The recent viral pandemic in Wuhan, Hubei, China has led to the identification of a new species of beta-coronaviruses, able to infect humans, the 2019-nCoV, later named SARS-CoV-2. SARS-CoV-2 causes a clinical syndrome named COVID-19, which presents with a spectrum of symptoms ranging from mild upper respiratory tract infection to severe pneumonia, with acute respiratory distress syndrome and death. All age groups are susceptible to the infection, but children, especially infants, seem to be partially spared, having a more favorable clinical course than other age groups. There is currently no clear evidence showing vertical transmission and intrauterine SARS-CoV-2 infection in fetuses of women developing COVID-19 pneumonia in late pregnancy, and even if transmission is possible, the SARS-CoV2 positivity of the mother does not require delivery by caesarean section, does not contraindicate the management of the infant in rooming-in and allows breastfeeding.
1. Introduction
Coronaviridae is a family of enveloped, positive-strand RNA viruses that include four genera: alpha-coronavirus (α-CoV), beta-coronavirus (β-CoV), gamma-coronavirus (γ-CoV) and delta-coronavirus (δ-CoV)
[1]. Six species of human coronaviruses (HCoV) were previously known. Four of them produce mild respiratory symptoms: HCoV-229E and HCoV-NL63 belong to α-CoV genus and HCoV-OC43 an HCoV-HKU1 belong to β-CoV genus
[2][3]. Other two β-coronaviruses (severe acute respiratory syndrome coronavirus—SARS-CoV—and Middle East respiratory syndrome coronavirus—MERS-CoV), that moved from animal reservoirs, have been identified as causing agents of severe respiratory outbreaks.
Severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) is the seventh coronavirus that is contagious in humans. In December 2019, it was identified in China as the causing agent of coronavirus disease 2019 (COVID-19). The World Health Organization (WHO) declared this outbreak as a pandemic in March 2020. SARS-CoV-2 is a new β-CoV, associated with high contagiousness and fatality rate, up to 2% in infected subjects. Human-to-human transmission occurs primarily via respiratory droplets within a range of about 1.8 m
[4], but can also occur through close contact, the fecal–oral route and ocular surface
[5][6].
The virus has been identified using reverse transcription polymerase chain reaction (RT-PCR) in bronchoalveolar lavage fluid, saliva, and particularly in the nasopharyngeal swab of infected patients. The incubation period can vary between 2 and 14 days, being in most cases 3–7 days. SARS-CoV-2 primarily affects the respiratory tract, but potentially involves all organs and systems, including the placenta.
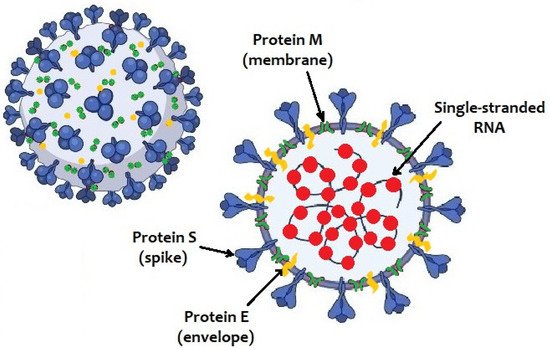
The SARS-CoV-2 genome consists in a positive-sense single-stranded RNA. Like other coronaviruses, it has four structural proteins: spike protein (S), membrane protein (M), nucleocapsid protein (N) and envelope protein (E) ().
Figure 1. Structure of SARS-CoV-2 virus.
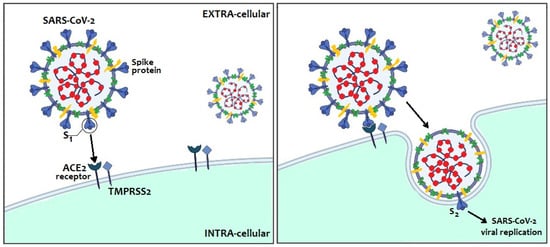
Protein S plays a key role in the pathogenesis of infection in humans since it allows to the virus to anchor itself to the host receptors and entry into the host cells. It has two major subunits, S1 and S2. The distal S1 subunit, with two structural domains (the receptor-binding domain—RBD—and the N-terminal galectin-like domain—S1-NTD), has a role in receptor recognition and binding to the angiotensin-converting enzyme 2 (ACE2) receptor of host cells, while the membrane-anchored S2 subunit mediates the fusion of the viral and the host cell membranes
[7] ().
