
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Seeram Ramakrishna | + 1826 word(s) | 1826 | 2021-06-10 08:31:43 | | | |
| 2 | Catherine Yang | Meta information modification | 1826 | 2021-06-22 03:56:21 | | | | |
| 3 | Catherine Yang | Meta information modification | 1826 | 2021-06-23 10:11:02 | | |
Video Upload Options
Atomization is an intricate operation involving unstable and complex networks with rupture and fusion of liquid molecules. There are diverse details that typify the spray formation, which are the technique and configuration of the atomization process, dimension and structure of the nozzle, experimental parameters, etc. .
1. Introduction
The process of the destruction of feed liquid into drops or fine spray by the influence of surface tension together with interior or exterior forces is referred to as atomization. When there is no influence of disturbances, the surface tension of the liquid will pull it into a sphere-shape in which the surface energy is less. When it is disturbed with other forces, the surface tension will result in instability that leads to the destruction of feed liquid (thin ligaments or sheets of liquid) into smaller drops [1]. Some examples of atomization that we come across in our day-to-day life are antiperspirant or hair spray, shower head, garden sprinkler, cologne mist, etc. The outcome of the atomization process is to generate spray, which is a gathering of fluid beads that are in an organized motion and each sphere-shaped drop is also known as a particle [2]. Thus, to generate particles of preferred thinness, the measure of the external force or any kind of force that causes disruption, should either level or top the force applied by the surface tension of the particular fluid [1]. There are factors that affect the atomization process and its result of the fine spray of liquid droplets. One of the major factors is the properties of the fluid that is being used in the process and those are surface tension, density and viscosity of the fluid. All the three have a similar effect, that is the higher these properties, the larger the droplets. Another factor that can affect this is the temperature at which atomization is taking place. The temperature has an inversely proportional effect on surface tension of the fluid, i.e., higher temperature reduces the surface tension of the fluid and thus affects the droplet size [2].
2. Kinetics of Liquid Jets and Sheets
Even though there are literature surveys on various atomization processes [3][4][5][6], the complexity of the technique, the possession of the atomizer on the droplet qualities, imprecision in computation strategies are still not well understood [7][8]. The type of nozzle and the pressure determines whether a jet or sheet is obtained before atomization. Since, there is liquid jet or sheet; instability of liquid jet and liquid sheet are considered for the formation of the droplets. These theories are also used to study the growth of perturbation waves, which is used to understand atomization to generate drops by the breakup of these liquid jets and liquid sheets [9].
The liquid jet originated from the nozzle comes in contact with the surrounding gas, and this jet is broken down into drops when put through interferences like pressure variation, surface shift, velocity change of the apparatus or the liquid jet and also variations in temperature, consistency of the liquid and feature of the liquid surface to endure superficial exertion [10][11][12]. These interferences cause instability in the liquid jet causing disturbance waves in it and the instability is characterized by studying the development of this wave, droplet emergence and rupture length of the jet [13][14]. The pressure variation is obtained by using acoustic waves, change in velocity is obtained by vibrating the flow, temperature is varied by providing regular heating and the surface is disturbed by using an electrifying sensor [9]. Rayleigh studied the vulnerability of the liquid jet and he procured an equation to calculate the growth rate of the disturbances that cause instability in the jet.
Studies on instability of a liquid jet reveal that a required wavenumber is necessary to generate stable liquid jet and when the wavenumber is less than the required value, it generates an unstable jet leading the breakup of the jet into a large droplet and one or more small drops. Figure 1 represents such a breakup of the liquid jet. By adjusting the interferences, we can generate droplets without the small drops (satellites).
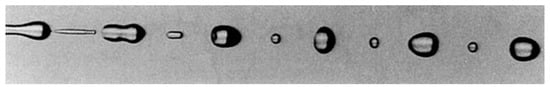
Figure 1. Instability of liquid jet with satellite drops; reproduced with permission from [15].
