Infrared spectroscopy has long been used to characterize chemical compounds, but the applicability of this technique to the analysis of biological materials containing highly complex chemical components is arguable. However, recent advances in the development of infrared spectroscopy have significantly enhanced the capacity of this technique in analyzing various types of biological specimens. Consequently, there is an increased number of studies investigating the application of infrared spectroscopy in screening and diagnosis of various diseases. The lack of highly sensitive and specific methods for early detection of cancer has warranted the search for novel approaches. Being more simple, rapid, accurate, inexpensive, non-destructive and suitable for automation compared to existing screening, diagnosis, management and monitoring methods, Fourier transform infrared spectroscopy can potentially improve clinical decision-making and patient outcomes by detecting biochemical changes in cancer patients at the molecular level. Besides the commonly analyzed blood and tissue samples, extracellular vesicle-based method has been gaining popularity as a non-invasive approach. Therefore, infrared spectroscopic analysis of extracellular vesicles could be a useful technique in the future for biomedical applications.
1. Introduction
Cancer is a major cause of death worldwide, accounting for a staggering estimated 9.6 million deaths in 2018
[1]. Globally, approximately one in three individuals will be diagnosed with cancer in his or her lifetime and one in six deaths is caused by cancer
[1]. The poor survival rates reflect the fact that most patients are diagnosed at a stage which is not responsive to current treatments. Early cancer detection is crucial as the condition of a patient may be irreversible once the clinical symptoms appear. In fact, early detection of the disease and identification of at-risk individuals may delay or prevent further progression with suitable treatments and would greatly increase survival rate of patients. However, current screening and diagnostic methods including imaging techniques, normally detect cancer in late stage when tumor mass is visible and existing screening tests lack the necessary sensitivity and specificity at early stage of the disease
[2].
To date, the gold standard for most cancer diagnosis is still the microscopic evaluation of stained tissue samples by pathologists, which is performed when cancerous or pre-cancerous lesions are observable and already contain significant genetic changes. Moreover, the use of the histopathological diagnosis is invasive, time consuming and has limited sensitivity as it depends heavily on the subjective judgement of pathologists which leads to intra- and inter-observer variations. Therefore, misdiagnosis with high false negative and false positive rates is common in tissue assessment
[3]. In fact, approximately 10% of pathologic evaluation could not result in a firm diagnosis because either certain tumors are histologically similar or the tissue of origin could not be identified from the poorly differentiated cells
[4]. The method also involves the complex process of histochemical staining techniques for the tissue samples, whereby the most commonly used hematoxylin and eosin (H&E) dyes are non-specific for cancer
[5].
Biomarkers, which are defined as disease-related molecular changes in body fluids and tissues
[6], are essential in facilitating screening and diagnosis to allow clinical interventions to begin as soon as possible. Conventional clinical analysis of blood samples for cancer diagnosis examines individual parameters which include tumor markers such as carcinoembryonic antigen (CEA), cancer antigen 15-3 (CA 15-3), prostate-specific antigen (PSA) and tissue polypeptide antigen (TPA). Nevertheless, these markers have low sensitivity and/or specificity
[7][8][9]. Thus, the lack of highly specific and sensitive biomarkers for cancer as well as the limited number of non-invasive and cost-effective tests demand the discovery of novel biomarkers and diagnostic methods. Thus, combinations of biomarkers have been investigated to improve the current situation
[10][11] and the use of multi-molecular biochemical analysis techniques such as Fourier transform infrared (FTIR) spectroscopy could support this purpose. As biochemical changes are preceded or accompanied with morphological alteration and symptomatic appearance correlated with disease progression or therapeutic treatment, the use of the vibrational spectroscopic technique can reveal these differences at molecular level and could serve as a screening and diagnostic tool
[12]. In addition, instead of evaluating morphological differences as in current histopathology methods, the application of FTIR spectroscopy which analyses tissue samples at the molecular level before morphological changes arise without the need for staining has been investigated. This permits objective assessment of the samples which allows early detection and increases accuracy as well as minimizes discrepancies in the interpretation of pathologists.
Traditionally, FTIR spectroscopy has been used by chemists for the characterization of molecular structures. Nonetheless, the potential of the technique to analyze biological specimens as a cancer diagnostic tool has been recognized for decades
[13]. The relatively simple and reproducible technique is reagent-free, non-destructive to samples and only require nanograms to micrograms of them with minimal preparation. The sensitivity of FTIR spectroscopy to chemical changes during the transition from normal to a pathological state or during treatment can lead to the identification of novel biomarkers associated with a disease
[14]. Hence, FTIR spectroscopy is a robust tool with great potential for clinical application which extends beyond screening, diagnosis and prognosis of cancer to continuous monitoring of treatment response and disease progression or regression in personalized medicine.
