ethane (DDT, 50 µg/mL) is used as an internal analytical standard; β-glucuronidase (Abalone) |
| 2 | Standard curves over a linear dynamic range of 0.5–100 μg/mL (ppm) | Shimadzu #220-91239-21 |
| 3 | Instrument: Shimadzu Prominence LC-2030C UV Column: Restek Ultra C18 (250 mm × 4.6 mm, 5 μm dp) Guard column: Restek Ultra C18 Guard (10 mm × 2.1 mm, 5 μm dp) Mobile-phase A: 0.1% formic acid in water Mobile-phase B: 100% acetonitrile Flow rate: 1 mL/min; Column temperature: 30 °C Injection volume: 20 μL; Detection: 220 nm |
Cannabinoid totals are calculated as the sum of the neutral form and the acid form multiplied by the conversion factors (0.877 for THCA and CBDA; 0.878 for CBGA) |
It is generally accepted that Δ9-THC found in Cannabis is a partial agonist of both CB1 and CB2 receptors similar to the endocannabinoids, with a slightly higher affinity for CB1 in the Ki range 4–40 nM [24], and exerts both psychotropic as well pain- and emesis-controlling effects in the CNS via CB1 and 5HT3 receptors activation [25].
6. Phytochemical Complexity of Hemp Oils
Overall, the hemp plant produces in excess of 140 cannabinoids that determine its potency and over 100 terpenes that define its distinctive flavor and aroma [26]. A typical high-resolution high-performance liquid chromatography (HPLC) analytical method with UV detection is capable of a full baseline resolution of 11 major cannabinoids including their native (acid) and neutral (decarboxylated) forms (Figure 2b).
7. ECS and the Oxidative Stress: Role of Dietary Antioxidants
The ECS tone is also critically associated with other systemic function of the body including inflammation and oxidative stress. Cellular defense mechanisms against oxidative, inflammatory, and toxic biochemical and environmental challenges constitute an important preservation mechanism to maintain optimal cellular structure and metabolism. The ability of the body to detoxify free radicals from an imbalanced physiological manifestation of ROS and reactive nitrogen species (RNS) may lead to a state of oxidative stress. The cellular redox homeostasis is challenged due to the formation of ROS and the prolonged oxidative stress is the underlying cause for unresolved inflammation leading to the development of several chronic metabolic conditions [27]. The uncontrolled ROS production and activity of the native antioxidant enzymes superoxide dismutase, glutathione peroxidase, nitric oxide synthase and catalase leads to increased peroxidation of polyunsaturated fatty acids, DNA oxidation, and cell death [28], therefore, the balanced redox system is critical in preserving the healthy metabolic conditions. Phytocannabinoids, including CBD, decreased the neuronal damage associate with β-amyloid accumulation and NADPH oxidase activity in SH-SY5Y neuroblastoma and BV-2 microglia cells, thus ameliorating oxidative stress [29].
Additional dietary Nrf2 activators such as glucosinolates found in cruciferous vegetables may potentiate induction of genes that regulate defenses against oxidative stress, inflammation, and DNA-damaging electrophiles when consumed as a part of the healthy diet or dietary intervention strategy. Studies have shown that many isothiocyanates, particularly sulforaphane, increased expression of antioxidant enzymes via the activation of Nrf-2 dependent pathway [30]. The biological actions of glucosinolate-derived isothiocyanates (ITCs) with active myrosinase component enhanced the bioavailability of ITCs thereby enhancing Nfr2 activation and promoting cellular antioxidant capacity [31].
8. Conclusions
Dietary PUFAs are major sources for the biosynthesis of endocannabinoids, both by contribution of omega-6 fatty acids to the arachidonic biosynthesis pathway and modulation of the ECS tone by dietary omega-3 EPA and DHA esterified to phospholipids. Understanding this intricate balance could hold potential to reduce over activation of central and circulating endocannabinoids observed in subjects with metabolic disorders [32], as well as modifying the onset and progression of many chronic metabolic and immune disorders associated with elevated inflammation and oxidative stress. The role of multiple cannabinoids in the integrated and often opposing control of these outcomes may be considered direct evidence in favor of developing novel strategies that deliver appropriate quantities and ratios of different metabolites found in full-spectrum hemp oils and provide a more discriminating means of eliciting a balanced and personalized response. While no single bioactive principle may balance these complex health outcomes, novel intervention strategies and selected nutrient supplementation regimes that specifically target the ECS, and metabolic health outcomes could support key mediators and inflammatory resolution pathways critical in maintaining a well-balanced systemic function of the body.