Later in 1931, Weber introduced the effect of viscosity in the exploration of jet split-up using the 3D partial differential equations of Newtonian viscous fluid and found that viscosity of the fluid plays a role in slowing down the growth rate of the interference and thus elongating the wavelength, without altering the wavenumber [16]. Similarly, Bogy [17] and Sirignano et al. [18] are the two other groups of scientists who provided the theoretical examination of the instability of the liquid jet.
Like liquid jets, in some cases liquid sheets originate from the nozzle and this sheet will be split into small ligaments through oscillations. The ligaments are then broken into small droplets to form a fine spray. The size of the droplets in this spray will directly depend on the thickness of the liquid sheet that initially originated [9].
The instability of the liquid sheet can be studied by relating to time and space occupied and there are many literatures explaining the mechanism of instability [19][20][21]. were the group of scientists who studied the impact of various factors in fracturing the liquid sheet into droplets and they realized that the liquid sheets with an elevated velocity are encountering a wave motion [22][23][24]. In 1999, Senecal et al. studied the mechanism of instability of a viscous fluid sheet in gas and established two well-defined systems for the atomization of the liquid sheet. One system is with a very low Weber number, the liquid sheet is disrupted by long waves and in this case the viscosity of the liquid is not considered.
3. Atomization Processes
Table 1 briefly describes the different types of atomization processes and its characteristics.
Table 1. Different atomization processes and its characteristics; Based on [1][2][25][26][27][28][29][30].
| Types | Energy Source | Mechanism | Class of Spray | Advantages | Disadvantages |
|---|---|---|---|---|---|
| Hydraulic | Fluid pressure | Pressure exerted on the fluid drives it through the nozzle to generate fluid sheets with high velocity and leading its disruption to fragments and then to droplets by friction between liquid and air | Non-uniform, rough | Economical, utilize small rooms for drying | Not relevent for viscous fluids, wide range of droplet size dispersion |
| Pneumatic | Air pressure | Fluid at low velocity passing through the nozzle is surrounded by high velocity flow of air, boosting friction between two medium causing disruption of fluid sheet | Heterogenous, average roughness | Uniform droplets, applicable for viscous fluids, superior productivity | Expensive, ensuing instability |
| Rotary | Centrifugal force | Atomizer has a spinning disc at the center to which the fluid is introduced through the nozzle; centrifugal force takes the fluid to the margin of the disc and flip off the boundary setting up ligaments that then breaks into droplets | Heterogenous | Uniform droplets, no clogging of atomizer, superior productivity | Expensive, not relevant for viscous fluids, requires larger rooms for drying |
| Electrostatic | Electric charge | An electric field applied between atomizer and workpiece to make it conductive; fluid passed through the electric field and the repulsive force disrupt the fluid into droplets and is gathered at the workpiece | Finer, homogenous | Fine & uniform droplets, no clogging of atomizer | Varying film thickness due to diverse electrostatic excitation in the core & shell of the system |
| Ultrasonic | Electromechanical device | Fluid is passed through a vibrating electromechanical device causing the disruption of the fluid into droplets | Very fine and homogenous | Control spray size by altering the vibrational frequency | Not relevant for viscous fluids, restriction in scaling up of the system |
Figure 2 represents the different types of commercially used atomizers, the device used for the atomization process to develop atomized sprays for various applications.
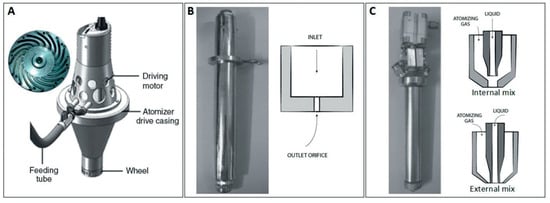
Figure 2. Schematic representation of atomizers used in: (A) rotary atomization, (B) hydraulic atomization and (C) pneumatic atomization; reproduced from [15].
