Your browser does not fully support modern features. Please upgrade for a smoother experience.

Submitted Successfully!
Thank you for your contribution! You can also upload a video entry or images related to this topic.
For video creation, please contact our Academic Video Service.
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Satriya Pranata | + 2102 word(s) | 2102 | 2021-06-21 08:18:35 | | | |
| 2 | Satriya Pranata | Meta information modification | 2102 | 2021-06-21 12:11:13 | | |
Video Upload Options
We provide professional Academic Video Service to translate complex research into visually appealing presentations. Would you like to try it?
Cite
If you have any further questions, please contact Encyclopedia Editorial Office.
Pranata, S. Precision Health Care for Diabetes. Encyclopedia. Available online: https://encyclopedia.pub/entry/11073 (accessed on 08 February 2026).
Pranata S. Precision Health Care for Diabetes. Encyclopedia. Available at: https://encyclopedia.pub/entry/11073. Accessed February 08, 2026.
Pranata, Satriya. "Precision Health Care for Diabetes" Encyclopedia, https://encyclopedia.pub/entry/11073 (accessed February 08, 2026).
Pranata, S. (2021, June 21). Precision Health Care for Diabetes. In Encyclopedia. https://encyclopedia.pub/entry/11073
Pranata, Satriya. "Precision Health Care for Diabetes." Encyclopedia. Web. 21 June, 2021.
Copy Citation
Tailoring integrated care through interdisciplinary collaborative practice among patients, nurses, and physicians based on the patient’s genetics or lifestyle, glycemic target, biodata- or evidence-based practice, patient preferences, and priority for improving patient self-management to achieve glycemic control.
personal health services
diabetes mellitus
glycemic control
patient care team
precision health care
1. Introduction
Diabetes is a chronic disease and a major health concern in modern society. diabetes is typically characterized by an abnormal increase in glucose levels caused by one or two mechanisms: inadequate insulin production by the pancreas or inadequate cell sensitivity to the action of insulin caused by the reduced function of insulin receptors. In 2019, an estimated 463 million patients had diabetes. By 2045, this number is predicted to increase to 700.2 million [1].
Other data indicate that people with diabetes have a greater risk of experiencing various complications than other individuals. People with diabetes are 2–3 times more likely to develop cardiovascular diseases and up to 10 times more likely to develop end-stage renal disease and part or complete amputation of a lower limb; further, an amputation is performed somewhere in the world every 30 s. In 2019, the total health care expenditures on diabetes for people aged 20–79 years were an estimated US$760 billion, such expenditure may reach US$825 billion in 2030 and US$845 in 2045 [1][2].
Hospital- and community-based interventions to avoid diabetes-related complications have been based on evidence-based practice and guidelines for diabetes care. A guideline typically provides a set of recommendations along with eligibility criteria that restrict their applicability to a specific group of patients for disseminating such knowledge and standardizing care to ensure the highest quality of care [3]. Furthermore, interventions based on hospital guidelines involve the promotion of self-efficacy, health education, self-management, and health coaching [4][5]. Guidelines and evidence-based practice are crucial for defining the quality of care. Nevertheless, in certain situations, deviating from such guidelines and practice is desirable and helps address the needs and peculiarities of patients with diabetes. Specifically, with the availability of health data related to patients with diabetes, precise identification based on treatment demands and targets can be executed, posing challenges on diabetes care to clinical guideline recommendations. However, studies related to preference regarding integrated individualized health care in diabetes are limited.
Research efforts in this direction are termed “precision medicine”. The vision of precision medicine is that this medication is predictive, preventive, personalized, and participatory [6][7]. Moreover, doctors and researchers adopt treatment and prevention strategies more accurately through precision medicine and consider differences between individuals rather than the one-size-fits-all approach [8]. As an individual’s experience of both health and disease is unique at the molecular, cellular, and organ levels, treating the causes rather than the symptoms of diseases is achievable [9]. The precision medicine approach is becoming a trend in clinical settings, especially after former US President Obama’s launch of the Precision Medicine Initiative in early 2015. The primary aim of precision medicine is to improve clinical outcomes for individual patients through precise treatment targeting by leveraging genetic, biomarker, phenotypic, or psychosocial characteristics that distinguish a given patient from others with similar clinical presentations [3].