Figure 2. SARS-CoV-2 entry into host cells through its receptor-binding domain (RBD) and its affinity to human receptor ACE2.
SARS-CoV-2 infects humans through binding between the RBD-domain of protein S and human receptor ACE2, widely expressed in many human cells, such as those of the respiratory tract, gut, uterus, ovaries and placenta
[8][9]. Furthermore, protein S must be cleaved by certain host proteases, transmembrane peptidase/serine subfamily member 2 (TMPRSS2), as well as other lysosomal peptidases (e.g., cathepsins L and B), to maintain its infectious capacity and lead to virion entry and fusion into the human cells.
2. Transplacental Transmission of SARS-CoV-2
Despite the growing body of literature about the current SARS-CoV-2 pandemic, the impact of the virus contracted during pregnancy on the mother and fetus is still to be determined
[10]. In past SARS and influenza H1N1 pandemics, pregnant women appeared to be more susceptible to infection in its most serious clinical forms, with a mortality much higher than that observed in the general population
[11]. Available data on SARS-CoV-2 infection in pregnancy during the current pandemic are yet to be interpreted. It has been reported that pregnant women would be partially spared from infections with a more severe course, although pathophysiological changes associated with pregnancy (elevation of the diaphragm, increased oxygen consumption, impaired immune response) makes this category of patients more susceptible to more serious respiratory infections. In addition, infants of women with a SARS-CoV-2-positive placenta rarely manifest the disease, as later described. How the placenta enacts this protective effect on the fetus remains to be explained. Human receptor ACE2, which carries the virus into the host cells, is widely expressed in the placenta. In particular, ACE2 has been identified in the villi (syncytiothrophoblasts, cytotrophoblasts, vascular endothelium of the villi, smooth muscle of the primary villi), in the extra-villous trophoblasts, and in the decidua cells. Its expression increases along with the trimester of pregnancy, being able to transfer the virus transplacentally to the fetus much more during the later stage of gestation
[12]. This would justify the lack of reports describing negative outcomes of maternal infection in the early stages of gestation, although we cannot yet exclude that they exist. Serine-protease type 2 (TMPRSS2) also found a similar dynamic alteration. Furthermore, the co-expression of serine protease type 2 (TMPRSS2) can promote entry by stimulating the fusion of viral particles with the host cell membrane and the related viral replication. Similar to other infections due to RNA viruses in pregnancy, SARS-CoV-2 infection causes placental lesions with signs of fetal and/or maternal vascular malperfusion and signs of inflammation. However, almost all infants of infected mothers were found to be negative at birth by RT-PCR and asymptomatic. Furthermore, it has been observed that the viral load in the placenta can be at least twice that of maternal blood and nasopharynx, increasing the possible risk of intrapartum transmission
[13][14]. The theoretical risk of vertical transmission therefore has a biological plausibility and can occur both through the hematogenous route and, more rarely, by the ascending route
[15].
A great caution is required in interpreting these data, because a complete collection from all tissues of both mother and baby is not always possible at appropriate times; currently available data often derive from case reports or case series rather than from multicentric studies. Regarding the unfavorable fetal/neonatal outcomes of COVID-19 infection contracted during pregnancy and perinatal outcomes, only four cases of miscarriage or abortion (1.4%) were reported
[16]. No confirmed teratogenic effects were reported in cases where the infection was contracted early in gestation and the overall malformations’ rate in SARS-CoV-2-infected women was similar to that in uninfected women, according to data from a Spanish multicenter study
[17].
In conclusion, transplacental transmission is possible or more likely in the last weeks of pregnancy. Vivanti’s recent report appears to have demonstrated neonatal viraemia following placental infection: the placenta showed signs of acute and chronic intervillous inflammation consistent with the severe systemic maternal inflammatory state triggered by SARS-CoV-2 infection. Both maternal and neonatal blood samples were positive, and the neonate had symptoms similar to those of infected adults
[18][19].
