
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Alireza Sadeghi Mahoonak | + 2874 word(s) | 2874 | 2021-05-11 06:06:48 | | | |
| 2 | Vivi Li | Meta information modification | 2874 | 2021-05-28 03:52:15 | | |
Video Upload Options
The orange seed is an important by-product obtained from the juice production industry. The juice industry considers seed as a by-product that contains between 17.9% to 26.5% of protein [18]. In fact, the protein obtained from defatted flour can be used as a low-cost source for the generation of peptides through hydrolysis that could have an important role as antioxidant, anticancer, antidiabetic, or antihypertensive peptides.
1. Introduction
In recent years, the importance of using bioactive compounds in diet and their role in reducing diseases and disorders due to improper nutrition has increased. Proteins and peptides are the most important bioactive compounds among natural sources [1]. When proteins and peptides have been hydrolyzed, they can exert an important biological role via antioxidant [2][3][4][5][6], antihypertensive [4][6][7][8], antidiabetic [9][10], or anticancer [2] activities. Therefore, numerous studies have been focused on the production of bioactive peptides. These studies include the production of peptides from royal jelly [11], orange seed flour [12], pumpkin seed [13], mung bean [14], rainbow trout skin [15], whey [16], or tomato seed [17]. Protein hydrolysates obtained from food industry by-products can be used as a rich source of bioactive peptides in nutritional supplements.
The orange seed is an important by-product obtained from the juice production industry. The juice industry considers seeds as a by-product that contains between 17.9% to 26.5% of protein [18]. In fact, the protein obtained from defatted flour can be used as a low-cost source for the generation of peptides through hydrolysis that could have an important role as antioxidant, anticancer, antidiabetic, or antihypertensive peptides.
The main objective of the present study was to hydrolyze orange seed proteins using the pepsin enzyme, a protease from porcine gastric mucosa, to obtain bioactive peptides showing the highest antioxidant, antihypertensive, and hypoglycemic capacity as well as to study the stability of such activity after simulated conditions of gastrointestinal digestion. After the separation of peptides using size-exclusion chromatography and RP-HPLC, the most active fractions were analyzed to identify the peptides using mass spectrometry in tandem before and after simulated gastrointestinal digestion.
2. Evaluation of Biological Activity of the Hydrolyzed Proteins
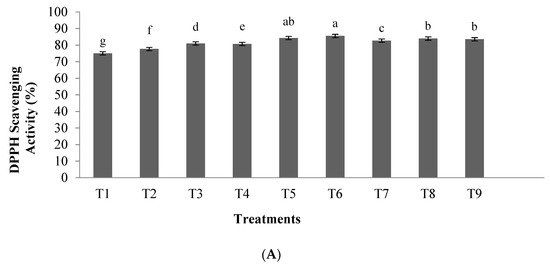
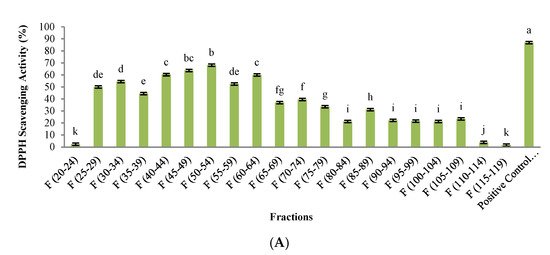
2.1. Antioxidant Activity
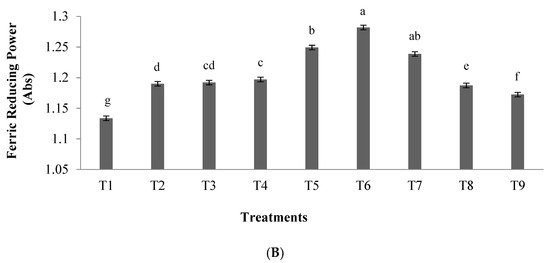
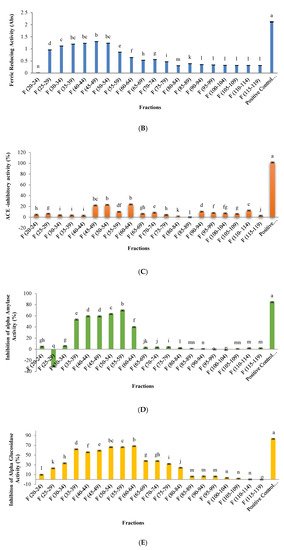
2.2. ACE-Inhibitory Activity