In addition, the terminologies defined above can be applied to other approaches in the form of precision health care (PHC). PHC involves patient care preferences, patient-oriented care, evidence-based care, and self-management [10][11][12]. PHC is a care delivery model that relies heavily on data, analytics, and personal information [11][13].
2. Discovery of the Elements and Their Concept Description
We discovered the PHC elements for diabetes through the literature study and discussed the concept description of eight elements. We identified the concepts by extracting the data in each article, comparable definitions used in precision medicine and health then integrated the results. Therefore, we reached a final consensus after discussion with the research team. The concept description and strategy of each element can be seen in Table 1 and are explained below.
Table 1. Elements, Concept Descriptions, and Clinical Strategies on PHC for Diabetes.
| No | Elements | Concept Description | Clinical Strategies |
|---|---|---|---|
| 1. | Personalized genetic or lifestyle | - Genetic or lifestyle analysis; genomic test screening for diabetes autoantibodies that remain after a drug or insulin dose, gene encoding glucokinase, presence of HNF1A and HNF4A that are associated with forms of diabetes onset; C-peptide is a biomarker that can be used as a guide to treatment choice (insulin deficiency); single-nucleotide polymorphisms provide information regarding drug toxicity | - Assessment of risk of complication by using risk prediction charts, genotype, or electronic health records |
| 2. | Biodata-or evidence-based | - Genetic examination to detect various potential health problems, cardiovascular disease, a person’s metabolic ability to a nutrient, and HbA1c target | - Electronic health records and ADA guidelines |
| 3. | Glycemic target | - Based on ADA guidelines, target and therapy differ based on the features and responses of each individual (including HbA1c, blood pressure, and cholesterol) | - Shared decision-making assessment tool |
| 4. | Patient preferences | - Identification of whether the patient needs additional medication and their concern regarding hyper/hypoglycemia, further expressing their decision | - Shared decision-making assessment tool |
| 5. | Glycemic control | - Supporting the use of a potent drug to achieve a reduction in HbA1c to <6.5%. | - HbA1c based on ADA guidelines |
| 6. | Interdisciplinary collaboration practice | - Teamwork entails discussion of the most appropriate treatment for patients | - Shared decision-making among patients, nurses, physicians, etc. |
| 7. | Self-management | - Individualizing therapy so that patients can effectively self-manage their disease through increasing self-efficacy | - Diabetes SM education, self-efficacy enhancing intervention program |
| 8. | Patient priority direct care | - Assess the individual as a whole including the complex interplay of comorbid conditions, psychosocial, functional status, and individual need | - Shared decision-making assessment tool |
Note: ADA: American Diabetes Association; SM: self-management.
2.1. Personalized Genetic or Lifestyle
With the advances in medical technology, various new methods continue to emerge, including the trend of genetic testing or genomic test to determine a person’s genetic profile. Genetic or lifestyle analysis helps delineate complication risk, glycemia tolerance, the side effect of diabetes treatment through gender analysis, age, comorbid condition, cholesterol, and blood pressure [14][15]. The genomic test can be used for screening for diabetes autoantibodies remains drug or insulin dose. Furthermore, a genomic test is performed by observing the gene encoding glucokinase, HNF1A, and HNF4A, which are associated with diabetes onset [16]. The patient genotype influences response to the side effect of diabetes treatment, and C-peptide is a biomarker to guide treatment choice (insulin deficiency). Moreover, single-nucleotide polymorphism provides information regarding drug toxicity [17][14][16]. Genetic or lifestyle factors can be assessed during PHC for diabetes by using risk prediction charts [18]. Furthermore, data on genotype or electronic health records from hospital records can be used to determine the most appropriate diabetes care for patients [11][19].
2.2. Biodata- or Evidence-Based Requirement
Health profiling should further include other social and environmental data of the patient. The PHC approach can provide precise therapy to patients based on their genetic characteristics [8]. Moreover, genetic examination can detect various potential health problems, including cardiovascular disease and a person’s metabolic ability toward a nutrient. Furthermore, electronic health medical records and health insurance databases contain rich clinical information, including data from remote sources to identify disease risk factors through computation and bioinformatic methods [20][21].
2.3. Glycemic Target
According to American Diabetes Association (ADA) guidelines, target and therapy differ based on each individual's features and responses, namely, (A1c, Blood pressure, and Cholesterol). Factors that must be considered in PHC are patient age, individual treatment goals, HbA1c on current clinical data, lifestyle, and physical activity of patients [22]. The shared decision-making assessment tool can be used to assess patient goals based on their preferences [23].
