Your browser does not fully support modern features. Please upgrade for a smoother experience.

Submitted Successfully!
Thank you for your contribution! You can also upload a video entry or images related to this topic.
For video creation, please contact our Academic Video Service.
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Bahiru Tsegaye | + 2334 word(s) | 2334 | 2021-06-03 10:38:59 | | | |
| 2 | Catherine Yang | -16 word(s) | 2318 | 2021-06-17 03:24:07 | | |
Video Upload Options
We provide professional Academic Video Service to translate complex research into visually appealing presentations. Would you like to try it?
Cite
If you have any further questions, please contact Encyclopedia Editorial Office.
Tsegaye, B. Food Waste Biorefinery. Encyclopedia. Available online: https://encyclopedia.pub/entry/10927 (accessed on 07 February 2026).
Tsegaye B. Food Waste Biorefinery. Encyclopedia. Available at: https://encyclopedia.pub/entry/10927. Accessed February 07, 2026.
Tsegaye, Bahiru. "Food Waste Biorefinery" Encyclopedia, https://encyclopedia.pub/entry/10927 (accessed February 07, 2026).
Tsegaye, B. (2021, June 16). Food Waste Biorefinery. In Encyclopedia. https://encyclopedia.pub/entry/10927
Tsegaye, Bahiru. "Food Waste Biorefinery." Encyclopedia. Web. 16 June, 2021.
Copy Citation
Food waste biorefineries for the production of biofuels, platform chemicals, and other bio-based materials can significantly reduce a huge environmental burden and provides opportunities for a sustainable way of chemical and material production.
biorefinery
platform chemicals
biofuels
biopolymer
Bio-based materials
Sustainable Production
Sustainable development goal
Food waste
circular bioeconomy
green hydrogen
1. Introduction
The environmental problem is one of the most difficult issues challenging the world today. The fast-growing world population accelerates the need for food and other basic materials, which is accompanied by the bulk generation of waste biomass. This directly contributes to the increased cost of waste disposal and causes significant environmental problems. The growing population is directly proportional to the increased demand of food and subsequently the larger quantity of food production that is accompanied by bulk generation of food wastes. According to the United Nations Food and Agricultural Organization (FAO), 1/3 of the total food produced was lost in the supply chain and harvesting which contributed to the estimated value of USD 1 trillion annual loss [1]. The drink industries are leading by generating around 26% of the total food waste, followed by the dairy industry which contributes 21%, fruit and vegetable industry 14.8%, and cereal industry 12.9% [2]. Other than the economic impact, food waste is a potent greenhouse gas emitter (mainly methane) contributing to environmental pollution. More recently, food waste is directly connected to water loss, air pollution, water pollution, biodiversity loss, soil degradation, and climate change. The loss of food as waste which was intended to be for human consumption is likely to be linked to nutritional loss in diet.
Food waste includes spoiled foods, crops left in the field, fruit and vegetable waste, leftovers on the plate from hotels, homes, and restaurants, and any other food lost at any stages of the supply chains. It is impossible to completely avoid food waste, however it is possible to reduce the amount of wasted food. Therefore, crafting ways or methods of valorizing food waste are crucial for developing sustainable bioeconomy and for achieving United Nations (UN) sustainable development goal of 2030 [3]. Due to their homogeneity, food waste has high potential for the production of biofuels, platform chemicals and bio-based materials by applying the concept of biorefinery [4][5]. The valorization of food waste under the biorefinery framework has recently gained momentum for the implementation and achievement of the sustainable development goals policies set by the European Unions (EU), such as the bioeconomy strategy and the circular economy goals of the EU [6][7][8]. According to the bioeconomy council, “The bioeconomy is the knowledge-based production and use of biological resources to provide products, processes and services in all economic sectors within the frame of a sustainable economic system”. The European bioeconomy strategy focused on the needs of the sustainability and circularity of processes and products [6]. The European commission defined the circular economy as the elimination/minimization of waste generations during the processing and production of products, materials and resources by maintaining the value of the product as long as possible [7]. The concept of the circular bioeconomy is described as the production of energy, food, platform chemicals, and other bio-based materials and compounds from biomass in a sustainable and integrated/cascaded manner (biorefinery) while generating zero waste [6][7].