4. Electrospray Atomization and Its Characteristics
Electro–hydro dynamic atomization is also known as electrostatic/electrospray atomization and it is usually considered as an alternative methodology to other techniques utilizing heat and some chemical substances. Electrospray atomization is the basic principle behind electrospinning (which gives rise to fibres) and electrospray (initiating fine spray containing particles). Electrospraying is the most widely used procedure in which a very high voltage electric current is applied to influence the liquid stream coming out of an orifice to either form a cone-jet which is the most common, dripping or multi-jet and ultimately generating long fibres or small droplets based on its application [31]. Micro/nanofibers are the result of electrospinning which has a range of applications in the field of tissue engineering [32], cancer therapy [33], antibacterial coating [34], etc.
Electro hydrodynamic atomization was first studied in the year 1600 by the scientist Gilbert [35] and later Rayleigh theoretically and experimentally calculated the maximum electric charge that a liquid droplet can carry and he named it as Rayleigh limit after his name [36]. In 1960, Taylor mathematically explained the development of cone-jet in the liquid stream in the action of an electric current and the cone is known as Taylor cone [37]. Further modification in the experimental parameters lead to the development of biomolecules [38] with a range of applications in different fields.
Electrospray constitutes a syringe setup with a metallic needle to which an electric current is connected from a high voltage generator. The needle of the syringe acts as an orifice through which the fluid comes out. The syringe is connected to a syringe pump with which we can apply the required pressure to push the working fluid in the syringe.
Sometimes the whole setup explained above will be enclosed in a chamber through which air or nitrogen will be allowed to flow continuously so that the atomized particles will be transported to the collector region. This also enhances the production of small, smooth and uniform droplets [39][40]. The forces acting on the Taylor cone are the gravitational pull which will be negligible if the viscosity of the working fluid is high, viscosity and surface tension of the fluid and the working parameter that is the electric field.
Table 2 compares electrospray to other common techniques in generating micro/nano polymeric particles applied for drug delivery applications.
Table 2. Comparing electrospray to other common techniques.
| Techniques | Merits | Challenges | Ref |
|---|---|---|---|
| Electrospray | One step process; confined particle size that is micro and nanoparticles; polymers with higher molecular weights can be employed; surfactant free; utilizes less solvents; drugs that are slightly soluble in water can be processed | Low yield; sometimes requires cross-linking factors; advancing the technique for bulk manufacturing is not possible | [41] |
| Spray drying | Develops inorganic polymeric microparticles; scale-up production | Uses higher temperature gases as transporter; denatures heat sensitive substances | [42][43] |
| Nanoprecipitation | Easy procedure; nanoparticles generated by desorption; efficiently encloses hydrophobic drugs; surfactant free | Utilizes substantial amount of solvent; drug loaded in the particles are low | [44] |
| Emulsion solvent vaporization | Adaptable technique; generates diverse biomolecular particles | Only uses low molar mass polymers; not free of surfactants and solvents; not a single step technique; wide range of particle size | [41] |
| Sheet by sheet fabrication | Accurate; multi-tiered particles; uniform sheet thickness; regulated drug delivery | Tiresome and lengthy process; advancing the technique for bulk manufacturing is not possible | [45] |
References
- Lapple, C.E.; Henry, J.P.; Blake, D.E. Atomization—A Survey and Critique of the Literature; Special Report; Department of the Army: Edgewood Arsenal, MD, USA, 1967; pp. 11–14. Available online: (accessed on 6 March 2021).
- Graco, Atomization Concept and Theory, 1–18. Available online: (accessed on 6 March 2021).
- Lefebvre, A.H.; McDonell, V.G. Atomization and Sprays; CRC Press: Boca Raton, FL, USA, 2017; Available online: (accessed on 14 March 2021).
- Kooij, S.; Astefanei, A.; Corthals, G.L.; Bonn, D. Size distributions of droplets produced by ultrasonic nebulizers. Sci. Rep. 2019, 9, 6128.
