
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Nuno Mariz-Ponte | + 2626 word(s) | 2626 | 2021-05-14 04:30:40 | | | |
| 2 | Nora Tang | + 22 word(s) | 2648 | 2021-06-11 03:53:19 | | |
Video Upload Options
Daily UV-supplementation during the plant fruiting stage of tomato (Solanum lycopersicum L.) growing indoors may produce fruits with higher nutraceutical value and better acceptance by consumers. However, it is important to ensure that the plant’s performance during this stage is not compromised by the UV supplement. We studied the impact of UV-A (1 and 4 h) and UV-B (2 and 5 min) on the photosynthesis of greenhouse-grown tomato plants during the fruiting/ripening stage. After 30 d of daily irradiation, UV-B and UV-A differently interfered with the photosynthesis. UV-B induced few leaf-necrotic spots, and effects are more evidenced in the stimulation of photosynthetic/protective pigments, meaning a structural effect at the Light-Harvesting Complex. UV-A stimulated flowering/fruiting, paralleled with no visible leaf damages, and the impact on photosynthesis was mostly related to functional changes, in a dose-dependent manner. Both UV-A doses decreased the maximum quantum efficiency of photosystem II (Fv/Fm), the effective efficiency of photosystem II (ΦPSII), and gas exchange processes, including net carbon assimilation (PN). Transcripts related to Photosystem II (PSII) and RuBisCO were highly stimulated by UV supplementation (mostly UV-A), but the maintenance of the RuBisCO protein levels indicates that some protein is also degraded. Our data suggest that plants supplemented with UV-A activate adaptative mechanisms (including increased transcription of PSII peptides and RuBisCO), and any negative impacts on photosynthesis do not compromise the final carbohydrate balances and plant yield, thus becoming a profitable tool to improve precision agriculture.
1. Introduction
Tomato (Solanum lycopersicum L.) is amongst the most popular and consumed crop species worldwide, with vast agricultural and economic importance. Around 182.3 million tons of tomato fruits were produced in 2018 (www.fao.org/faostat/, accessed in September of 2020), generating a global revenue of 190.4 billion dollars [1]. Its excellent acceptance by the consumers is due to its multiple gastronomic uses and other features like its taste, color, and high nutritional value [2][3].
This crop is produced in open fields and indoors, including greenhouses [4]. Tomato production outdoors, which is widely used in the pulp industry, allows plants to grow so that they are naturally adapted to the solar ultraviolet (UV) radiation. In indoor cultures (mostly dedicated to table tomato), UV-exposure is usually neglected, since greenhouse building materials are diverse and include glass and polycarbonate, which totally or partially filter solar UV radiation. Additionally, artificial light may be used as a supplement or as the only source of light [5]. Although previous studies demonstrated that fruits and vegetables can be grown indoors out of season, they are described as having low nutritional and organoleptic attractiveness [6].
Nevertheless, in the last decade, a new agricultural paradigm emerged, which considers UV-supplementation as a strategy to improve crop yield and/or quality [7][8][9][10][11]. Besides, UV-irradiation systems do not pose legal restrictions, and some systems (e.g., UV-A, LED-UV) are easily affordable. Controlled UV-supplementation can be a powerful tool not only to control indoors-crop pests and diseases but also to promote the synthesis of defense compounds and increase the nutritional quality and organoleptic attributes of the edible parts, which can advance the current agriculture requirements [11][12][13]. However, it is also necessary to ensure that the application of these UV supplements does not compromise the plant’s performance (namely photosynthesis) during the fruiting stage.
For instance, plants exposed to excessive UV rays, especially UV-B, suffer biological changes, which include decreased growth and yield, and leaf chlorosis and/or necrosis [14][15]. Defense mechanisms in response to harmful light (intensity or quality) develop defense mechanisms mediated by molecular photoreceptors [13][16][17]. Photoreceptors sense and transduce light signals through distinct intracellular signaling pathways (which include the modulation of light-regulated genes) and ultimately lead to adaptive changes at the cellular and systemic levels [18]. Effects of UV-B are usually more deleterious than a similar dose of UV-A, but high levels of UV-A may lead to responses identical to those induced by lower levels of UV-B [19].