2. Food Waste Biorefinery
Food waste biorefinery is a process by which a broader range of food wastes are converted into biofuels, platform chemicals and bio-based materials. For food waste valorization, it is essential to know the compositions, the interaction of its components, and the desired final products for choosing an efficient biorefinery process [9]. In general, food waste biorefinery processes are categorized into three major groups: (i) biological pathway: a process by which food wastes are converted into value-added product via enzymes or microorganisms; (ii) thermochemical process: a process by which food wastes are treated at elevated temperature using chemicals as a solvent. This includes liquefaction, pyrolysis, and gasification; (iii) chemical process: a process by which chemicals are used as a solvent and as a catalyst in food waste valorizations. The combination of two or more of the above processes in an integrated manner has been attracting the attention of many researchers due to higher conversion efficiencies.
2.1. Bioconversion Processes
2.1.1. Anaerobic Digestion
Anaerobic digestion is a biological process by which organic matters are metabolized and transformed by complex reactions into biogas in the absence of oxygen [10][11]. Anaerobic digestion is commonly found in nature such as in animal digestive system, in swaps and wetlands. Anaerobic digestion is most-commonly practiced throughout the world in many ways, such as the digestion of primary and secondary sewage sludge, upflow of anaerobic sludge blanket reactors, and activated sludge plants [12][13][14]. The process consists of four steps: hydrolysis, acidogenesis, acetogenesis, and methanogenesis, which may occur sequentially or simultaneously in a single stage. Products such as methane (CH4), volatile fatty acids (VFAs), such as propionic acid, butyric acid, acetic acid, iso-butyric acid, valeric acid, iso-valeric acid, and hydrogen (H2) are produced from food waste via anaerobic digestion or anaerobic fermentation.
The anaerobic digestion can be either performed in single stage or two stage operations. In the single stage configuration, all reactions are carried out in a single reactor that helps toward low operational costs and low reactor complexity. However, the formation of intermediate products accelerates inhibition of the subsequent processes. Hence, the lower conversion efficiency and lower product yield are obtained in such reactor configurations. Generally, in single stage reactor configurations, process instability, reactor acidifications, and the combined production of hydrogen and methane are common problems [15][16]. The two-stage process in which the acidogenic and methanogenic processes are physically separated appears to be effective, overcoming problems associated with single stage digestion [17]. The anaerobic digestion of mixtures of food waste, poultry litter, and sewage sludge enhanced the biogas yield to 640 L/kg VS when mixed in the ratio of 2:1:1, sewage sludge: food waste: poultry litter [18]. The anaerobic digestion of food waste for methane production at mesophilic temperature (34 °C) generated 276.5 mL CH4/g VS while 307.5 mL CH4/g VS was obtained at the thermophilic temperature of 55 °C [19]. A study carried out by Patinvoh et al. [20] observed that the yield of VFAs was enhanced by controlling the pH of acidogenesis process (at pH 6) during the anaerobic digestion of food waste [20]. The highest yield of VFA (0.8 g VFA/g VS) was achieved at an inoculum to substrate ratio of 1:3 [20]. The integration of dark fermentation (acidogenesis) and methanogenesis of food waste enhanced the biohythane (H2 + CH4) production by 1.22 times [21]. The yield of methane from the anaerobic digestion of one ton of food waste can be as high as 90.6 m3 [22]. The reaction configurations of anaerobic digestion are highly influenced and controlled by process parameters (pH, acidity, temperature, substrate composition, C/N ratio, reaction time, and inoculum) and the desired final product. Therefore, optimizing the process parameters enhances the yield of the desired final product.