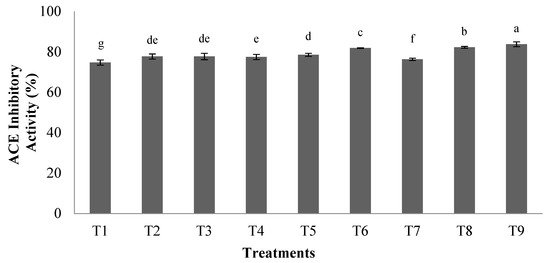
2.3. α-Amylase and α-Glucosidase Inhibitory Activity
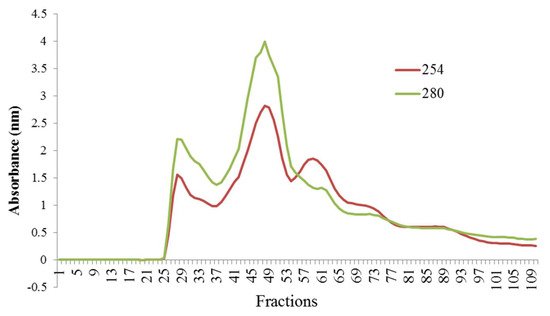
3. Fractionation of Hydrolyzed Orange Seed Proteins Using SEC
4. Evaluation of Biological Activity in Peptides Isolated from SEC
5. Discussion
Results showed that by increasing the hydrolysis time and the enzyme to substrate ratio, the inhibitory activity of ACE in the hydrolyzed samples increased. Furthermore, using an enzyme concentration of 0.03 and hydrolysis time of 3.5 h, the antioxidant activity and the inhibitory activity of α-amylase and α-glucosidase enzymes increased. These results are in agreement with previous studies of the antioxidant activity of hydrolyzed peanut proteins and tuna liver proteins, where similarly, by increasing hydrolysis time and concentration, an increase in the antioxidant activity was observed [19][20].
In another study, the highest ACE-inhibitory activity of 83.70% was obtained using an enzyme to substrate ratio of 0.03 and hydrolysis time of 5 h. In general, increasing hydrolysis time leads to the production of peptides smaller than 3 kDa, which could show higher ACE-inhibitory activity [21]. Je et al. [20] reported an ACEI-inhibitory activity in fish liver protein of 36% after hydrolysis using various enzymes such as Flavourzyme, Neutrase, Alcalase, and Protamex, which is lower than the activity observed in the present study (83.70%), probably due to the differences in matrices and types of protein. Possible mechanisms of action of peptides showing ACEI-inhibitory activity are (i) the peptides bind to the active site of the ACE enzyme, or (ii) they may bind to the inhibitory sites located on the ACEI enzyme. These binding alter the protein’s structure and prevent the binding of substrate (angiotensin) to the active site of the enzyme [22]. Moreover, the highest ACEI inhibition was associated with peptides with high hydrophobicity, because hydrophobic peptides show affinity to the active site of the ACEI enzyme [23]. Furthermore, differences in the ACEI-inhibitory activity between treatments were described as differences in the molecular weight and the sequence of amino acids in the peptides [24].
In this study, the highest antidiabetic potential was observed using an enzyme to substrate ratio of 0.03 with a hydrolysis time of 3.5 h. The results indicated that the highest inhibitory activity of α-amylase and α-glucosidase were 42.3% and 45.4%, respectively. Previous studies showed that the content in aromatic amino acids including phenylalanine, tryptophan, and tyrosine in the peptide chain are key factors in the ability to inhibit α-glucosidase or α-amylase enzymes [25][26]. Therefore, the high anti-diabetic capacity of hydrolyzed orange seed proteins could be due to the presence of aromatic amino acids in their structure.