References
- Gertsch, J. Cannabimimetic Phytochemicals in the Diet - an Evolutionary Link to Food Selection and Metabolic Stress Adaptation? Br. J. Pharmacol. 2017, 174, 1464–1483.
- Di Marzo, V. “Endocannabinoids” and Other Fatty Acid Derivatives with Cannabimimetic Properties: Biochemistry and Possible Physiopathological Relevance. Biochim. Biophys. Acta 1998, 1392, 153–175.
- Freitas, H.R.; Isaac, A.R.; Malcher-Lopes, R.; Diaz, B.L.; Trevenzoli, I.H.; De Melo Reis, R.A. Polyunsaturated Fatty Acids and Endocannabinoids in Health and Disease. Nutr. Neurosci. 2018, 21, 695–714.
- Chen, C.-A.; Wang, T.-Y.; Varadharaj, S.; Reyes, L.A.; Hemann, C.; Talukder, M.A.H.; Chen, Y.-R.; Druhan, L.J.; Zweier, J.L. S-Glutathionylation Uncouples ENOS and Regulates Its Cellular and Vascular Function. Nature 2010, 468, 1115–1118.
- Han, Z.; Varadharaj, S.; Giedt, R.J.; Zweier, J.L.; Szeto, H.H.; Alevriadou, B.R. Mitochondria-Derived Reactive Oxygen Species Mediate Heme Oxygenase-1 Expression in Sheared Endothelial Cells. J. Pharmacol. Exp. Ther. 2009, 329, 94–101.
- Rajasekaran, N.S.; Varadharaj, S.; Khanderao, G.D.; Davidson, C.J.; Kannan, S.; Firpo, M.A.; Zweier, J.L.; Benjamin, I.J. Sustained Activation of Nuclear Erythroid 2-Related Factor 2/Antioxidant Response Element Signaling Promotes Reductive Stress in the Human Mutant Protein Aggregation Cardiomyopathy in Mice. Antioxid. Redox Signal. 2011, 14, 957–971.
- Sies, H.; Berndt, C.; Jones, D.P. Oxidative Stress. Annu. Rev. Biochem. 2017, 86, 715–748.
- Ander, B.P.; Dupasquier, C.M.; Prociuk, M.A.; Pierce, G.N. Polyunsaturated Fatty Acids and Their Effects on Cardiovascular Disease. Exp. Clin. Cardiol. 2003, 8, 164–172.
- Pertwee, R.G.; Ross, R.A. Cannabinoid Receptors and Their Ligands. Prostaglandins Leukot. Essent. Fatty Acids 2002, 66, 101–121.
- Busquets-Garcia, A.; Bains, J.; Marsicano, G. CB1 Receptor Signaling in the Brain: Extracting Specificity from Ubiquity. Neuropsychopharmacology 2018, 43, 4–20.
- Liu, Q.-R.; Canseco-Alba, A.; Zhang, H.-Y.; Tagliaferro, P.; Chung, M.; Dennis, E.; Sanabria, B.; Schanz, N.; Escosteguy-Neto, J.C.; Ishiguro, H.; et al. Cannabinoid Type 2 Receptors in Dopamine Neurons Inhibits Psychomotor Behaviors, Alters Anxiety, Depression and Alcohol Preference. Sci. Rep. 2017, 7, 17410.
- Navarrete, F.; García-Gutiérrez, M.S.; Jurado-Barba, R.; Rubio, G.; Gasparyan, A.; Austrich-Olivares, A.; Manzanares, J. Endocannabinoid System Components as Potential Biomarkers in Psychiatry. Front. Psychiatry 2020, 11.
- Roques, B.P.; Fournié-Zaluski, M.-C.; Wurm, M. Inhibiting the Breakdown of Endogenous Opioids and Cannabinoids to Alleviate Pain. Nat. Rev. Drug Discov. 2012, 11, 292–310.
- Godlewski, G.; Alapafuja, S.O.; Bátkai, S.; Nikas, S.P.; Cinar, R.; Offertáler, L.; Osei-Hyiaman, D.; Liu, J.; Mukhopadhyay, B.; Harvey-White, J.; et al. Inhibitor of Fatty Acid Amide Hydrolase Normalizes Cardiovascular Function in Hypertension without Adverse Metabolic Effects. Chem. Biol. 2010, 17, 1256–1266.
- Cao, Z.; Mulvihill, M.M.; Mukhopadhyay, P.; Xu, H.; Erdélyi, K.; Hao, E.; Holovac, E.; Haskó, G.; Cravatt, B.F.; Nomura, D.K.; et al. Monoacylglycerol Lipase Controls Endocannabinoid and Eicosanoid Signaling and Hepatic Injury in Mice. Gastroenterology 2013, 144, 808–817.e15.