2.1.2. Dark Fermentation
Dark fermentation is a microbial conversion process in which hydrogen is produced by anaerobic bacteria from organic matters via glycolysis pathway. It is performed in the absence of light by a diverse group of bacteria. The cost effectiveness and the possibilities of utilizing wide ranges of substrates in the dark fermentation for biohydrogen production have been studied by numerous authors [23][24]. However, problems associated with low hydrogen yield and high cost of production is a challenge for scale up and commercialization of dark fermentation technology [24]. Theoretically, 12 moles of biohydrogen are expected from one mole of glucose, however maximum yield of four moles of biohydrogen were obtained when acetic acid was the end product, while two moles were produced when butyric acid was the end product. With VFAs formations, 2–3 moles of biohydrogen are obtained from one mole of glucose [25]. Dark fermentation of food waste collected from cafeterias yielded 1.77 moles of H2/mole of hexose [26]. However, sequential dark fermentation and photofermentation increased the biohydrogen production by 2.5-folds producing 5.4-moles of H2/mole of hexose [26]. Dark fermentation using food waste at the mesophilic temperature of 34 °C led to a biohydrogen yield of 53.5 mL H2/g VS, while 37.6 mL H2/g VS were obtained at the thermophilic temperature of 55 °C [19]. Nguyen et al. [27] studied the single stage dark fermentation of food waste mixed with condensed molasses to produce biohythane (H2 + CH4). Biogas comprising 10–60% H2, and 5–20% CH4 was obtained depending on the ratio of food to microorganism [27]. The co-existence of a wide range of microorganisms can significantly reduce the yields of biohydrogen by either utilizing the produced biohydrogen or metabolizing the substrate into other products [28]. The operating conditions highly influence the specific microbial communities and the final product. Acetate and butyrate pathways are linked to higher biohydrogen yields while alcohol and lactate production pathways are linked with lower biohydrogen yields [29][30]. Optimizing the fermentation conditions significantly enhances the biohydrogen yield but is far from reaching the near theoretical yield. Adjustment of the reactor configurations for utilization of the intermediate products during co-culturing or sequential photofermentation can greatly enhance the biohydrogen yield. Metabolic engineering has great potential to alter the current barriers of dark fermentation, and the application of metabolic engineering principles to the selected strains of microorganisms has a promising future, which could revolutionize the whole biorefinery process.
2.1.3. Electro-Fermentation
Electro-fermentation is a new type of hybrid technology that combines the old fermentation principles and electromicrobiology for the improvement of product yields. It uses polarized electrodes to redirect the transfer of small number of electrons into and/or from the medium. The main source of electrons during the electro-fermentation process is the organic material in the medium because the number of electrons exchanged at the polarized electrode is low compared to the microbial electrosynthesis [31][32][33]. The interactions of the microorganisms with the electrode during electro-fermentation are either through DIET (direct interspecies electron transfer mechanisms) or MIET (indirect interspecies electron transfer mechanisms) [34]. The electron transfers are achieved by mediators/shuttles produced by cells such as flavins, formate, phenazines, and H2 in the case of MIET while electrically conductive pilus or proteins such as cytochromes are used in the case of DIET [34][35][36]. Shewanella oneidensis and Geobacter sufurreducens are the two most commonly studied electroactive bacteria and are considered as a model for DIET. This impressive capability observed in some bacteria can be exploited for biohydrogen production. Recently, electro-fermentation has been employed on food waste valorization and promising results were obtained [37][38][39]. About 26.3% improvement in methane production was achieved by limiting the amount of volatile fatty acids to 129 mg/L from the electro-fermentation of food waste [38]. Hydrogen recovery was also improved by the sequential process of electro-fermentation of food waste from the effluents of dark fermentation [39]. Therefore, further studies are required to fully exploit the microbial potential for biohydrogen production as well as for other biomaterials from different food wastes.
2.1.4. Photofermentation
Photofermentation is a fermentation process in which light is used as an additional source of energy. The purple non-sulfur bacteria (PNSB) are the most common photosynthetic bacteria. Electrons are driven out from the organic food waste by nitrogenase enzyme of the photosynthetic bacteria to produce carbon dioxide and hydrogen [40]. Food wastes such as glycerol that contain simpler organic compounds and short chains fatty acids are ideal substrates for photofermentation [40][41]. Photofermentation as a green technology has great potential and capability for production of biohydrogen from food waste as evident from wastewater treatments emerging from industries such as dairies [42], distilleries [43], brewery [44], and sugar refinery [45]. The production cost of 1 kg of hydrogen by photofermentation was estimated to be about EUR 2.83, while electrolysis-based technology costs from EUR 4–24 [46]. The presence of inhibitory compounds in the waste, lower light penetrations due to the turbidity of the waste, and the rate of cell wash out exceeding the specific growth rates are some of the major challenges hindering the production of biohydrogen by photofermentation [47][48]. The immobilization of microbial cells is an effective approach to overcome the over washing, while other drawbacks need to be resolved [49]. To use the full power of photofermentation, the drawbacks have to be resolved. Therefore, intensive research is required to develop feasible and sustainable photofermentation technology to utilize food waste for high-value product production.