- Lefebvre, A.H. Airblast atomization. Prog. Energy Combust. Sci. 1980, 6, 233–261.
- Faeth, G.; Hsiang, L.-P.; Wu, P.-K. Structure and breakup properties of sprays. Int. J. Multiph. Flow 1995, 21, 99–127.
- Desjardins, O.; McCaslin, J.; Owkes, M.; Brady, P. Direct numerical and large-eddy simulation of primary atomization in complex geometries. At. Sprays 2013, 23, 1001–1048.
- Ramamurthi, K.; Tharakan, T.J. Flow Transition in Swirled Liquid Sheets. AIAA J. 1998, 36, 420–427.
- Ashgriz, N. Handbook of Atomization and Sprays—Theory and Applications; Springer: Toronto, ON, Canada, 2011; Chapters 1 & 3; Available online: (accessed on 5 March 2021).
- McCarthy, M.; Molloy, N. Review of stability of liquid jets and the influence of nozzle design. Chem. Eng. J. 1974, 7, 1–20.
- Bazilevskii, A.V.; Rozhkov, A.N. Dynamics of capillary breakup of elastic jets. Fluid Dyn. 2014, 49, 827–843.
- Mashayek, F.; Ashgriz, N. Instability of liquid coatings on cylindrical surfaces. Phys. Fluids 1995, 7, 2143–2153.
- Donnelly, R.J.; Glaberson, W. Experiments on the Capillary Instability of a Jet. Proc. R. Soc. Lond. A 1966, 209, 547–556.
- Goedde, E.F.; Yuen, M.C. Experiments on liquid jet instability. J. Fluid Mech. 1970, 40, 495–511.
- Vassallo, P.; Ashgriz, N. Satellite formation and merging in liquid jet breakup. Proc. R. Soc. Lond. Ser. A 1991, 433, 269–286.
- Weber, C. To the disintegration of a jet of liquid. ZAMM J. Appl. Math. Mech. 1931, 11, 136–154.
- Bogy, D.B. Drop Formation in a Circular Liquid Jet. Annu. Rev. Fluid Mech. 1979, 11, 207–228.
- Sirignano, W.; Mehring, C. Review of theory of distortion and disintegration of liquid streams. Prog. Energy Combust. Sci. 2000, 26, 609–655.
- Michell, J.H. On the Theory of Free Stream Lines. Philos. Trans. R. Soc. A. 1890, 181, 389–431. Available online: (accessed on 15 March 2021).
- Hagerty, W.W.; Shea, J.F. A Study of the Stability of Plane Fluid Sheets. J. Appl. Mech. 1955, 22, 509–514.
- Rangel, R.H.; Sirignano, W.A. The Linear and Nonlinear Shear Instability of a Fluid Sheet. Phys. Fluids 1999, 3, 2392–2400.
- Dombrowski, N.; Fraser, R.P. A Photographic Investigation into the Disintegration of Liquid Sheets. Phil. Trans. 1954, 247, 101–130.
- Dombrowski, N.; Hasson, D.; Ward, D. Some aspects of liquid flow through fan spray nozzles. Chem. Eng. Sci. 1960, 12, 35–50.
- Dombrowski, N.; Hooper, P. The effect of ambient density on drop formation in sprays. Chem. Eng. Sci. 1962, 17, 291–305.
- Santos, D.; Mauricio, A.C.; Sencadas, V.; Santos, J.D.; Fernandez, M.H.; Gomes, P.S. Spray Dryin—An Overview. In Biomaterials—Physics and Chemistry—New Edition; IntechOpen: London, UK, 2017; Chapter 2; pp. 9–35.
- Vasilyev, A.Y.; Domrina, E.S.; Kaufman, S.V.; Maiorova, A.I. Classification of Atomization Devices. J. Phys. Conf. Ser. 2019, 1359, 012131.
- Cal, K.; Sollohub, K. Spray Drying Technique. I: Hardware and Process Parameters. J. Pharm. Sci. 2010, 99, 575–586.