- Rouzer, C.A.; Marnett, L.J. Endocannabinoid Oxygenation by Cyclooxygenases, Lipoxygenases, and Cytochromes P450: Cross-Talk between the Eicosanoid and Endocannabinoid Signaling Pathways. Chem. Rev. 2011, 111, 5899–5921.
- Forsell, P.K.A.; Brunnström, A.; Johannesson, M.; Claesson, H.-E. Metabolism of Anandamide into Eoxamides by 15-Lipoxygenase-1 and Glutathione Transferases. Lipids 2012, 47, 781–791.
- Burdge, G.C.; Calder, P.C. Conversion of Alpha-Linolenic Acid to Longer-Chain Polyunsaturated Fatty Acids in Human Adults. Reprod. Nutr. Dev. 2005, 45, 581–597.
- Calder, P.C. Marine Omega-3 Fatty Acids and Inflammatory Processes: Effects, Mechanisms and Clinical Relevance. Biochim. Biophys. Acta 2015, 1851, 469–484.
- Badal, S.; Smith, K.N.; Rajnarayanan, R. Analysis of Natural Product Regulation of Cannabinoid Receptors in the Treatment of Human Disease. Pharmacol. Ther. 2017, 180, 24–48.
- Howlett, A.C.; Breivogel, C.S.; Childers, S.R.; Deadwyler, S.A.; Hampson, R.E.; Porrino, L.J. Cannabinoid Physiology and Pharmacology: 30 Years of Progress. Neuropharmacology 2004, 47 (Suppl. 1), 345–358.
- Matsuda, L.A.; Lolait, S.J.; Brownstein, M.J.; Young, A.C.; Bonner, T.I. Structure of a Cannabinoid Receptor and Functional Expression of the Cloned CDNA. Nature 1990, 346, 561–564.
- Russo, E.B. Taming THC: Potential Cannabis Synergy and Phytocannabinoid-Terpenoid Entourage Effects. Br. J. Pharmacol. 2011, 163, 1344–1364.
- Compton, D.R.; Rice, K.C.; De Costa, B.R.; Razdan, R.K.; Melvin, L.S.; Johnson, M.R.; Martin, B.R. Cannabinoid Structure-Activity Relationships: Correlation of Receptor Binding and in Vivo Activities. J. Pharmacol. Exp. Ther. 1993, 265, 218–226.
- Tramèr, M.R.; Carroll, D.; Campbell, F.A.; Reynolds, D.J.; Moore, R.A.; McQuay, H.J. Cannabinoids for Control of Chemotherapy Induced Nausea and Vomiting: Quantitative Systematic Review. BMJ 2001, 323, 16–21.
- Hanuš, L.O.; Meyer, S.M.; Muñoz, E.; Taglialatela-Scafati, O.; Appendino, G. Phytocannabinoids: A Unified Critical Inventory. Nat. Prod. Rep. 2016, 33, 1357–1392.
- Varadharaj, S.; Kelly, O.J.; Khayat, R.N.; Kumar, P.S.; Ahmed, N.; Zweier, J.L. Role of Dietary Antioxidants in the Preservation of Vascular Function and the Modulation of Health and Disease. Front. Cardiovasc. Med. 2017, 4, 64.
- Pisoschi, A.M.; Pop, A. The Role of Antioxidants in the Chemistry of Oxidative Stress: A Review. Eur. J. Med. Chem. 2015, 97, 55–74.
- Janefjord, E.; Mååg, J.L.V.; Harvey, B.S.; Smid, S.D. Cannabinoid Effects on β Amyloid Fibril and Aggregate Formation, Neuronal and Microglial-Activated Neurotoxicity in Vitro. Cell. Mol. Neurobiol. 2014, 34, 31–42.
- Bryan, H.K.; Olayanju, A.; Goldring, C.E.; Park, B.K. The Nrf2 Cell Defence Pathway: Keap1-Dependent and -Independent Mechanisms of Regulation. Biochem. Pharmacol. 2013, 85, 705–717.
- Fahey, J.W.; Wehage, S.L.; Holtzclaw, W.D.; Kensler, T.W.; Egner, P.A.; Shapiro, T.A.; Talalay, P. Protection of Humans by Plant Glucosinolates: Efficiency of Conversion of Glucosinolates to Isothiocyanates by the Gastrointestinal Microflora. Cancer Prev. Res. 2012, 5, 603–611.
- Simon, V.; Cota, D. MECHANISMS IN ENDOCRINOLOGY: Endocannabinoids and Metabolism: Past, Present and Future. Eur. J. Endocrinol. 2017, 176, R309–R324.