2.2. Integrated Approach
Integrated approaches are considered in order to improve the economics of food waste treatments, enhancing product yields, and reducing the current high production costs. Two stage dark fermentation integrated with microalgal cultivation (MC) was applied to improve overall energy and resource recovery [50]. Enriching starchy waste-water with poultry manure to increase the nitrogen supplement in dark fermentation enhanced the biohydrogen yield from 4.11 mol/kg COD (chemical oxygen demand) to 5.03 mol/kg COD, while the remaining spent was utilized for biodiesel production by Chlamydomonas reinhardtii [50]. On the other hand, thermal pretreatments (at 121 °C for 15 min) of starch wastewater enriched with groundnut de-oiled cake showed an improved biohydrogen production of 3.24 L/L and biohydrogen yield of 12.05 mol H2 kg−1 COD [51]. The addition of nano-metal oxides in rice mill wastewater during dark fermentation by Clostridium beijerinckii DSM 791 showed improved biohydrogen production, while the addition of NiO and CoO nanoparticles enhanced biohydrogen yields by 109% and 90% respectively [52]. The integration of dark fermentation and photofermentation significantly improves the biohydrogen yield. In this hybrid system, biohydrogen and organic acids are produced during dark fermentation and enhanced biohydrogen was produced by dark fermentation using purple nonsulfur bacteria [53][54]. The mode of operation of this hybrid system is either in a single stage (combined system) or sequential (two stage), and was found to be very efficient for biohydrogen production. The two-stage system (sequential) is more promising, as the metabolic products of dark fermentation sometimes require treatment and different optimal conditions [55]. The overall reaction of integrated dark fermentation and photofermentation in a sequential manner is:
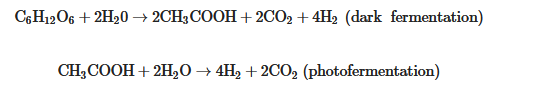
These show the potential of the integrated food waste biorefinery process for opening up the way for the circular economy. More investigations and research studies on how to improve the efficiencies of conversion and product yield in the pilot scale and commercial scale are key for the transition to bioeconomy. The overall complexity of the food industries and the relationships with the circular bioeconomy and sustainability are described in Figure 1.
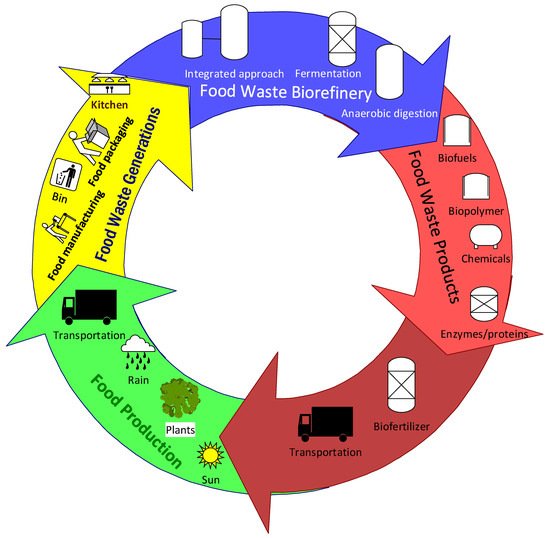
Figure 1. The prospect of circular bioeconomy in food industries.
References
- FAO. Global Food Losses and Food Waste—Extent, Causes and Prevention; FAO: Rome, Italy, 2011; ISBN 9781788975391.
- Baiano, A. Recovery of biomolecules from food wastes—A review. Molecules 2014, 19, 14821–14842.
- UN. Transforming Our World: The 2030 Agenda for Sustainable Development Preamble; United Nations: New York, NY, USA, 2015; ISBN 9781138029415.
- Ong, K.L.; Kaur, G.; Pensupa, N.; Uisan, K.; Lin, C.S.K. Trends in food waste valorization for the production of chemicals, materials and fuels: Case study South and Southeast Asia. Bioresour. Technol. 2018, 248, 100–112.
- Matharu, A.S.; de Melo, E.M.; Houghton, J.A. Opportunity for high value-added chemicals from food supply chain wastes. Bioresour. Technol. 2016, 215, 123–130.
- EU. A Sustainable Bioeconomy for Europe: Strengthening the Connection between Economy, Society and the Environment; European Commission: Brussels, Belgium, 2018.
- EU. Closing the Loop—An EU Action Plan for the Circular Economy; Europe Union: Brussels, Belgium, 2015.