In order to identify the main peptides responsible for the antioxidant, antihypertensive, and antidiabetic activities, sample T6 (0.03 E/S, 3.5 h) was analyzed using SEC. Regarding the observed results, Khantaphant et al. reported that differences in the molecular weight of the peptides’ profile may be related to differences in enzymes’ specific breakdown sites [27]. Thus, the active site of the pepsin enzyme contains thiol groups and breaks the bonds adjacent to the tryptophan, tyrosine, and phenylalanine amino acids [27].
SEC has been used as a common procedure for the peptides’ isolation before RP-HPLC [28][29]. The highest antioxidant activity in the peptide fractions isolated from SEC was in fractions with molecular weights lower than 13,000 Da, and the maximum inhibitory activity of ACEI enzyme was related to peptide fractions with molecular weight less than 13,000 Da. Other studies also implied that low-molecular-weight peptides show higher ACE-inhibitory activity [30][24][31]. In addition, it has been reported that peptides with high antidiabetic properties contain high amounts of hydrophobic amino acids [32], and they may inhibit α-amylase and α-glucosidase enzymes by binding to the active site of the enzyme through hydrophobic bonds [25]. The affinity of binding to the enzyme active site was enhanced by increasing the peptide bond breakdown points in a protein chain and producing shorter peptide chains [22]. The results showed that by increasing hydrophobicity, the amount of antioxidant activity also increased. Andersen et al. [33] suggested that the most powerful antioxidant compounds usually are strong reducing agents. This is probably due to the high amount of aromatic and hydrophobic amino acids in these peptide fractions. Several researchers documented that the antioxidant properties, ACEI-inhibitory activity, and antidiabetic potential have been closely associated with the ratio of hydrophilic to hydrophobic peptides [11][30][28][29][34].
During the separation of peptides using RP-HPLC, peptide fractions that were more retained in the C18 column showed the highest DPPH activity [35]. The identification of the peptides in this fractions confirmed the presence in the sequence of hydrophobic amino acids such as leucine, phenylalanine, valine, and tryptophan [35]. Power et al. [35] revealed that active antioxidant peptides mainly contain hydrophobic amino acids in their structure. Tryptophan also plays an important role in the inhibitory activity of DPPH through its hydrogenating role [11][17][35]. These results are similar to the results obtained after the hydrolysis of royal jelly [11] and tomato seeds [17]. In relation to the inhibitory activity of the ACE enzyme, fraction 32 showed the highest inhibitory activity in the obtained peptides. It has been suggested that polar groups present in hydrophilic peptides and non-polar or aromatic groups in hydrophobic peptides may bind to the active site or to the inhibitory site in ACE enzyme. These bindings would alter the spatial structure of the enzyme, preventing angiotensin from binding to the active site of ACE [23][36][37]. On the other hand, peptides with high hydrophilicity have less access to the ACE active site [38][23][24]. Meanwhile, the peptides of the primary chromatogram fractions appeared to be multifunctional peptides, and the reason for this can be related to the balanced content of polar groups of hydrophilic amino acids in the primary fractions [11][22]. The fractions 28 and 30 showed the maximum α-amylase and α-glucosidase enzyme inhibitory activities, respectively. Our outcomes indicated that there was a direct relationship between the hydrophobic content of bioactive peptides and their biological activity. The results of this study were consistent with the results obtained from the biological activity of bioactive peptides from hydrolysis of royal jelly [11] and tomato seeds [17].
In the current study, the bioavailability of peptide fractions was evaluated by simulated gastrointestinal digestion. The stability of the biological activity of peptides depends not only on the maintenance of their structure but also on other factors [39]. Peptide behavior in simulated gastrointestinal digestion may provide important information about their possible behavior in the digestive system. The changes in the peptides profile observed after HPLC separation indicated the important effect of gastrointestinal enzymes on the sequences. Moreover, acidic conditions with pH 2 and increasing to pH 7 could cause changes in peptide sequences. Marambe et al. (2011) demonstrated that acidic treatment reduced some peaks of high-molecular-weight peptides in the chromatogram and increased other peptides of 1 kDa and less [40]. Moreover, Meshginfar et al. reported that the neutral polar charge of ionizable amino acid group in the sequence of peptides was very effective on the stability of peptides in digestive conditions [17]. They also indicated that digestion in antioxidant peptides causes minimal changes in their biological activity [17]. Thus, it seems that the hydrolyzed orange seed protein peptide fractions may retain their biological activity after the gastrointestinal digestion stage.