3. Which Tests Are to Be Used in the Neonate?
To date, the molecular detection of SARS-CoV-2 is considered the gold standard test to diagnose COVID-19. The technique is PCR (RT-PCR), that consists in two PCRs in sequence: first a rt-PCR and, subsequently, a real-time PCR for the detection of viral RNA. The test displays the presence of genetic fragments of the virus both in asymptomatic/paucisymptomatic subjects and in symptomatic subjects but does not assess whether the virus is in the process of replication.
The molecular test is performed on mucus and respiratory secretions, collected with a nasopharyngeal swab through the nostril and/or pharynx. The molecular technique converts the viral RNA (+) genome into the complementary single-stranded DNA (cDNA) sequence, using reverse transcriptase. Real-time PCR is performed on the obtained cDNA, which simultaneously amplifies and quantifies the available DNA, while maintaining the relative concentration ratios of the viral RNA of SARS-CoV-2. For this reason, rt-real-time PCR is used in the diagnosis of COVID-19 (albeit with qualitative indications)
[20].
Currently, the molecular test on nasopharyngeal swab is considered the most reliable for diagnosis, even in the newborn (with very high sensitivity and specificity), if the sampling technique and the analytical process are correct. A single positive RT-PCR result in a respiratory sample from a newborn can have several meanings: indicating active viral replication, or the presence of viral fragments acquired during passage through the birth canal or from external environmental contact soon after birth, or surface contamination that does not necessarily result in neonatal infection, symptomatic or not
[21].
According to the before-mentioned WHO expert report, it appears that in the report of a universal nasopharyngeal RT-PCR screening for SARS-CoV-2, 2.2% (9/418) of neonates tested positive within 24 h of birth; of these, eight had mothers with negative RT-PCR on nasopharyngeal swab and without symptoms, and seven retested infants were negative on a second test
[22]. This finding illustrates how complex it is sometimes to correctly interpret a positive molecular test in neonates. A negative test at 24 h from birth must be repeated, even on samples other than the nasopharyngeal, such as bronchoalveolar lavage fluid when indicated, blood and feces.
All neonates born to mothers with suspected or confirmed COVID-19 infection should undergo diagnostic rt-RT-PCR test on the respiratory tract exudate (nasopharynx, oropharynx, nose) at 24 h of life, regardless of the presence of symptoms in both the newborn and in the mother. A test performed too early may be indeterminate, but the best time to test the newborn is still unclear
[22]. If the test is negative or indeterminate, it must be repeated at 48 h of life. Asymptomatic infants who can be discharged before 48 h after birth should be tested before discharge (at 24–48 h of age). There is no need to wait for the test result to discharge the neonate if he or she appears clinically stable.
Serological tests search for SARS-CoV-2-specific antibodies in serum. They should not be used for diagnostic purposes and do not replace the identification of viral genetic material from the nasopharyngeal swab. The use of serological tests aims to evaluate the progress of the infection in the individual and in the community. In the newborn, they are not indicated, and interpretation can be very difficult. In fact, during the second and third trimester of gestation, maternal immunoglobulin G (IgG) are transferred to the fetus through the placenta. The presence of IgG antibodies in the newborn at birth may simply reflect the transfer of maternal antibodies and does not allow the diagnosis of intrauterine infection. Maternal IgM and IgA do not cross the placenta unless there are lesions and are thought to represent the immune response of the fetus to intrauterine infection. However, the sensitivity and specificity of IgM tests vary and are usually lower and less reliable than the molecular test. Therefore, a positive serological test in a newborn at birth always requires confirmation with a molecular test. To date, the presence of positive SARS-CoV-2 IgM in a newborn between birth and 7 days of life suggests the suspicion of a fetal response to an intrauterine infection, while positive IgM after 7 days suggests an early intrapartum or postnatal infection
[22].
During COVID-19 infection, there are no specific laboratory data in neonates and little infants. Leukopenia and lymphocytopenia have frequently been observed, while C-reactive protein and procalcitonin are usually normal. Other anomalies observed were mild to medium thrombocytopenia, an increase in creatine phosphokinase, alkaline phosphatase, alanine aminotransferase and lactic dehydrogenase. Lung lesions are better seen with pulmonary axial computed tomography rather than radiography alone and consist mostly of multiple radiopaque nodular images, single or multiple foci of parenchymal consolidation mostly located at the periphery of the lung parenchyma
[23].