- Cristóbal, J.; Caldeira, C.; Corrado, S.; Sala, S. Techno-economic and profitability analysis of food waste biorefineries at European level. Bioresour. Technol. 2018, 259, 244–252.
- Carmona-Cabello, M.; Garcia, I.L.; Leiva-Candia, D.; Dorado, M.P. Valorization of food waste based on its composition through the concept of biorefinery. Curr. Opin. Green Sustain. Chem. 2018, 14, 67–79.
- Xu, Y.; Lu, Y.; Zheng, L.; Wang, Z.; Dai, X. Perspective on enhancing the anaerobic digestion of waste activated sludge. J. Hazard. Mater. 2020, 389, 121847.
- Kumar, A.; Samadder, S.R. Performance evaluation of anaerobic digestion technology for energy recovery from organic fraction of municipal solid waste: A review. Energy 2020, 197, 117253.
- Feng, L.; Chen, Y.; Zheng, X. Enhancement of waste activated sludge protein conversion and volatile fatty acids accumulation during waste activated sludge anaerobic fermentation by carbohydrate substrate addition: The effect of pH. Environ. Sci. Technol. 2009, 43, 4373–4380.
- Chernicharo, C.A.L.; van Lier, J.B.; Noyola, A.; Bressani Ribeiro, T. Anaerobic sewage treatment: State of the art, constraints and challenges. Rev. Environ. Sci. Biotechnol. 2015, 14, 649–679.
- Carneiro, R.B.; Gonzalez-Gil, L.; Londoño, Y.A.; Zaiat, M.; Carballa, M.; Lema, J.M. Acidogenesis is a key step in the anaerobic biotransformation of organic micropollutants. J. Hazard. Mater. 2020, 389, 121888.
- Mari, A.G.; Andreani, C.L.; Tonello, T.U.; Leite, L.C.C.; Fernandes, J.R.; Lopes, D.D.; Rodrigues, J.A.D.; Gomes, S.D. Biohydrogen and biomethane production from cassava wastewater in a two-stage anaerobic sequencing batch biofilm reactor. Int. J. Hydrogen Energy 2020, 45, 5165–5174.
- Feng, K.; Wang, Q.; Li, H.; Zhang, Y.; Deng, Z.; Liu, J.; Du, X. Effect of fermentation type regulation using alkaline addition on two-phase anaerobic digestion of food waste at different organic load rates. Renew. Energy 2020, 154, 385–393.
- Srisowmeya, G.; Chakravarthy, M.; Nandhini Devi, G. Critical considerations in two-stage anaerobic digestion of food waste—A review. Renew. Sustain. Energy Rev. 2020, 119, 109587.
- Lohani, S.P.; Shakya, S.; Gurung, P.; Dhungana, B.; Paudel, D.; Mainali, B. Anaerobic co-digestion of food waste, poultry litter and sewage sludge: Seasonal performance under ambient condition and model evaluation. Energy Sources Part A Recover. Util. Environ. Eff. 2021.
- Ghimire, A.; Luongo, V.; Frunzo, L.; Lens, P.N.; Pirozzi, F.; Esposito, G. Biohythane production from food waste in a two-stage process: Assessing the energy recovery potential. Environ. Technol. 2021.
- Patinvoh, R.J.; Millati, R.; Sárvári-horváth, I.; Taherzadeh, M.J. Factors influencing volatile fatty acids production from food wastes via anaerobic digestion production. Bioengineered 2020, 11, 39–52.
- Sarkar, O.; Santhosh, J.; Dhar, A.; Mohan, S.V. Green hythane production from food waste: Integration of dark-fermentation and methanogenic process towards biogas up- gradation. Int. J. Hydrogen Energy 2021.
- Kuo, J.; Dow, J. Biogas production from anaerobic digestion of food waste and relevant air quality implications. J. Air Waste Manag. Assoc. 2017, 67, 1000–1011.
- Pandey, A.; Srivastava, S.; Rai, P.; Duke, M. Cheese whey to biohydrogen and useful organic acids: A non-pathogenic microbial treatment by L. acidophilus. Sci. Rep. 2019, 9, 1–9.
- Ghimire, A.; Frunzo, L.; Pirozzi, F.; Trably, E.; Escudie, R.; Lens, P.N.L.; Esposito, G. A review on dark fermentative biohydrogen production from organic biomass: Process parameters and use of by-products. Appl. Energy 2015, 144, 73–95.