2.4. Patient Preferences
2.5. Glycemic Control
A more potent drug can be used to achieve a greater reduction in HbA1c to <6.5% [22]. Individual GTs based on ADA consensus in addition to patient-specific factors are approximately <6.5%, <7%, and <8% [24]. In Precision health care, the participant and facilitator will set a goal and develop a detailed written action plan for achieving glycemic control in reducing HbA1C. All medication and nursing management of patients will be tailored based on patient needs, priorities, and preferences. Patient difficulties, frequency, and intensity of intervention for the action plan will discuss with patients.
2.6. Interdisciplinary Collaboration Practice
Interdisciplinary collaboration can be accomplished if all professionals from different backgrounds learn to work together as a part of the health care team [25]. Teamwork entails discussion to decide on the most appropriate treatment for patients, which can then be shared through decision-making tools [14][26][27].
2.7. Self-Management
Diabetes self-management education, self-efficacy enhancing intervention program, self-management are ongoing processes comprising strategies to help individuals with chronic conditions, and their families and caregivers, to better understand and manage their illness and enhance health behaviour, [28]. Self-management in PHC includes focusing on self-identified needs or problems that require continual monitoring and taking appropriate actions [4]. Individualizing therapy allows patients to effectively self-manage their disease through increased self-efficacy [4][29].
2.8. Patient Priority Direct Care
We can improve outpatient care decisions through improved support systems that prioritize care recommendations and enhance communication of treatment-relevant information to patients with diabetes [20]. For example, through shared decision-making in clinical practice, the patient condition can be improved through consideration of the whole individual, including the complex interplay of comorbid conditions, psychosocial and functional status, and individual needs [29][20]. Table 1 presents the elements, concept description, and clinical strategies on PHC for diabetes.
3. Definition of PHC
We integrated the elements in the literature for an operational definition of PHC and PHC for patients with diabetes as below.
We identified six elements of PHC, namely purpose of health, care preferences, patient priority direct care, interdisciplinary collaborative practice, popular biodata- or evidence-based practice, and patient self-management [10][11][13][25]. Therefore, we defined PHC as providing integrated care based on individual needs, including interdisciplinary cooperation and patient involvement in decision-making regarding health goals, providing care that meets patient expectations and preferences, providing patient-oriented care, and using biodata as evidence-based care disposal to improve patient self-management.
Moreover, based on the comprehensive literature analysis and data integration, we summarized that personalized genetic or lifestyle, biodata- or evidence-based, glycemic target, patient preferences, glycemic control, interdisciplinary collaboration practice, self-management, and patient priority direct care were the core elements for patients with diabetes. Furthermore, the operational definition of PHC for diabetes was tailoring integrated care through interdisciplinary collaborative practice among patients, nurses, and physicians based on the patient’s genetics or lifestyle, glycemic target, biodata- or evidence-based practice, patient preferences, and priority for improving patient self-management to achieve glycemic control.
4. Strategies of PHC for Diabetes in Clinical Practice
After discovering the PHC elements and defining them for diabetes, we developed strategies for the application of these elements for treating diabetes in clinical practice. The strategies can be elaborated as follows:
SDM became crucial in PHC because, through this method, patients with diabetes have an opportunity to choose either insulin injection or oral drugs for glucose control; furthermore, health professionals must discuss the best suitable treatment with patients. The shared decision-making process, which combines knowledge, communication, and respect to establish a glycemic target and medical consensus, is based on patients’ preferences and values. Shared decision-making can be achieved by using the shared decision-making tool for assistance to implement PHC. When analyzing elements of PHC for diabetes, clinical strategies or intervention for clinical application is essential. The clinical strategies or interventions are illustrated in Figure 1.
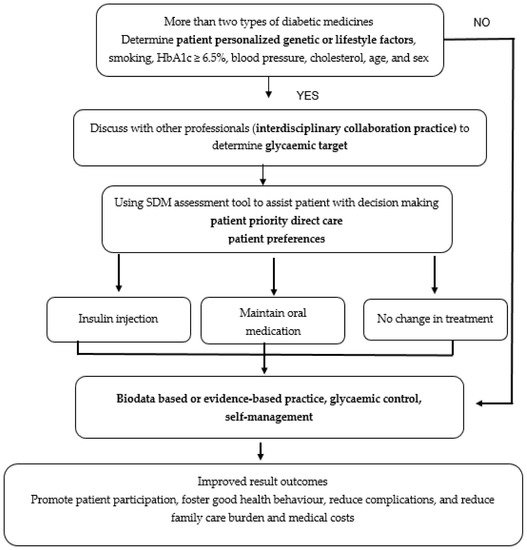
Figure 1. Strategies to clinically apply eight PHC elements for treating patients with diabetes.