Various types of biological materials including blood
[15][16][17][18][19], tissues
[4][20][21][22][23][24], extracellular vesicles (EVs)
[25][26][27], urine
[28], bladder wash
[29], bile
[30] and sputum
[31][32] specimens have been studied using FTIR spectroscopy to develop better alternatives for cancer diagnosis and management. Blood and tissue samples are widely used in current clinical diagnostics for various diseases compared to other types of specimen. Nevertheless, the immense biological variability found in these complex biological specimens may mask specific spectral changes and hinder the identification of biomarkers. EVs have lately gained research interests due to their association with cancer. These vesicles have a diameter which ranges from 30–1000 nm and contain various biomolecules such as proteins, lipids and nucleic acids
[33]. They are secreted by all cell types in the body and play a major role in intercellular communication. Intriguingly, EVs may reflect the condition of their originating cells and provide information of disease progression
[34]. Therefore, EVs can be isolated from biofluids which are obtained non-invasively, such as urine, for FTIR spectroscopic analysis. This could remove the abundant uninformative and uncorrelated data to effectively identify biomarkers.
2. Wavenumber Range and Computational Models in Fourier Transform Infrared Spectroscopic Analysis of Biological Specimens
FTIR spectroscopy detects biochemical compositions including nucleic acids, proteins, lipids and carbohydrates within biological samples by precisely identifying molecular conformations, bonding types, functional groups and intermolecular interactions of which the specimen is composed. As each molecule has a unique spectrum depending on the wavelength and quantity of infrared radiation being absorbed, IR spectroscopy produces a signature spectral fingerprint of absorbance peaks for multiplex parameters of genome, lipidome, proteome and metabolome in the examined sample. Essentially, the biochemical fingerprint changes are unique to the molecular alterations in specific diseases, providing valuable diagnostic information for each patient’s health status. As biological materials absorb energy in the mid-IR region (4000–400 cm
−1 of the electromagnetic spectrum, the spectral regions typically measured for examining these specimens are the fingerprint region (1450–600 cm
−1) as well as the amide I and II region (1700–1500 cm
−1). Higher-wavenumber region (3500–2550 cm
−1) is correlated to stretching vibrations including C–H, O–H, N–H and S–H, while lower-wavenumber regions are usually associated with bending and carbon skeletal fingerprint vibrations
[35]. shows the assignment of typical absorption bands identified in biological IR spectra
[36][37][38].
Table 1. Assignment of typical absorption bands observed in biological IR spectra.
| Wavenumber (cm−1) |
Assignment |
| 3080–2800 |
Anti-symmetric and symmetric C–H stretches from proteins and lipids |
| 1745–1725 |
Ester carbonyl of lipids |
| 1700–1500 |
Amide I and II groups in peptide linkages of proteins |
| 1270–1080 |
Anti-symmetric and symmetric C−O and P−O areas in DNA, RNA and phospholipids |
| 1200–900 |
Carbohydrate vibrations of glucose, fructose and glycogen |
By quantitatively measuring the vibrational modes
[39] and analyze spectroscopic data to identify disease patterns with artificial intelligent systems, FTIR spectroscopy allows the advancement of next-generation clinical systems that could revolutionize disease diagnostics. Due to the molecular complexity of biological specimens, common techniques such as chemometrics which combine statistical and mathematical procedures are utilized to generate chemo-physical evidence from spectral data
[40]. One of the chemometric techniques is principal component analysis (PCA), which is the most basic feature extraction unsupervised method based upon the analysis of feature variance within the full spectrum
[36]. This technique has been coupled with FTIR spectroscopy in numerous studies for various applications in cancer, including cancer identification
[41] and monitoring of chemotherapy efficacy
[42]. Meanwhile, clustering unsupervised techniques including discriminant analysis (DA), hierarchical cluster analysis (HCA), support vector machines (SVM), artificial neural networks (ANN) and k-nearest neighbours (KNN) are applied to identify biological subtypes within a specimen. On the other hand, partial least squares (PLS) is the most extensively used supervised multivariate data analysis method which quantifies and estimates components in a sample
[36]. Additionally, physics-based computational models have also been applied in FTIR spectroscopic analysis for cancer classification
[43] and treatment monitoring to determine therapeutic efficacy
[44][45].