- Sinha, P.; Pandey, A. An evaluative report and challenges for fermentative biohydrogen production. Int. J. Hydrogen Energy 2011, 36, 7460–7478.
- Zong, W.; Yu, R.; Zhang, P.; Fan, M.; Zhou, Z. Efficient hydrogen gas production from cassava and food waste by a two-step process of dark fermentation and. Biomass Bioenergy 2009, 33, 1458–1463.
- Nguyen, M.T.; Hung, P.; Vo, T. Effect of food to microorganisms (F/M) ratio on biohythane production via single-stage dark fermentation. Int. J. Hydrogen Energy 2020.
- Cabrol, L.; Marone, A.; Tapia-Venegas, E.; Steyer, J.P.; Ruiz-Filippi, G.; Trably, E. Microbial ecology of fermentative hydrogen producing bioprocesses: Useful insights for driving the ecosystem function. FEMS Microbiol. Rev. 2017, 41, 158–181.
- Saady, N.M.C. Homoacetogenesis during hydrogen production by mixed cultures dark fermentation: Unresolved challenge. Int. J. Hydrogen Energy 2013, 38, 13172–13191.
- Liu, C.G.; Xue, C.; Lin, Y.H.; Bai, F.W. Redox potential control and applications in microaerobic and anaerobic fermentations. Biotechnol. Adv. 2013, 31, 257–265.
- Toledo-Alarcón, J.; Fuentes, L.; Etchebehere, C.; Bernet, N.; Trably, E. Glucose electro-fermentation with mixed cultures: A key role of the Clostridiaceae family. Int. J. Hydrogen Energy 2021, 46, 1694–1704.
- Toledo-Alarcón, J.; Moscoviz, R.; Trably, E.; Bernet, N. Glucose electro-fermentation as main driver for efficient H2-producing bacteria selection in mixed cultures. Int. J. Hydrogen Energy 2019, 2230–2238.
- Moscoviz, R.; Toledo-Alarcón, J.; Trably, E.; Bernet, N. Electro-Fermentation: How to Drive Fermentation Using Electrochemical Systems. Trends Biotechnol. 2016, 34, 856–865.
- Creasey, R.C.G.; Mostert, A.B.; Nguyen, T.A.H.; Virdis, B.; Freguia, S.; Laycock, B. Microbial nanowires—Electron transport and the role of synthetic analogues. Acta Biomater. 2018, 69, 1–30.
- Thrash, J.C.; Coates, J.D. Review: Direct and indirect electrical stimulation of microbial metabolism. Environ. Sci. Technol. 2008, 42, 3921–3931.
- Hirose, A.; Kouzuma, A.; Watanabe, K. Towards development of electrogenetics using electrochemically active bacteria. Biotechnol. Adv. 2019, 37, 107351.
- Zhang, Y.; Li, J.; Meng, J.; Wang, X. A cathodic electro-fermentation system for enhancing butyric acid production from rice straw with a mixed culture. Sci. Total Environ. 2021, 767, 145011.
- Liu, S.; Deng, Z.; Li, H.; Feng, K. Contribution of electrodes and electric current to process stability and methane production during the electro-fermentation of food waste. Bioresour. Technol. 2019, 288, 121536.
- Jia, X.; Li, M.; Wang, Y.; Wu, Y.; Zhu, L.; Wang, X.; Zhao, Y. Enhancement of hydrogen production and energy recovery through electro-fermentation from the dark fermentation effluent of food waste. Environ. Sci. Ecotechnology 2020, 1, 100006.
- Hanipa, M.A.F.; Abdul, P.M.; Jahim, J.M.; Takriff, M.S.; Reungsang, A.; Wu, S.Y. Biotechnological approach to generate green biohydrogen through the utilization of succinate-rich fermentation wastewater. Int. J. Hydrogen Energy 2020, 45, 22246–22259.
- Basak, N.; Jana, A.K.; Das, D.; Saikia, D. Photofermentative molecular biohydrogen production by purple-non-sulfur (PNS) bacteria in various modes: The present progress and future perspective. Int. J. Hydrogen Energy 2014, 39, 6853–6871.
- Seifert, K.; Waligorska, M.; Laniecki, M. Hydrogen generation in photobiological process from dairy wastewater. Int. J. Hydrogen Energy 2010, 35, 9624–9629.
- Laurinavichene, T.; Tekucheva, D.; Laurinavichius, K.; Tsygankov, A. Utilization of distillery wastewater for hydrogen production in one-stage and two-stage processes involving photofermentation. Enzyme Microb. Technol. 2018, 110, 1–7.