Figure 1 shows an example and the process of applying PHC for diabetes based on eight elements. Figure 1 explains the process in a real condition at a hospital or a clinical setting, wherein patients often show HbA1c values ≥ 6.5% despite taking two types of medication namely oral drug medication and insulin injection. The patient and health professionals should discuss and analyze the most appropriate treatment using the following five steps, which forms the acronym SHARE: (1) Seek patients’ participation by describing the problem clearly and openly to make the patients understand that a decision needs to be made; (2) Help patients explore and compare treatment options; (3) Assess the patient’s values and preferences; (4) Reach a decision with the patient; (5) Evaluate the patient’s decision of using insulin injection, maintaining oral medication, or no treatment change [30]. In addition, patients must be encouraged to use a more potent drug to achieve a greater reduction in HbA1c (<6.5%) based on patient-specific factors (age, individual treatment goals, HbA1c ADA guidelines, current clinical data, diet, lifestyle, and physical activity). It cannot be overstated that PHC allows patients to effectively self-manage their disease by increasing self-efficacy. Result outcomes can be improved through the promotion of patient participation, good health behaviour, and reduced complications, family care burden, and medical costs.
References
- IDF. International Diabetes Federation Diabetes Atlas, 9th ed.; Innis Communication: Brussels, Belgium, 2019; Available online: (accessed on 11 January 2020).
- WHO. World Health Organization/International Society of Hypertension (WH0/ISH) Risk Prediction Charts; WHO: Geneva, Switzerland, 2014; pp. 1–40. Available online: (accessed on 13 March 2020).
- Parimbelli, E.; Marini, S.; Sacchi, L.; Bellazzi, R. Patient similarity for precision medicine: A systematic review. J. Biomed. Inform. 2018, 83, 87–96.
- Sherifali, D.; Viscardi, V.; Bai, J.W.; Ali, R.M.U. Evaluating the Effect of a Diabetes Health Coach in Individuals with Type 2 Diabetes. Can. J. Diabetes 2016, 40, 84–94.
- Sherifali, D. Diabetes coaching for individuals with type 2 diabetes: A state-of-the-science review and rationale for a coaching model. J. Diabetes 2017, 9, 547–554.
- Weston, A.D.; Hood, L. Systems Biology, Proteomics, and the Future of Health Care: Toward Predictive, Preventative, and Personalized Medicine Introduction: Paradigm Changes in Health Care. J. Prot. Res. 2004, 3, 179–196.
- Hood, L.; Balling, R.; Auffray, C. Revolutionizing medicine in the 21st century through systems approaches. Biotechnol. J. 2012, 7, 992–1001.
- Jameson, J.L.; Longo, D.L. Precision medicine—Personalized, problematic, and promising. N. Engl. J. Med. 2015, 372, 2229–2234.
- Flores, M.; Glusman, G.; Brogaard, K.; Price, N.D.; Hood, L. P4 medicine: How systems medicine will transform the healthcare sector and society. Per. Med. 2013, 10, 565–576.
- Bierman, A.S.; Tinetti, M.E. Precision medicine to precision care: Managing multimorbidity. Lancet 2016, 388, 2721–2723.
- Corwin, E.; Redeker, N.S.; Richmond, T.S.; Docherty, S.L.; Pickler, R.H. Ways of knowing in precision health. Nurs. Outlook 2019, 67, 293–301.
- Davies, A.K.; McGale, N.; Humphries, S.E.; Hirani, S.P.; Beaney, K.E.; Bappa, D.A.S.; McCabe, J.G.; Newman, S.P. Effectiveness of a self-management intervention with personalised genetic and lifestyle-related risk information on coronary heart disease and diabetes-related risk in type 2 diabetes (CoRDia): Study protocol for a randomised controlled trial. Trials 2015, 16, 1–11.
- Tinetti, M.E.; Esterson, J.; Ferris, R.; Posner, P.; Blaum, C.S. Patient Priority-Directed Decision Making and Care for Older Adults with Multiple Chronic Conditions. Clin. Geriatr. Med. 2016, 32, 261–275.