3. Sensitivity, Specificity and Accuracy in Cancer Detection
The power of a test to differentiate patients from healthy individuals defines its accuracy and diagnostic value
[46]. The characteristics which reflects the abilities of a test include accuracy, sensitivity and specificity. The accuracy of a test is defined as the ability to correctly distinguish the patient and healthy cases. The ideal diagnostic test would identify the cases with 100% accuracy. However, the accuracy of a test varies in different situations and for different diseases. Meanwhile, the sensitivity and specificity of a test are its ability to correctly determine the patient and healthy cases, respectively
[47]. Even though these measures are usually regarded as fixed properties of a diagnostic test, they are subjected to multiple variations such as the severity of the disease under investigation and the population case mix
[48]. In cancer diagnostic tests, current biomarkers used to detect the disease often have low sensitivity and/or specificity. For instance, the prostate-specific antigen (PSA) test has been used to detect prostate cancer. Although the test has a high specificity of approximately 87–95%, it has a much lower sensitivity which ranges from 33–59%
[7].
FTIR spectroscopy has been shown to be a prospective novel diagnostic method for many different types of cancer by being able to distinguish cancer samples from normal ones at high sensitivity, specificity and accuracy. Sheng et al. have demonstrated the use of this technique in the diagnosis of leukemia
[17] and gastric cancer
[18] by analyzing serum samples. RNA/DNA and peak height ratios demonstrated high sensitivity and specificity of approximately 80–100% for the diagnosis of both cancers. Likewise, up to 98% of sensitivity and 100% of specificity have been reported by Backhaus et al.
[15] in the analysis of serum samples for breast cancer diagnosis using FTIR spectroscopy as well as cluster analysis (CA) and artificial neural networks (ANN). Remarkably, these findings showed significant improvement over conventional clinical analysis of the tumor markers cancer antigen 15-3 (CA 15-3), carcinoembryonic antigen (CEA) and tissue polypeptide antigen (TPA) for breast cancer monitoring, which have only at most 60–70% of sensitivity and specificity
[8][9][49]. Notably, the study also revealed the ability to distinguish breast cancer from other diseases such as Alzheimer’s disease, hepatitis C, coronary heart diseases as well as other types of cancer. On the other hand, Khanmohammadi et al.
[4] applied attenuated total reflection (ATR)-FTIR microspectroscopy and chemometric techniques, such as CA, analysis of variance (ANOVA) and linear discriminant analysis (LDA), to diagnose colon cancer. Reproducible and clear differences between the spectra of cancer and normal colon tissues result in classification with high sensitivity, specificity and accuracy of 100%, 93.1% and 95.8%, respectively.
Furthermore, FTIR spectroscopy has been utilized for cancer diagnosis by analyzing other types of biological materials besides the commonly examined blood and tissue samples, such as urine
[28], bladder wash
[29], bile
[30] and sputum
[31][32] samples. Paraskevaidi et al.
[28] have performed ATR-FTIR spectroscopic analysis of urine samples using classification models, including partial least squares discriminant analysis (PLS-DA), principal component analysis with support vector machines (PCA-SVM) and genetic algorithm with linear discriminant analysis (GA-LDA), for the non-invasive diagnostic test of ovarian and endometrial cancers. Urine samples from patients with ovarian or endometrial cancer and healthy controls were evaluated and achieved up to 100% of sensitivity, specificity as well as accuracy with the identified biomarkers for both types of cancers. Evidently, these results demonstrated the potential of the technique for improved diagnosis when compared to the most commonly used serum biomarker for ovarian cancer, cancer antigen 125 (CA-125), which has been found to be unacceptable for early-stage detection due to its low sensitivity of 50–60% and it is only elevated in approximately 60% of patients
[50][51]. Apart from that, Lewis et al.
[31] have applied FTIR spectroscopy combined with hierarchical cluster analysis (HCA) and principal component analysis (PCA) to examine sputum samples for the diagnosis of lung cancer. Likewise, Lewis et al.
[31] obtained prominent significant wavenumbers which separate spectra between cancer and normal sputum samples. Interestingly, the spectral analysis showed that the wavenumbers were also able to differentiate lung cancer patients who had been previously diagnosed with breast cancer. The findings suggest that the techniques applied to sputum may have high sensitivity and specificity of greater than 80% for diagnosis using the small panel of significant wavenumbers, which compares more than favorably with current techniques of lung cancer detection. This enables the development of a non-invasive, cost-effective and high-throughput screening method for lung cancer.