During maturation, fruit tissues suffer a transition from partially to true heterotrophy, meaning dramatic metabolic changes to the plant [3]. During this stage, leaf photosynthesis is crucial to export fixed carbon (mostly sucrose) to non-photosynthetic fruit sink-tissues [20]. Thus, any change in the leaf photosynthetic efficiency during the fruiting/ripening stage will alter this “source-sink” dynamics, influencing fruit yield and quality. However, beneficial effects of small amounts of UV radiation on crop yield, and how UV-triggers photosynthetic signals that may be used in precision agriculture/horticulture are far less studied, being the majority of available studies on the UV impacts on photosynthesis, and at the same time not using the crop’s fruiting/ripening stage, thus mainly focused on the negative effects of excessive UV rather than on physiologically tolerable UV-doses [21][22][23][24][25]. The beneficial effects of small amounts of UV radiation on crop yield, and how UV-triggers photosynthetic signals that may be used in precision agriculture/horticulture are far less studied.
UV-A light that ranges between 315 and 400 nm has beneficial effects on the growth and vigor of seedlings [10]. It also increases chlorophyll and carotenoid levels and stimulates polyphenol pathways and levels [26][27], thus it may be used to improve the yield and/or quality of crops. In this line, Lee et al. [15] used LED UV-A to increase the biomass of kale plants. We have shown that UV-A irradiation during the pre-harvest period is effective in increasing ripening synchronization and the fruit’s nutritional properties, potentially making these fruits more likable to buyers [11].
The way moderate doses of UV-rays (quality and quantity) modulate specific targets of the leaf photosynthesis (including the thylakoidal electron transport chain and the Calvin cycle) during the fruiting/ripening stage remains a matter of debate. After selecting the ideal type, dose, and period of UV supplementation to enhance the nutraceutical quality of fruits and vegetables (reviewed by Huché-Thélier et al. [28]), we must evaluate its impact on the leaf photosynthesis, including at the transcriptional and post-transcriptional levels.
Moreover, the plant’s responses to UV are an integration of its cross-talks with both environmental factors and the developmental stage of the plant (reviewed by Yadav et al. [29]). UV-B inhibited RuBisCO activity [21], but Gao et al. [24] showed that a low dose of UV-B upregulated proteins related to chloroplast structure, light reactions, oxygen-evolving enhancer proteins, and ATP synthase. The physiological understanding of the effects to low doses of UV-A/B [25][30][31], namely regarding the photosynthetic performance and carbon metabolism, is essential to evaluate the possible application of these irradiations in indoors crop production.
Following our previous work that showed biochemical and organoleptic benefits of UV supplementation to tomato fruit [11], we aim to unveil how the different quality and intensity of low (physiologically tolerated) doses of UV-A and UV-B influence the plant photosynthesis and carbon metabolism during the ripening stage of the plants, referring to physiological, biochemical and molecular tools. By distinguishing characteristic effects associated with specific UV wavelengths, and dose, we may be able to select only precise UV-supplementation indoors, namely in greenhouses, for precision horticulture.
2. Discussion
With the paradigm of producing “more with less”, the exponential population growth, and the alarming climate changes and pests and diseases that affect crops worldwide, the control of multiple variables in indoor production systems represent a new era of precision agriculture and a challenge to understand the mechanisms underlying the physiological and molecular responses of the crops to those variables. Mimicking natural conditions, including natural solar light, is a key challenge to increase the organoleptic and nutritional value of edible parts. Greenhouses create an ideal environment for intensive and precision crop production. However, they are usually built with UV-absorbing materials that reduce the benefits of UV-A and UV-B on crops along their life cycle. Comparing with their outdoor growing counterparts, crops growing indoors may have repressed metabolic pathways that are triggered by UV-A and UV-B plant sensors [32]. A previous study showed that the fruits produced indoors with the supplementation of low UV-A/B radiation had a higher quality to the consumers [11]; even so, it is necessary to ensure that this supplementation (quality and dose) is not too deleterious to plant growth and photosynthesis during fruiting and ripening stages.