The fraction 31–32 obtained from the last purification step (RP-HPLC) was applied for the identification of the peptides using nano-LC-MS/MS. A total of 952 sequences were identified in samples before gastrointestinal digestion in comparison with 26 peptide fractions that were identified after gastrointestinal digestion. This decrease in the amount of peptide sequences identified could be due to the intense hydrolysis occurring during gastrointestinal digestion that results in the generation of very small peptides that are out of the analysis range in the mass spectrometer. Selma et al. (2018) suggested that the highest antidiabetic and antihypertensive activity of peptides detected from protein hydrolysates from Octopus vulgaris by nano-LC-MS/MS after isolation with RP-HPLC were related to fractions with a molecular mass of about 400–2500 Da and mainly contain the hydrophobic amino acids [32]. Another study on the antioxidant activity of bioactive peptides from the porcine plasma protein hydrolysis concluded that the DPPH scavenging activity and Fe3+ ion reduction power were the highest in molecules that weigh less than 3 kDa [41]. Torkova et al. (2016) [42] studied the hydrolyzed peptides of poultry protein and the effect of in vitro gastrointestinal digestion conditions on the bioactive activity of these peptides and showed that all peptides inhibiting the ACEI enzyme had 5 to 9 amino acids in their peptide chain [42]. Our outcomes were consistent with the results of previous studies including Maqsoudlou et al., Meshginfar et al., and Mazloomi et al. [11][17][43], and it was revealed that histidine, proline, serine, aspartic acid, and glutamic acid were the important amino acids in peptides with antioxidant properties. Histidine, proline, serine, glutamic acid, and tyrosine were also the important amino acids in peptides with ACEI-inhibitory properties. Ultimately, proline, serine, aspartic acid, and glutamic acid were also important amino acids in peptides with antidiabetic potential. Generally, the restriction on the number of detected peptides in the hydrolyzed pepsin and the presence of specific amino acids at the end of these peptide chains has been due to the presence of specific amino acids in the active site of the pepsin enzyme [20][27]. This was evident in the amino acid sequence of the detected pepsin-hydrolyzed peptides. Depending on their specific structure, some bioactive peptides can resist the protease enzymes’ action. For example, peptides containing proline at the end of the carboxyl group are more resistant to gastrointestinal digestion and are, therefore, not hydrolyzed by digestive enzymes [11]. On the other hand, due to their molecular weight, and hydrophobicity, some antihypertensive peptides are structurally resistant when passing through intestinal epithelial cells as well as digestive digestion [11]. Mazloomi et al. (2020) [43] showed that orange seeds can be used to produce beneficial bioactive peptides that are resistant to digestive enzymes, and Alcalase-hydrolyzed orange seed proteins can be also suggested as a health-beneficial product to reduce blood pressure and for diabetes management. The study on the peptide sequences after gastrointestinal digestion shows that these peptides may retain their biological activity in many cases and in some cases become new peptides with new health-beneficial characteristics.
References
- De Deckere, E.; Verschuren, P. Functional fats and spreads. In Functional Foods; Elsevier: Amsterdam, The Netherlands, 2000; pp. 233–257.
- Taha, F.S.; Mohamed, S.S.; Wagdy, S.M.; Mohamed, G.F. Antioxidant and antimicrobial activities of enzymatic hydrolysis products from sunflower protein isolate. World Appl. Sci. J. 2013, 21, 651–658.
- Selamassakul, O.; Laohakunjit, N.; Kerdchoechuen, O.; Yang, L.; Maier, C.S. Isolation and characterisation of antioxidative peptides from bromelain-hydrolysed brown rice protein by proteomic technique. Process Biochem. 2018, 70, 179–187.