- Spiegel, A.M.; Hawkins, M. “Personalized medicine” to identify genetic risks for type 2 diabetes and focus prevention: Can it fulfill its promise? Health Aff. 2012, 31, 43–49.
- Floyd, J.S.; Psaty, B.M. The application of genomics in diabetes: Barriers to discovery and implementation. Diabetes Care 2016, 39, 1858–1869.
- Pearson, E.R. Personalized medicine in diabetes: The role of “omics” and biomarkers. Diabet. Med. 2016, 33, 712–717.
- Meneghini, L.; Reid, T. Individualizing insulin therapy. J. Fam. Pract. 2012, 61, 13–27.
- Otgontuya, D.; Oum, S.; Buckley, B.S.; Bonita, R. Assessment of total cardiovascular risk using WHO/ISH risk prediction charts in three low and middle income countries in Asia. BMC Public Health 2013, 13, 539.
- Hickey, K.T.; Bakken, S.; Byrne, M.W.; Bailey, D.C.E.; Demiris, G.; Docherty, S.L.; Dorsey, S.G.; Guthrie, B.J.; Heitkemper, M.M.; Jacelon, C.S.; et al. Precision Health: Advancing Symptom and Self-Management Science [Internet]; Nursing Outlook; Elsevier Inc.: Amsterdam, The Netherlands, 2019; Volume 67, pp. 462–475.
- Holt, R.I.G. Personalized medicine for diabetes: A special issue. Diabet. Med. 2016, 33, 711.
- Mayor, S. Individualising treatment and care of patients with diabetes. Prescriber 2017, 28, 23–25.
- Subramanian, S.; Hirsch, I.B. Personalized diabetes management: Moving from algorithmic to individualized therapy. Diabetes Spectr. 2014, 27, 87–91.
- Paschou, S.A.; Leslie, R.D. Personalizing guidelines for diabetes management: Twilight or dawn of the expert? BMC Med. 2013, 11, 161.
- Miñambres, I.; Mediavilla, J.J.; Sarroca, J.; Pérez, A. Meeting individualized glycemic targets in primary care patients with type 2 diabetes in Spain. BMC Endocr. Disord. 2016, 16, 10.
- Sexton, M.; Baessler, M. Interprofessional Collaborative Practice. J. Contin. Educ. Nurs. 2016, 47, 156–157.
- Fradkin, J.E.; Hanlon, M.C.; Rodgers, G.P. NIH precision medicine initiative: Implications for diabetes research. Diabetes Care 2016, 39, 1080–1084.
- Rich, S.S.; Cefalu, W.T. The impact of precision medicine in diabetes: A multidimensional perspective. Diabetes Care 2016, 39, 1854–1857.
- Wu, S.F.V.; Lee, M.C.; Liang, S.Y.; Lu, Y.Y.; Wang, T.J.; Tung, H.H. Effectiveness of a self-efficacy program for persons with diabetes: A randomized controlled trial. Nurs. Health Sci. 2011, 13, 335–343.
- Sherifali, D. Diabetes Management in Older Adults: Seeing the Forest for the Trees. Can. J. Diabetes 2016, 40, 10–11.
- Agency for Healthcare Research Quality (AHRQ). The SHARE Approach Essential Steps of Shared Decision Making; Agency for Healthcare Research Quality: Rockville, MD, USA, 2016.
More
Information
Subjects:
Medicine, General & Internal
Contributor
MDPI registered users' name will be linked to their SciProfiles pages. To register with us, please refer to https://encyclopedia.pub/register
:
View Times:
936
Revisions:
2 times
(View History)
Update Date:
21 Jun 2021
Notice
You are not a member of the advisory board for this topic. If you want to update advisory board member profile, please contact office@encyclopedia.pub.
OK
Confirm
Only members of the Encyclopedia advisory board for this topic are allowed to note entries. Would you like to become an advisory board member of the Encyclopedia?
Yes
No
${ textCharacter }/${ maxCharacter }
Submit
Cancel
Back
Comments
${ item }
|
More
No more~
There is no comment~
${ textCharacter }/${ maxCharacter }
Submit
Cancel
${ selectedItem.replyTextCharacter }/${ selectedItem.replyMaxCharacter }
Submit
Cancel
Confirm
Are you sure to Delete?
Yes
No