This work shows that UV-B may reduce plant length, principally with UV-B 2 min, which may indicate that at this low UV-B level, there is already an influence in cell division and cell expansion as reported for UV-B by Bandurska et al. [33]. Cell expansion depends on variables like the leaf water content, turgor pressure, and cell extensibility. This relation may thus support the correlation of length with %WC, shown by the PCA (Figure 1). This correlation also suggests an adjustment of the metabolism of the UV-treated tomato plants to restrict water use, which is supported by the reduced stomata aperture (Figure 1).
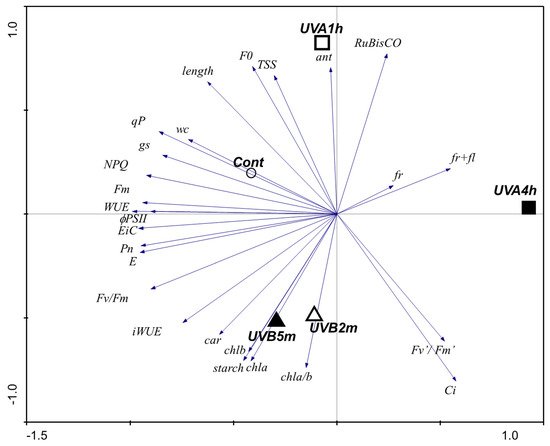
Figure 1. PCA analysis of functional responses of tomato fruit plants exposed to UV-A (1 and 4 h) and to UV-B (2 and 5 min) for 30 d.
The flowering and fruiting/ripening stages impose dramatic changes to the “source-sink” mobilization of photo-assimilates. In this process, leaf photosynthesis plays a critical role in the supply of carbohydrates to flowers/fruits. Under the stimulus of moderate UV-B, leaves shifted their metabolism to alternative secondary pathways, towards increasing, for example, flavonoids/phenols [34][35]. While this shift may enrich the edible parts with valuable secondary compounds increasing their nutraceutical and/or sensorial value [11], it also implies that the leaves may suffer a decrease in the necessary photo-assimilates to supply the fruits.
As can be seen from the PCA, the low levels of UV-B and UV-A used here differently affect the photosynthetic parameters. UV-B 5 min increased Chl a and Chl b, supporting that UV-B stimulates the pathways involved in chlorophyll synthesis. Likewise, a short-term pre-harvest supplement of UV-B did not compromise the levels of Chl a, Chl b, or carotenoids in Ocimum basilicum leaves [26]. In tomato, the UV-B 5 min dose used stimulated the carotenoid pathways, supporting investment in these UV-protecting pigments and scavengers of reactive oxygen species (ROS), towards protecting chlorophylls from photo-oxidative damage caused by any excessive UV-B irradiation, as also proposed by Yadav et al. [29]. Phenolic compounds are also important protective compounds, associated with sensorial attributes (smell and taste). At these doses, UV-A has little impact on pigment levels, but compromises the efficiency of PSII photochemistry, although not significantly compromising the plant’s performance, growth, or fruit yield. Mariz-Ponte et al. [11] used the same doses of UV-A and demonstrated that, besides stimulating the antioxidant and antiradical activity of the fruit, and richness in phenolic compounds (e.g., flavonoids), UV-A also led to fruits more attractive to the consumers, and thus more likable to buyers. These beneficial aspects occur despite the negative correlation of UV-A with some photosynthetic parameters in the same plants, as shown here. The Fv/Fm ratio is a widely used indicator of photoinhibition or other injuries at the PSII complexes [36]. However, the UV-A plants were able to maintain Fv/Fm values close to 0.8, despite the decrease in UV-A conditions, showing a ratio characteristic of unstressed plants [37].