- Jo, C.; Khan, F.F.; Khan, M.I. Marine bioactive peptides: Types, structures, and physiological functions. Food Rev. Int. 2016, 33, 44–61.
- De Domenico, S.; De Rinaldis, G.; Paulmery, M.; Piraino, S.; Leone, A. Barrel Jellyfish (Rhizostoma pulmo) as Source of Antioxidant Peptides. Mar. Drugs 2019, 17, 134.
- Ngo, D.H.; Kim, S.K. Marine Bioactive Peptides as Potential Antioxidants. Curr. Protein Pept. Sci. 2013, 14, 189–198.
- Szwajkowska, M.; Wolanciuk, A.; Barłowska, J.; Krol, J.; Litwińczuk, Z. Bovine milk proteins as the source of bioactive peptides influencing the consumers’ immune system—A review. Anim. Sci. Pap. Rep. 2011, 29, 269–280.
- Soleymanzadeh, N.; Mirdamadi, S.; Mirzaei, M.; Kianirad, M. Novel β-casein derived antioxidant and ACE-inhibitory active peptide from camel milk fermented by Leuconostoc lactis PTCC1899: Identification and molecular docking. Int. Dairy J. 2019, 97, 201–208.
- Chakrabarti, S.; Jahandideh, F.; Wu, J. Food-derived bioactive peptides on inflammation and oxidative stress. BioMed Res. Int. 2014, 2014, 608979.
- Yang, H.J.; Kwon, D.Y.; Kim, M.J.; Kang, S.; Park, S. Meju, unsalted soybeans fermented with Bacillus subtilis and Aspergilus oryzae, potentiates insulinotropic actions and improves hepatic insulin sensitivity in diabetic rats. Nutr. Metab. 2012, 9, 37.
- Maqsoudlou, A.; Mahoonak, A.S.; Mora, L.; Mohebodini, H.; Toldrá, F.; Ghorbani, M. Peptide identification in alcalase hydrolysated pollen and comparison of its bioactivity with royal jelly. Int. Food Res. J. 2019, 116, 905–915.
- Mazloomi, S.N.; Mahoonak, A.S.; Ghorbani, M.; Houshmand, G. Physicochemical properties of chitosan-coated nanoliposome loaded with orange seed protein hydrolysate. J. Food Eng. 2020, 280, 109976.
- Mazloomi-Kiyapey, S.N.; Sadeghi-Mahoonak, A.; Ranjbar-Nedamani, E.; Nourmohammadi, E. Production of antioxidant peptides through hydrolysis of medicinal pumpkin seed protein using pepsin enzyme and the evaluation of their functional and nutritional properties. ARYA Atheroscler. 2019, 15, 218.
- Xie, J.; Du, M.; Shen, M.; Wu, T.; Lin, L. Physico-chemical properties, antioxidant activities and angiotensin-I converting enzyme inhibitory of protein hydrolysates from Mung bean (Vigna radiate). Food Chem. 2019, 270, 243–250.
- Ramezanzade, L.; Hosseini, S.F.; Nikkhah, M. Biopolymer-coated nanoliposomes as carriers of rainbow trout skin-derived antioxidant peptides. Food Chem. 2017, 234, 220–229.
- Mohan, A.; Rajendran, S.R.; Thibodeau, J.; Bazinet, L.; Udenigwe, C.C. Liposome encapsulation of anionic and cationic whey peptides: Influence of peptide net charge on properties of the nanovesicles. LWT 2018, 87, 40–46.
- Meshginfar, N.; Mahoonak, A.S.; Hosseinian, F.; Tsopmo, A. Physicochemical, antioxidant, calcium binding, and angiotensin converting enzyme inhibitory properties of hydrolyzed tomato seed proteins. J. Food Biochem. 2019, 43, e12721.
- Mohamed, B.; El-Shenawi, M. Functional properties and In-vitro digestibility of bitter orange (Citrus aurantium) seed flour. Merit Res. J. Agric. Sci. Soil Sci. 2013, 1, 47.