4. How to Treat SARS-CoV-2 Infection
The approach to SARS-CoV-2 infection depends on the patient’s symptoms. From the respiratory point of view, if respiratory support is required, the neonate born to a mother with suspected or confirmed SARS-CoV-2 infection should be managed with devices currently used in clinical practice, carefully limiting the generation of aerosols and the spreading of droplets exhaled during oxygen delivery and ventilation strategies. The risk of aerosol transmission from neonates with SARS-CoV-2 to healthcare workers during respiratory support may be low. However, further studies on viral dispersion with different-sized droplets and various types of neonatal respiratory support are needed
[24]. Therefore, given the still not fully known characteristics of the current pandemic, it is prudent that health workers wear complete personal protective equipment during respiratory assistance to the newborn. The elective intubation of newborns born to SARS-CoV-2-positive women is not recommended and patient management must be tailored to the needs. When assembling the mechanical ventilator, it may be useful to place a hydrophobic antiviral/antibacterial filter on the expiratory branch of the circuit, paying attention to the effect of the increase in dead space in extremely preterm infants.
From the pharmacological point of view, most clinical trials did not include pregnant women with ongoing infection, although the safety of various drugs (i.e., lopinavir-ritonavir and remdesivir) during pregnancy has already been evaluated previously
[25]. In neonatal age, no therapy is usually required in asymptomatic cases, while supportive therapy and paracetamol are recommended in mild–moderate cases. In severe cases (clinical worsening, respiratory distress, sepsis, shock), it is possible to start intravenous remdesivir at the loaded dose of 5 mg/kg/per day iv (in 30 min) on the first day, and of 2.5 mg/kg/per day in the following nine days. Note that the most appropriate dosage of remdesivir in the first 2 weeks of life and for babies with a body weight lower than 2.5 kg has not yet been established
[26][27][28]. A higher-than-expected incidence of thromboembolic complications has been described when the clinical course of COVID-19 is severe. It may therefore be appropriate to start enoxaparin at a dose of 150–300 IU/kg/per day
[29][30]. Sometimes, the cytokine storm in progress leads to COVID-19-associated multisystem inflammatory (MIS-C). In such a situation, it could be appropriate to start the immunomodulatory therapy using IV immunoglobulins (400 mg/kg/day for 5 days or 1 g/kg/day for 2 days) or monoclonal (anakinra or tocilizumab)
[31][32][33].
5. Breastfeeding and SARS-CoV-2 Infection
SARS-CoV-2 virus has not been detected in human milk samples in most reports
[34][35]. Although in the detection of SARS-CoV-2 RNA in some human milk samples, the risk of the milk contamination by maternal respiratory secretions could not be ruled out, as declared by the same authors
[36]. Breastfeeding seemed to be not associated with SARS-CoV-2 infection, revealing that viral transmission through milk, if any, is very rare. A particular caution should be acknowledged when analyzing these results, considering the method of milk collection, sample handling and storage, validation of used assays, viral quantification and viability
[35]. The presence of viral particles does not imply the possibility of transmitting the infection. Conversely, the benefits of breastfeeding concern the transfer of specific antibodies to the neonates, the quality of nutrition and prevention of obesity in late life, the development of the infant immunity by immune-stimulating factors and microorganisms colonizing the gastrointestinal tract
[37].
Therefore, the current indications of all the Italian and foreign scientific societies and of the CDC invite mothers with suspected or confirmed COVID-19, if in good clinical condition, to breastfeed their babies, adequately applying all the correct hygiene rules (precautions from contact and droplets)
[38][39]. Both parents must be informed of the remote possibility of transmission and an informed consent should be signed.
It is clear to date that milk produced by infected mothers is a source of anti-SARS-CoV-2 IgA and IgG and can neutralize SARS-CoV-2 activity, supporting the recommendations to continue breastfeeding although mild-to-moderate COVID-19 illness
[40].