Taken together, despite the sensitivity, specificity and accuracy of FTIR spectroscopy as a cancer diagnostic tool in these studies may be affected by factors such as the severity of the disease (e.g., stage and grade of cancer) in patient cases just as other diagnostic tests, the technique shows promising results for many different types of cancer when compared to existing diagnostic tests. Moreover, studies comparing different classification, stages and grades of cancer through FTIR analysis have been performed and are discussed in the following section. Although some studies may be preliminary and the sample size may be limited, these investigations demonstrated the strong potential of FTIR spectroscopy as a highly sensitive, specific and accurate cancer diagnostic tool that is valuable for further investigation and development.
4. Fourier Transform Infrared Spectroscopic Analysis of Cancer-Derived Extracellular Vesicles
4.1. Diagnostic Value of Extracellular Vesicles
Extracellular vesicles (EVs) are cell-derived membrane nanovesicles that are released to the extracellular space and circulation. They contribute to intercellular communication and reflect the physiological as well as pathological conditions in the body
[34]. EVs have recently gained the attention of researchers in the field of clinical research. These vesicles are a heterogenous population of particles varying between 30–1000 nm in diameter that are classified into a few subsets based on their size, density, morphology, biogenesis, origin, lipid composition, sedimentation characteristics and biochemical markers specifically present on their surface
[52][53]. These subsets include exosomes (30–100 nm), microvesicles and late endosomes (50–1000 nm), ectosomes (100–350 nm) as well as microparticles (100–1000 nm). Additionally, several apoptotic bodies (0.5–5 μm) and “small-size microparticles” (<50 nm) are also grouped as EVs.
Recent studies suggest that EVs are the transport form for various molecules such as mRNAs, miRNAs, cytokines, hormones, autoantigens, surface receptors and tissue coagulation factors, which might be paracrine regulators of target cell function and metabolism from their parental cells
[54][55][56]. Furthermore, the biological molecules encompassed in EVs are involved in diverse processes including proliferation, malignancy, vasculogenesis, inflammation, infections, tissue repair as well as growth and differentiation of tissues
[57][58]. EVs contribute to disease progression by modulating local and systemic effects in the body. In fact, EVs have been identified in many biological tissues and fluids, including blood and saliva, as targets of treatments and biomarkers of diseases
[59][60]. Studies have found that the characteristic morphological and molecular features of salivary exosomes of oral cancer patients were different from that of healthy individuals
[61][62]. To date, numerous evidences are present suggesting the use of EVs in diagnosis with promising predictive value in various diseases including cancer
[63][64], diabetes
[65], cardiovascular
[66][67] and autoimmune
[68] diseases as well as central nervous system disorders
[69]. Being released into bodily fluids, EVs could serve in nanomedicine as an invaluable source of non-invasive diagnostic specimen that is simpler and yet representative of the pathophysiological conditions in the body which overcome the limitations of the commonly used blood and tissue samples
[70]. Moreover, EVs can provide diagnostic information at successive time points for early detection and monitoring of both local and systemic diseases.
4.2. Analysis of Extracellular Vesicles Using Fourier Transform Infrared Spectroscopy
Despite numerous studies demonstrating the potential use of EVs in disease diagnosis, FTIR spectroscopic analysis of EVs for cancer diagnosis has not been widely investigated. One of such investigation is from Krafft et al.
[25] who have examined the diagnostic value of EVs in the screening of prostate cancer by using FTIR and Raman spectroscopy to perform a comprehensive comparative analysis between cancer and non-cancer EVs. Differential centrifugation of plasma and serum from patients with prostate cancer or benign prostatic hyperplasia as well as a healthy donor isolated two distinct EV fractions enriched with microvesicles and exosomes, which are the most abundant and well-investigated EVs. Cancer spectral signature of the blood EVs was utilized in a pilot study to detect prostate cancer from a test cohort of patients, including four with benign prostatic hyperplasia and another four with high-grade prostate carcinoma. The study concluded that the identified EV signature is a useful screening tool in cancer detection. Notably, the authors demonstrated that the isolation of EV fraction as an analyte is necessary because both the IR and Raman data resulted from direct analysis of unprocessed serum and plasma from cancer and healthy donors were almost identical and are insufficient to detect changes for cancer screening. Samples are usually dried before spectroscopic analysis to avoid the strong spectral contributions from water in the specimen from masking the spectrum differences. Hence, broad variations in the study observed between 1200 and 1400 cm
−1 from liquid but not in dry phases of the serum and plasma samples were considered irrelevant
[25].