- Jamdar, S.; Rajalakshmi, V.; Pednekar, M.; Juan, F.; Yardi, V.; Sharma, A. Influence of degree of hydrolysis on functional properties, antioxidant activity and ACE inhibitory activity of peanut protein hydrolysate. Food Chem. 2010, 121, 178–184.
- Je, J.-Y.; Lee, K.-H.; Lee, M.H.; Ahn, C.-B. Antioxidant and antihypertensive protein hydrolysates produced from tuna liver by enzymatic hydrolysis. Int. Food Res. J. 2009, 42, 1266–1272.
- Coscueta, E.R.; Amorim, M.M.; Voss, G.B.; Nerli, B.B.; Picó, G.A.; Pintado, M.E. Bioactive properties of peptides obtained from Argentinian defatted soy flour protein by Corolase PP hydrolysis. Food Chem. 2016, 198, 36–44.
- Mora, L.; Aristoy, M.-C.; Toldrá, F. Bioactive peptides in foods. In Encyclopedia of Food and Health; Caballero, B., Finglas, P.M., Toldrá, F., Eds.; Academic Press: Cambridge, MA, USA, 2016; pp. 395–400.
- Ktari, N.; Nasri, R.; Mnafgui, K.; Hamden, K.; Belguith, O.; Boudaouara, T.; El Feki, A.; Nasri, M. Antioxidative and ACE inhibitory activities of protein hydrolysates from zebra blenny (Salaria basilisca) in alloxan-induced diabetic rats. Process. Biochem. 2014, 49, 890–897.
- Nasri, R.; Younes, I.; Jridi, M.; Trigui, M.; Bougatef, A.; Nedjar-Arroume, N.; Dhulster, P.; Nasri, M.; Karra-Châabouni, M. ACE inhibitory and antioxidative activities of Goby (Zosterissessor ophiocephalus) fish protein hydrolysates: Effect on meat lipid oxidation. Int. Food Res. J. 2013, 54, 552–561.
- Lacroix, I.M.; Li-Chan, E.C. Inhibition of dipeptidyl peptidase (DPP)-IV and α-glucosidase activities by pepsin-treated whey proteins. J. Agric. Food Chem. 2013, 61, 7500–7506.
- Siow, H.-L.; Lim, T.S.; Gan, C.-Y. Development of a workflow for screening and identification of α-amylase inhibitory peptides from food source using an integrated Bioinformatics-phage display approach: Case study–Cumin seed. Food Chem. 2017, 214, 67–76.
- Khantaphant, S.; Benjakul, S.; Ghomi, M.R. The effects of pretreatments on antioxidative activities of protein hydrolysate from the muscle of brownstripe red snapper (Lutjanus vitta). LWT Food Sci Technol. 2011, 44, 1139–1148.
- Jemil, I.; Abdelhedi, O.; Mora, L.; Nasri, R.; Aristoy, M.-C.; Jridi, M.; Hajji, M.; Toldrá, F.; Nasri, M. Peptidomic analysis of bioactive peptides in zebra blenny (Salaria basilisca) muscle protein hydrolysate exhibiting antimicrobial activity obtained by fermentation with Bacillus mojavensis A21. Process. Biochem. 2016, 51, 2186–2197.
- Lassoued, I.; Mora, L.; Nasri, R.; Aydi, M.; Toldrá, F.; Aristoy, M.-C.; Barkia, A.; Nasri, M. Characterization, antioxidative and ACE inhibitory properties of hydrolysates obtained from thornback ray (Raja clavata) muscle. J. Proteom. 2015, 128, 458–468.
- Lassoued, I.; Mora, L.; Barkia, A.; Aristoy, M.-C.; Nasri, M.; Toldrá, F. Bioactive peptides identified in thornback ray skin’s gelatin hydrolysates by proteases from Bacillus subtilis and Bacillus amyloliquefaciens. J. Proteom. 2015, 128, 8–17.