The profile shown by NPQ in response to UV-A (a stimulation for lower doses and a drastic decrease with the increase in the dose, comparing to the control) suggests a hormesis effect, meaning that lower doses of UV-A promote heat dissipation (which includes photo-protective mechanisms), while higher doses compromise this strategy. NPQ of chlorophyll a fluorescence is an indicator of the level of non-radiative energy dissipation in the light-harvesting complex of PSII (LHC II), which is attributed to the prevention of electron transfer chain over-reduction, preventing photodamage. The decrease in NPQ observed at UV-A 4 h may be supported by the decrease in the light-harvesting antenna size (lowered by Fm) and/or by other causes of PSII inactivation [36]. The reaction centers, which are functionally involved in the qP, are stimulated by the lowest dose of UV-A. Interestingly, the higher UV-A dose tested here decreased both the availability of the reaction centers to receive photons and ΦPSII, which suggests that UV-A 4 h already imposes some mild stress to plants (Figure 2). This mild stress may be the basis for the biochemical changes observed in the fruits as previously described [11]. Conversely, UV-B did not induce functional stress on the fluorescence/quenching parameters, but rather changes in the protective LHC-pigments composition, thus meaning a structural change.
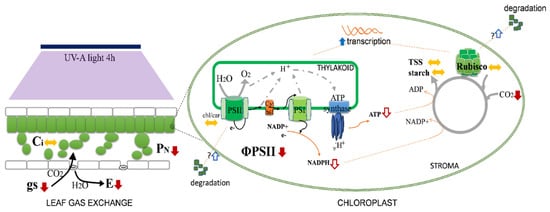
Figure 2. Major photosynthetic impacts and changes induced by moderate UV-A 4 h/day, for 30 d, in tomato flowering plants. Overall, the PSII fluorescence is affected by the decreased efficiency of ΦPSII, although LHC-pigment (Chl/car = chlorophyll/carotenoids) levels are not affected. This leads to fewer electrons being transported and thus a decrease in NADPH and ATP production and availability for the Calvin cycle. This reduction is related with the decrease in the net photosynthetic rate (PN), meaning that internal CO2 concentration (Ci) is not so depleted, and the stomatal conductance (gs) may decrease, therefore decreasing transpiration rate (E). Simultaneously, a degradation of RuBisCO may occur, but it can be replaced by new protein due to the stimulated accumulation of its transcripts (and increase its transcription), which overall may reset the negative impacts on the Calvin cycle, thus not having negative impacts on total amounts of soluble sugars and starch. Solid red arrows mean a decrease and solid blue arrows an increase. Dashed red and blue arrows mean a putative decrease and increase, respectively.
We unveil for the first time the relationship between genes involved in RuBisCO response to high doses of UV-A/B light supplementation versus tomato plants growing without UVs. These data showed a tendency (p < 0.05) to increase the RuBisCO transcripts in the high doses of UV-A/B tested, contrary to that demonstrated on pea leaves under UV-B [38]. UV-A acts at the level of transcriptional regulation by increasing the transcripts of psbB, which encode for CP47. This protein is located at the antenna pigment complex CP43-47 and binds to chlorophylls and carotenes, acting in the transfer of energy from the LHC antenna to the photochemical reaction center. The transcript psbA encoding for the protein D1 (a protein involved in receiving electrons in the PSII) presented a similar behavior. D1 protein has shown susceptibility to UV-light [39][40]. Additionally, UV-A increased several proteins of the PSII in Taxus ssp. plants [41]. Based on these findings, we hypothesize that in UV-exposed tomato leaves, there might be a light-induced degradation of these proteins in the PSII, compensated by an increase in transcription to synthesize new proteins. Nevertheless, the possibility, within a process adaptive to UV radiation, that new PSII centers may be under generation to compensate the lower ΦPSII, should also be considered.
The PCA clearly shows that UV-A 4 h has an impact in the gas exchange parameters by the negative correlation of the vectors. While stomatal closure occurred and PN decreased, the levels of Ci remained unchanged, which together with the increase in RuBisCO transcriptional levels may also support a higher investment on more RuBisCO protein to support Calvin cycle dynamics (Figure 2). This is supported by the increase in both rbcL and rbcS transcripts, which show that these high doses of UV already stimulated the synthesis of RuBisCO subunits. On the other hand, the fact that the relative content of RuBisCO is not affected supports the idea that UV might also induce RuBisCO degradation, which is offset by an increase in the transcript levels, allowing the maintenance of soluble sugars and/or starch in UV-supplemented leaves.