In another study, Zlotogorski-Hurvitz et al.
[27] assessed the diagnostic potential of salivary exosomes for early detection of oral cancer using ATR-FTIR spectroscopy and machine learning techniques, including principal component analysis-linear discriminant analysis (PCA-LDA) and support vector machine (SVM) classification. Exosomes were isolated from whole saliva samples of oral cancer patients and healthy individuals using differential centrifugation. The findings showed that the IR spectra were consistently different between the two groups and that specific spectral signature for the cancer salivary exosomes was accurately distinguished from exosomes of healthy individuals. Furthermore, classification of samples resulted with high sensitivity, specificity and accuracy of 100%, 89% and 95%, respectively. Recently, we have coupled ATR-FTIR analysis of urinary EVs with PCA-LDA statistic model () in our laboratory as a novel strategy for non-invasive early detection of prostate cancer
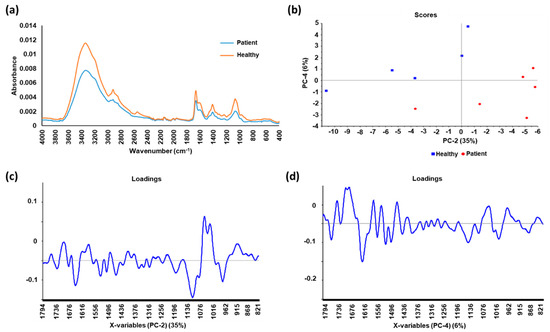
[26]. The spectral differences between the EVs from prostate cancer patients and healthy individuals as well as the analysis using linear discriminant analysis (LDA)-derived classifier, which achieved sensitivity of 83.33% and specificity of 60%, signifies the potential of ATR-FTIR technique as a point-of-care test for prostate cancer in urine.
Figure 1. The average spectra and principal component analysis (PCA) of urinary extracellular vesicle (EV) samples from prostate cancer patients and healthy individuals (a) The average spectra of the EV samples from patients has a lower absorbance compared to that from healthy individuals; (b) Score plot of second and fourth PCs, with corresponding percentage of explained variance in parentheses; (c) Loadings of the second PC with 35% of explained variance; (d) Loadings of the fourth PC with 6% of explained variance. Spectral peak differences between the two groups were revealed after analyzing the spectra with PCA, suggesting possible biomarkers for prostate cancer. Linear discriminant analysis (LDA) was used to derive a diagnostic classifier for prostate cancer from the spectra, which achieved sensitivity of 83.33% and specificity of 60%.
The ability of FTIR spectroscopy to provide accurate signature of biomolecular content of the analyte with small-scale preparation and high-speed detection, allows the development of a label-free disease-specific EV biomarker which is both time and cost-effective. However, the current most commonly used method in EV studies, differential centrifugation, only allows isolation of enriched fraction of the EV subtypes. Therefore, development of novel methodology for complete purification of each EV subtype would be required to aid in cancer biomarker identification as well as for the study of EVs and their role in cancer, which are still poorly understood. Furthermore, it is important to note that the promising findings of FTIR spectroscopy warrants further research for validation in larger patient cohorts with varying stages and different grades of cancer for clinical translation of the methodology.
5. Conclusions
Taken together, FTIR spectroscopy holds promise for use as a novel clinical tool for cancer. Importantly, this is evident through the numerous investigations using the technique on multiple cancer types (). The technique offers high sensitivity, specificity and accuracy in cancer detection when compared to currently used diagnostic methods. Furthermore, FTIR spectroscopy enables accurate and objective classification, staging and grading for cancer management as opposed to the present gold standard, histopathological diagnosis, to guide treatments and predict patient prognosis. Notably, automated marker-free FTIR spectroscopy allows higher accuracy and reproducibility in cancer diagnosis, while eliminating the need of complex and time-consuming clinical processing of tissue samples required in existing computer-aided histopathological diagnosis. In addition, it has been demonstrated that FTIR spectroscopy could be used to evaluate surgical resection margins rapidly and objectively to assist surgical decision making which will improve long-term survival and postoperative recovery of patients when compared to the common intraoperative pathological examination. The technique has also been utilized to monitor cancer treatment response and follow-up of patients for treatment planning, early detection of recurrences and support for mental or psychosocial stress with more rapid, sensitive and specific results as compared to current methods. Hence, FTIR spectroscopy would be crucial to accelerate point-of-care decisions and potentially revolutionize cancer diagnostics in personalized medicine.