- Anandharamakrishnan, C.; Ishwarya, S.P. Spray Drying Techniques for Food Ingredient Encapsulation; John Wiley & Sons: Hoboken, NJ, USA, 2015; Available online: (accessed on 16 March 2021).
- Barbosa-Canovas, G.V.; Ortega-Rivas, E.; Juliano, P.; Yan, H. Food Powders: Physical Properties, Processing, and Functionality; Springer: Berlin/Heidelberg, Germany, 2005.
- Anandharamakrishnan, C. Handbook of Drying for Dairy Products; John Wiley & Sons: Hoboken, NJ, USA, 2017; Available online: (accessed on 8 March 2021).
- Chen, C.; Liu, W.; Jiang, P.; Hong, T. Coaxial Electrohydrodynamic Atomization for the Production of Drug-loaded Micro/Nanoparticles. Micromachines 2019, 10, 125.
- Rahmati, M.; Mills, D.K.; Urbanska, A.M.; Saeb, M.R.; Venugopal, J.R.; Ramakrishna, S.; Mozafari, M. Electrospinning for tissue engineering applications. Prog. Mater. Sci. 2021, 117, 100721.
- Abid, S.; Hussain, T.; Raza, Z.A.; Nazir, A. Current applications of electrospun polymeric nanofibers in cancer therapy. Mater. Sci. Eng. C 2019, 97, 966–977.
- Soo, J.Z.; Chai, L.C.; Ang, B.C.; Ong, B.H. Enhancing the Antibacterial Performance of Titanium Dioxide Nanofibers by Coating with Silver Nanoparticles. ACS Appl. Nano Mater. 2020, 3, 5743–5751.
- Gilbert, W. Gilbert’s Book on Magnets, De Magnete, Londini, Anno MDC. 1600. Available online: (accessed on 18 March 2021).
- Rayleigh, L. On the Instability of Jets. Proc. Lond. Math. Soc. 1878, 10, 4–13.
- Taylor, G. The Force Exerted by an Electric Field on a Long Cylindrical Condutor. Proc. Royal Soc. A 1966, 291, 145–158.
- Grayson, M.A. John Bennett Fenn: A Curious Road to the Prize. J. Am. Soc. Mass Spectrom. 2011, 22, 1301–1308.
- Xie, J.; Lim, L.K.; Phua, Y.; Hua, J.; Wang, C.-H. Electrohydrodynamic atomization for biodegradable polymeric particle production. J. Colloid Interface Sci. 2006, 302, 103–112.
- Xie, J.; Ng, W.J.; Lee, L.Y.; Wang, C.H. Encapsulation of Protein Drugs in Biodegradable Microparticles by Coaxial Electrospray. J. Colloid Interface Sci. 2008, 317, 469–476.
- Hernandez, J.A.T.; Chavez, P.I.T.; Wong, B.R.; Chu, A.R.; Jatomea, M.P.; Urbina, C.G.B.; Vazquez, N.A.R.; Felix, F.R. Micro and Nanoparticles by Electrospray: Advances and Applications in Foods. J. Agric. Food Chem. 2015, 63, 4699–4707.
- Ré, M.-I. Formulating Drug Delivery Systems by Spray Drying. Dry. Technol. 2006, 24, 433–446.
- Peltonen, L.; Valo, H.; Kolakovic, R.; Laaksonen, T.; Hirvonen, J. Electrospraying, Spary Drying and Related Techniques for Production and Formulation of Drug Nanoparticles. Expert Opin. Drug Deliv. 2010, 7, 705–719.
- Bilati, U.; Allémann, E.; Doelker, E. Development of a nanoprecipitation method intended for the entrapment of hydrophilic drugs into nanoparticles. Eur. J. Pharm. Sci. 2005, 24, 67–75.
- Hammond, P.T. Building biomedical materials layer-by-layer. Mater. Today 2012, 15, 196–206.