In conclusion, we have demonstrated previously that UV-A/B supplementation of tomato plants during fruiting/ripening increases the number of fruits with a richer nutraceutical composition and their preference by consumers [11]. Using the same UV-supplementation conditions described by Mariz-Ponte et al. [11], we demonstrate here that both UV light quality and dose differently target the leaf photosynthesis during the fruiting/ripening stage of the plant. UV-A had more positive and negative effects on photosynthetic functional parameters, while UV-B acted more at the structural level (stimulating pigment levels). The identification of targets specifically susceptible to UV quality/quantity light may provide a useful tool to selectively use UV-supplementation to produce better fruits (increased nutraceutical and organoleptic value) without detrimental effects on the photosynthesis of the plant, thus improving greenhouse crop production, with UV-A treatment for 1 h presenting potential as a better supplementation for fruit quality.
References
- Research and Market. World-Tomato-Market Analysis, Forecast, Size, Trends and Insights; IndexBox Inc.: Walnut, CA, USA, 2020.
- Chaudhary, P.; Sharma, A.; Singh, B.; Nagpal, A.K. Bioactivities of phytochemicals present in tomato. J. Food Sci. Technol. 2018, 55, 2833–2849.
- Quinet, M.; Angosto, T.; Yuste-Lisbona, F.J.; Blanchard-Gros, R.; Bigot, S.; Martinez, J.P.; Lutts, S. Tomato Fruit Development and Metabolism. Front. Plant Sci. 2019, 10, 1554.
- Torres Pineda, I.; Lee, Y.D.; Kim, Y.S.; Lee, S.M.; Park, K.S. Review of inventory data in life cycle assessment applied in production of fresh tomato in greenhouse. J. Clean. Prod. 2020, 282, 124395.
- Wu, Q.; Su, N.; Shen, W.; Cui, J. Analyzing photosynthetic activity and growth of Solanum lycopersicum seedlings exposed to different light qualities. Acta Physiol. Plant 2014, 36, 1411–1420.
- Muñoz, P.; Antón, A.; Nuñez, M.; Paranjpe, A.; Ariño, J.; Castells, X.; Rieradevall, J. Comparing the environmental impacts of greenhouse versus open-field tomato production in the Mediterranean region. Int. Symp. High Technol. Greenh. Syst. Manag. Greensys2007 2007, 801, 1591–1596.
- Gil, M.; Bottini, R.; Berli, F.; Pontin, M.; Silva, M.F.; Piccoli, P. Volatile organic compounds characterized from grapevine (Vitis vinifera L. cv. Malbec) berries increase at pre-harvest and in response to UV-B radiation. Phytochemistry 2013, 96, 148–157.
- Barnes, P.; Flint, S.; Ryel, R.; Tobler, M.; Barkley, A.E.; Wargent, J. Rediscovering leaf optical properties: New insights into plant acclimation to solar UV radiation. Plant Physiol. Biochem. 2015, 93, 94–100.
- Neugart, S.; Schreiner, M. UVB and UVA as eustressors in horticultural and agricultural crops. Sci. Hortic. 2018, 234, 370–381.
- Mariz-Ponte, N.; Mendes, R.J.; Sario, S.; Melo, P.; Santos, C. Moderate UV-A supplementation benefits tomato seed and seedling invigoration: A contribution to the use of UV in seed technology. Sci. Hortic. 2018, 235, 357–366.
- Mariz-Ponte, N.; Martins, S.; Gonçalves, A.; Correia, C.M.; Ribeiro, C.; Dias, M.C.; Santos, C. The potential use of the UV-A and UV-B to improve tomato quality and preference for consumers. Sci. Hortic. 2019, 246, 777–784.
- Nelson, J.A.; Bugbee, B. Economic analysis of greenhouse lighting: Light emitting diodes vs. high-intensity discharge fixtures. PLoS ONE 2014, 9, e99010.
- Escobar-Bravo, R.; Chen, G.; Kim, H.K.; Grosser, K.; van Dam, N.M.; Leiss, K.A.; Klinkhamer, P.G. Ultraviolet radiation exposure time and intensity modulate tomato resistance to herbivory through activation of jasmonic acid signaling. J. Exp. Bot. 2019, 70, 315–327.