Table 2. Studies of various cancer types using FTIR spectroscopy discussed in this review article.
| Cancer Type |
Title of Study |
References |
| Colorectal Cancer |
Application of linear discriminant analysis and attenuated total reflectance Fourier transform infrared microspectroscopy for diagnosis of colon cancer |
[4] |
| The use of FTIR-ATR spectrometry for evaluation of surgical resection margin in colorectal cancer: a pilot study of 56 samples |
[24] |
| Early detection of colorectal cancer relapse by infrared spectroscopy in “normal” anastomosis tissue |
[23] |
| Use of FTIR spectroscopy and PCA-LDC analysis to identify cancerous lesions within the human colon |
[22] |
| Prostate Cancer |
Study of prostate cancer-derived extracellular vesicles in urine using IR spectroscopy |
[26] |
| Investigating FTIR based histopathology for the diagnosis of prostate cancer |
[20] |
| A specific spectral signature of serum and plasma-derived extracellular vesicles for cancer screening |
[25] |
| Leukemia |
Distinction of leukemia patients’ and healthy persons’ serum using FTIR spectroscopy |
[17] |
| Pre-screening and follow-up of childhood acute leukemia using biochemical infrared analysis of peripheral blood mononuclear cells |
[19] |
| Ovarian and/or Endometrial Cancers |
Potential of mid-infrared spectroscopy as a non-invasive diagnostic test in urine for endometrial or ovarian cancer |
[28] |
| Segregation of ovarian cancer stage exploiting spectral biomarkers derived from blood plasma or serum analysis: ATR-FTIR spectroscopy coupled with variable selection methods |
[16] |
| Lung Cancer |
Evaluation of FTIR spectroscopy as a diagnostic tool for lung cancer using sputum |
[31] |
| Marker-free automated histopathological annotation of lung tumor subtypes by FTIR imaging |
[21] |
| Oral, Oropharyngeal, and/or Laryngeal Cancer |
Fourier transform infrared for noninvasive optical diagnosis of oral, oropharyngeal, and laryngeal cancer |
[32] |
| FTIR-based spectrum of salivary exosomes coupled with computational-aided discriminating analysis in the diagnosis of oral cancer |
[27] |
| Gastric Cancer |
Comparison of serum from gastric cancer patients and from healthy persons using FTIR spectroscopy |
[18] |
| Breast Cancer |
Diagnosis of breast cancer with infrared spectroscopy from serum samples |
[15] |
| Bladder Cancer |
Bladder cancer diagnosis from bladder wash by Fourier transform infrared spectroscopy as a novel test for tumor recurrence |
[29] |
| MalignantBiliary Strictures |
Bile analysis using high-throughput FTIR spectroscopy for the diagnosis of malignant biliary strictures: a pilot study in 57 patients |
[30] |
| Skin Cancer |
FT-IR spectroscopy study in early diagnosis of skin cancer |
[71] |
Even though FTIR spectroscopy may not be able to identify specific molecules when compared to molecular tests, it allows the qualitative and quantitative analyses of different classes of molecules simultaneously such as nucleic acids, proteins and lipids. Therefore, the technique provides the overall status of the analyzed specimen and is ideal for complicated diseases like cancer, which are multifactorial and examining isolated molecules alone may not give complete information
[72]. Hence, this feature provides the advantage of being able to reveal the whole “omics” of the examined specimen and identify more than one cancer biomarker. Additionally, the comprehensive information of patients provided by FTIR spectroscopy also reveals new insights in cancer research by allowing better understanding of mechanisms underlying carcinogenesis, such as the significant quantitative changes in spectral regions of nucleic acids and proteins indicate metabolic dysfunction in cancer cells and can be considered as biomarkers
[22]. Interestingly, Kyriakidou et al.
[71] have reported spectroscopic data showing melanoma alters the permeability of cell membrane and changes the native B-DNA form to Z-DNA form, suggesting possible biomarker for skin cancer while further understanding the biochemical changes in the disease. The promising findings of FTIR spectroscopy in cancer screening, diagnosis, management and monitoring demonstrates its value for further research and development. Besides validating the technique in larger patient cohorts, challenges in technological development, standardization of sample collection, storage and preparation methods, data acquisition procedures, pre- and post-processing of spectral data as well as classification models will have to be overcome for successful clinical translation.