- Lu, H.; Zhang, G.; He, S.; Peng, C.; Ren, Z. Production of photosynthetic bacteria using organic wastewater in photobioreactors in lieu of a culture medium in fermenters: From lab to pilot scale. J. Clean. Prod. 2020, 259, 120871.
- Assawamongkholsiri, T.; Reungsang, A.; Plangkang, P.; Sittijunda, S. Repeated batch fermentation for photo-hydrogen and lipid production from wastewater of a sugar manufacturing plant. Int. J. Hydrogen Energy 2018, 43, 3605–3617.
- Sinigaglia, T.; Lewiski, F.; Santos Martins, M.E.; Mairesse Siluk, J.C. Production, storage, fuel stations of hydrogen and its utilization in automotive applications-a review. Int. J. Hydrogen Energy 2017, 42, 24597–24611.
- Hay, J.X.W.; Wu, T.Y.; Juan, J.C.; Jahim, J.M. Effect of adding brewery wastewater to pulp and paper mill effluent to enhance the photofermentation process: Wastewater characteristics, biohydrogen production, overall performance, and kinetic modeling. Environ. Sci. Pollut. Res. 2017, 24, 10354–10363.
- Ghosh, S.; Dairkee, U.K.; Chowdhury, R.; Bhattacharya, P. Hydrogen from food processing wastes via photofermentation using Purple Non-sulfur Bacteria (PNSB)—A review. Energy Convers. Manag. 2017, 141, 299–314.
- Du Toit, J.P.; Pott, R.W.M. Transparent polyvinyl-alcohol cryogel as immobilisation matrix for continuous biohydrogen production by phototrophic bacteria. Biotechnol. Biofuels 2020, 13, 1–16.
- Radhakrishnan, R.; Banerjee, S.; Banerjee, S.; Singh, V.; Das, D. Sustainable approach for the treatment of poultry manure and starchy wastewater by integrating dark fermentation and microalgal cultivation. J. Mater. Cycles Waste Manag. 2021.
- Mahata, C.; Dhar, S.; Ray, S.; Das, D. Effect of thermal pretreated organic wastes on the dark fermentative hydrogen production using mixed microbial consortia. Fuel 2021, 284, 119062.
- Rambabu, K.; Bharath, G.; Thanigaivelan, A.; Das, D.B.; Show, P.L.; Banat, F. Augmented biohydrogen production from rice mill wastewater through nano-metal oxides assisted dark fermentation. Bioresour. Technol. 2021, 319, 124243.
- Ventura, J.R.S.; Rojas, S.M.; Ventura, R.L.G.; Nayve, F.R.P.; Lantican, N.B. Potential for biohydrogen production from organic wastes with focus on sequential dark- and photofermentation: The Philippine setting. Biomass Convers. Biorefinery 2021.
- Mishra, P.; Krishnan, S.; Rana, S.; Singh, L.; Sakinah, M.; Ab Wahid, Z. Outlook of fermentative hydrogen production techniques: An overview of dark, photo and integrated dark-photo fermentative approach to biomass. Energy Strateg. Rev. 2019, 24, 27–37.
- Rai, P.K.; Singh, S.P. Integrated dark- and photo-fermentation: Recent advances and provisions for improvement. Int. J. Hydrogen Energy 2016, 41, 19957–19971.
More
Information
Subjects:
Food Science & Technology
Contributor
MDPI registered users' name will be linked to their SciProfiles pages. To register with us, please refer to https://encyclopedia.pub/register
:
View Times:
1.5K
Revisions:
2 times
(View History)
Update Date:
17 Jun 2021
Notice
You are not a member of the advisory board for this topic. If you want to update advisory board member profile, please contact office@encyclopedia.pub.
OK
Confirm
Only members of the Encyclopedia advisory board for this topic are allowed to note entries. Would you like to become an advisory board member of the Encyclopedia?
Yes
No
${ textCharacter }/${ maxCharacter }
Submit
Cancel
Back
Comments
${ item }
|
More
No more~
There is no comment~
${ textCharacter }/${ maxCharacter }
Submit
Cancel
${ selectedItem.replyTextCharacter }/${ selectedItem.replyMaxCharacter }
Submit
Cancel
Confirm
Are you sure to Delete?
Yes
No