- Bashri, G.; Singh, M.; Mishra, R.K.; Kumar, J.; Singh, V.P.; Prasad, S.M. Kinetin regulates UV-B-induced damage to growth, photosystem II photochemistry, and nitrogen metabolism in tomato seedlings. J. Plant Growth Regul. 2018, 37, 233–245.
- Lee, J.; Oh, M.; Son, K. Short-Term Ultraviolet (UV)-A Light-Emitting Diode (LED) Radiation Improves Biomass and Bioactive Compounds of Kale. Front. Plant Sci. 2019, 10, 1042.
- Dias, M.C.; Pinto, M.D.; Correia, C.; Silva, A.M.S.; Santos, C. UV-B radiation modulates physiology and lipophilic metabolite profile in Olea europaea. J. Plant Physiol. 2018, 222, 39–50.
- Katsoulas, N.; Bari, A.; Papaioannou, C. Plant Responses to UV Blocking Greenhouse Covering Materials: A Review. Agronomy 2020, 10, 1021.
- Yokawa, K.; Kagenishi, T.; Baluska, F. UV-B Induced Generation of Reactive Oxygen Species Promotes Formation of BFA-Induced Compartments in Cells of Arabidopsis Root Apices. Front. Plant Sci. 2015, 6, 1162.
- Brazaitytė, A.; Viršilė, A.; Jankauskienė, J.; Sakalauskienė, S.; Sirtautas, R.; Novičkovas, A.; Dabašinskasm, L.; Miliauskienė, J.; Vaštakaitė, V.; Bagdonavičienė, A.; et al. Effect of supplemental UV-A irradiation in solid-state lighting on the growth and phytochemical content of microgreens. Int. Agrophys. 2013, 29, 13–22.
- Osorio, S.; Ruan, Y.; Fernie, A.R. An update on source-to-sink carbon partitioning in tomato. Front. Plant Sci. 2014, 5, 516.
- Araújo, M.; Santos, C.; Costa, M.; Moutinho-Pereira, J.; Correia, C.; Dias, M.C. Plasticity of young Moringa oleifera L. plants to face water deficit and UVB radiation challenges. J. Photochem. Photobiol. B Biol. 2016, 162, 278–285.
- Inostroza-Blancheteau, C.; Acevedo, P.; Loyola, R.; Arce-Johnson, P.; Alberdi, M.; Reyes-Díaz, M. Short-term UV-B radiation affects photosynthetic performance and antioxidant gene expression in highbush blueberry leaves. Plant Physiol. Biochem. 2016, 107, 301–309.
- Khudyakova, A.Y.; Kreslavski, V.D.; Shmarev, A.N.; Lyubimov, V.Y.; Shirshikova, G.N.; Pashkovskiy, P.P.; Kuznetsov, V.V.; Allakhverdiev, S.I. Impact of UV-B radiation on the photosystem II activity, pro-/antioxidant balance and expression of light-activated genes in Arabidopsis thaliana hy4 mutants grown under light of different spectral composition. Photochem. Photobiol. B Biol. 2019, 194, 14–20.
- Gao, L.; Wang, X.; Li, Y.; Han, R. Chloroplast proteomic analysis of Triticum aestivum L. seedlings responses to low levels of UV-B stress reveals novel molecular mechanism associated with UV-B tolerance. Environ. Sci. Pollut. Res. 2019, 26, 7143–7155.
- Soriano, G.; Del-Castillo-Alonso, M.Á.; Monforte, L.; Tomás-Las-Heras, R.; Martínez-Abaigar, J.; Núñez-Olivera, E. Photosynthetically-active radiation, UV-A and UV-B, causes both common and specific damage and photoprotective responses in the model liverwort Marchantia polymorpha subsp. ruderalis. Photochem. Photobiol. Sci. 2019, 18, 400–412.
- Nascimento, L.B.; Brunetti, C.; Agati, G.; Lo Iacono, C. Short-Term Pre-Harvest UV-B Supplement Enhances the Polyphenol Content and Antioxidant Capacity of Ocimum basilicum Leaves during Storage. Plants 2020, 9, 797.
- Mariz-Ponte, N.; Mendes, R.J.; Sario, S.; de Oliveira, J.F.; Melo, P.; Santos, C. Tomato plants use non-enzymatic antioxidant pathways to cope with moderate UV-A/B irradiation: A contribution to the use of UV-A/B in horticulture. J. Plant Physiol. 2018, 221, 32–42.