6. How to Rethink In-Hospital Management of Mother and Neonate
If the pregnant woman has a suspected or confirmed SARS-CoV-2 infection, a dedicated path must be provided within the hospital structure, as recommended
[41][42]. Triage must be organized to separate infected patients from non-infected patients, placing them in dedicated rooms, where it is possible to assess them safely. Dedicated healthcare workers must be equipped with all the necessary personal protective equipment (PPE: gloves, surgical mask or FFP2 or FFP3, eye protection, disposable gown, overshoes). The patient must wear the mask from the moment of entry into the hospital. The birth must take place in a dedicated delivery room, with a multidisciplinary assistance team and with a minimum number of operators. The assistance and monitoring protocol is accorded to the routinary clinical practice of the unit, adapted to the clinical conditions of the pregnant woman, regarding caesarean section, continuous cardiotocography monitoring and steroid prophylaxis for fetal lung development. In symptomatic women, water birth is contraindicated. After the birth, the newborn is managed in the resuscitation unit, in the delivery room, at a distance of at least two meters from the mother, or in a dedicated room, next to the delivery room. Delayed cord clamping can be performed on a routine basis, depending on the condition of the newborn. Skin-to-skin contact, including kangaroo mother care for preterm or low birth weight infants, is recommended if the clinical conditions of the infant and mother allow it
[41][42].
When transferred to the NICU, the infant should be placed in a closed transport incubator, with immediate disinfection immediately after the transport. The assistance in the unit must be provided in an area isolated from other patients, within which the medical and nursing staff act following all the needed precautions, minimizing any contact with the patient. By following the appropriate preventive measures, the parents’ entry to the unit must be guaranteed. After delivery, when the baby and mother leave the delivery room, all devices must be removed and placed in closed plastic bags, with adequate disinfection of the environment and equipment. If the clinical condition of the newborn is stable and the mother is paucisymptomatic, she can independently manage the newborn in rooming-in and they must be managed together, pending the results of any tests performed. If the mother’s test is positive, rooming-in in a dedicated and isolated room can be performed
[43].
Infants born to SARS-CoV-2-positive mothers or infants whose mother’s test was pending should be considered persons under investigation (PUIs), even if PCR resulted negative at 24 h of life. Considering the incubation period (range, 2–14 days; mean 4–5 days), they should be retested to rule out the perinatal transmission of SARS-CoV-2 (due to contact with maternal secretions), before their status is resolved. The duration of transmission precautions has to be discussed on a case-by-case basis with an infection prevention and control team
[44].
Mother–newborn separation should occur only in the case in which the conditions of the mother are severe, especially from the respiratory point of view. It is recommended to directly breastfeed the infant at the mother’s breast if the mother is in stable clinical conditions or to express milk if the mother cannot breastfeed the infant due to compromised clinical conditions. The following PPE is recommended for operators assisting the mother and the newborn: water-repellent surgical masks and (where available) FFP2, disposable gown, gloves and devices that allow eye protection, overshoes. When maneuvers that allow aerosol transmission are necessary (i.e., during intubation, if the patient is undergoing ventilatory assistance, during aerosol therapy, during bronchoscopy or during cardio-pulmonary resuscitation), healthcare workers should wear FFP2 and, where available, FFP3 masks
[45].
Given the highly contagious nature of SARS-CoV-2, additional precautions should be considered during the transport of patients with suspected or confirmed COVID-19. Dedicated pathways have to be carefully planned. A recent European consensus recommended transporting the infant without parents or relatives, regardless of if they are symptomatic or not, to reduce the risk for the transport team of becoming infected during the transport
[46].
Healthcare workers should assist the infant in a closed transport incubator, avoiding reopening the portholes, except in the case of resuscitation procedures. At the end of transport, any exposed equipment should be disposed (i.e., masks or self-inflating bag) or sterilized (i.e., laryngoscope) according to routine procedures
[45]. All procedures must always be performed wearing disposable PPE. The vehicle requires decontamination with a universal detergent (i.e., ethanol) followed by cleaning of the entire interior of the vehicle with a chlorine-based solution at 1000 parts per million and waste should be disposed of as infectious waste, as per local policy
[46].