- Chen, N.; Yang, H.; Sun, Y.; Niu, J.; Liu, S. Purification and identification of antioxidant peptides from walnut (Juglans regia L.) protein hydrolysates. Peptides 2012, 38, 344–349.
- Salem, R.B.S.-B.; Ktari, N.; Bkhairia, I.; Nasri, R.; Mora, L.; Kallel, R.; Hamdi, S.; Jamoussi, K.; Boudaouara, T.; El-Feki, A. In vitro and in vivo anti-diabetic and anti-hyperlipidemic effects of protein hydrolysates from Octopus vulgaris in alloxanic rats. Int. Food Res. J. 2018, 106, 952–963.
- Andersen, M.L.; Lauridsen, R.K.; Skibsted, L.H. Optimising the Use of Phenolic Compounds in Foods; Woodhead Publishing Ltd.: Cambridge, UK, 2003.
- Moayedi, A.; Mora, L.; Aristoy, M.-C.; Hashemi, M.; Safari, M.; Toldrá, F. ACE-inhibitory and antioxidant activities of peptide fragments obtained from tomato processing by-products fermented using Bacillus subtilis: Effect of amino acid composition and peptides molecular mass distribution. Appl. Biochem. Biotechnol. 2017, 181, 48–64.
- Power, O.; Jakeman, P.; FitzGerald, R. Antioxidative peptides: Enzymatic production, in vitro and in vivo antioxidant activity and potential applications of milk-derived antioxidative peptides. Amino Acids 2013, 44, 797–820.
- Lee, S.-H.; Qian, Z.-J.; Kim, S.-K. A novel angiotensin I converting enzyme inhibitory peptide from tuna frame protein hydrolysate and its antihypertensive effect in spontaneously hypertensive rats. Food Chem. 2010, 118, 96–102.
- Wijesekara, I.; Kim, S.-K. Angiotensin-I-converting enzyme (ACE) inhibitors from marine resources: Prospects in the pharmaceutical industry. Mar. Drugs 2010, 8, 1080–1093.
- Lassoued, I.; Mora, L.; Barkia, A.; Aristoy, M.C.; Nasri, M.; Toldrá, F. Angiotensin I-converting enzyme inhibitory peptides FQPSF and LKYPI identified in Bacillus subtilis A26 hydrolysate of thornback ray muscle. Int. J. Food Sci. Technol. 2016, 51, 1604–1609.
- Megías, C.; Yust, M.d.M.; Pedroche, J.; Lquari, H.; Girón-Calle, J.; Alaiz, M.; Millán, F.; Vioque, J. Purification of an ACE inhibitory peptide after hydrolysis of sunflower (Helianthus annuus L.) protein isolates. J. Agric. Food Chem. 2004, 52, 1928–1932.
- Marambe, H.K.; Shand, P.J.; Wanasundara, J.P. Release of angiotensin I-converting enzyme inhibitory peptides from flaxseed (Linum usitatissimum L.) protein under simulated gastrointestinal digestion. J. Agric. Food Chem. 2011, 59, 9596–9604.
- Liu, R.; Zheng, W.; Li, J.; Wang, L.; Wu, H.; Wang, X.; Shi, L. Rapid identification of bioactive peptides with antioxidant activity from the enzymatic hydrolysate of Mactra veneriformis by UHPLC–Q-TOF mass spectrometry. Food Chem. 2015, 167, 484–489.
- Torkova, A.; Kononikhin, A.; Bugrova, A.; Khotchenkov, V.; Tsentalovich, M.; Medvedeva, U. Effect of in vitro Gastrointestinal Digestion on Bioactivity of Poultry Protein Hydrolysate. Curr. Res. Nutr. Food Sci. 2016, 4, 77–86.
- Mazloomi, S.N.; Mora, L.; Aristoy, M.; Sadeghi Mahoonak, A.; Ghorbani, M.; Houshmand, G.H.; Toldrá, F. Impact of Simulated Gastrointestinal Digestion on the Biological Activity of an Alcalase Hydrolysate of Orange Seed (Siavaraze, Citrus sinensis) by-Products. Foods 2020, 9, 1217.