- Huché-Thélier, L.; Crespel, L.; Gourrierec, J.L.; Morel, P.; Sakr, S.; Leduc, N. Light signaling and plant responses to blue and UV radiations-Perspectives for applications in horticulture. Environ. Exp. Bot. 2016, 121, 22–38.
- Yadav, A.; Bakshi, A.; Yadukrishnan, P.; Lingwan, M.; Dolde, U.; Wenkel, S.; Masakapalli, S.; Datta, S. The B-Box-Containing MicroProtein miP1a/BBX31 Regulates Photomorphogenesis and UV-B Protection. Plant Physiol. 2019, 179, 1876–1892.
- Kang, S.; Zhang, Y.; Zhang, Y.; Zou, J.; Yang, Q.; Li, T. Ultraviolet-A radiation stimulates growth of indoor cultivated tomato (Solanum lycopersicum) seedlings. HortScience 2018, 53, 1429–1433.
- Qian, M.; Kalbina, I.; Rosenqvist, E.; Jansen, M.A.; Teng, Y.; Strid, Å. UV regulates the expression of phenylpropanoid biosynthesis genes in cucumber (Cucumis sativus L.) in an organ and spectrum dependent manner. Photochem. Photobiol. Sci. 2019, 8, 424–433.
- Wargent, J.; Nelson, B.; McGhie, T.; Barnes, P. Acclimation to UV-B radiation and visible light in Lactuca sativa involves up-regulation of photosynthetic performance and orchestration of metabolome-wide responses. Plant Cell Environ. 2015, 38, 929–940.
- Bandurska, H.; Niedziela, J.; Chadzinikolau, T. Separate and combined responses to water deficit and UV-B radiation. Plant Sci. 2013, 213, 98–105.
- Guidi, L.; Brunetti, C.; Fini, A.; Agati, G.; Ferrini, F.; Gori, A.; Tattini, M. UV radiation promotes flavonoid biosynthesis, while negatively affecting the biosynthesis and the de-epoxidation of xanthophylls: Consequence for photoprotection? Environ. Exp. Bot. 2016, 127, 14–25.
- Machado, F.; Dias, C.; Pinho, P.; Araújo, A.; Pinto, D.; Correia, C.; Santos, C. Photosynthetic performance and volatile organic compounds profile in Eucalyptus globulus after UVB radiation. Environ. Exp. Bot. 2017, 140, 141–149.
- Hou, H.; Najafpour, M.M.; Moore, G.F.; Allakhverdiev, S.I. Photosynthesis: Structures, Mechanisms and Applications; Springer International Publishing: Cham, Switzerland, 2017; ISBN 978-3-319-48873-8.
- Maxwell, K.; Johnson, G.N. Chlorophyll fluorescence-a practical guide. J. Exp. Bot. 2000, 51, 659–668.
- Mackerness, S.H.; Surplus, S.L.; Jordan, B.R.; Thomas, B. Ultraviolet-B effects on transcript levels for photosynthetic genes are not mediated through carbohydrate metabolism. Plant Cell Environ. 1997, 20, 1431–1437.
- Kiss, E.; Kós, P.B.; Chen, M.; Vass, I. A unique regulation of the expression of the psbA, psbD, and psbE genes, encoding the 01, 02 and cytochrome b559 subunits of the PSII complex in the chlorophyll d containing cyanobacterium Acaryochloris marina. Biochim. Biophys. Acta 2012, 1817, 1083–1094.
- Nouri, M.Z.; Moumeni, A.; Komatsu, S. Abiotic stresses: Insight into gene regulation and protein expression in photosynthetic pathways of plants. Int. J. Mol. Sci. 2015, 16, 20392–20416.
- Zheng, W.; Komatsu, S.; Zhu, W.; Zhang, L.; Li, X.; Cui, L.; Tian, J. Response and Defense Mechanisms of Taxus chinensis Leaves Under UV-A Radiation are Revealed Using Comparative Proteomics and Metabolomics Analyses. Plant Cell Physiol. 2016, 57, 1839–1853.




