The integration of genetic and molecular components of a disease produces interacting networks, modules and phenotypes with clinical applications in complex cardiovascular entities. With the holistic principle of systems biology, some of the features of complexity and natural progression of cardiac diseases are approached and explained. Two important interrelated holistic concepts of systems biology are described; the emerging field of personalized medicine and the constraint-based thinking with downward causation. Constraints in cardiovascular diseases embrace three scientific fields related to clinical cardiology: biological and medical constraints; constraints due to limitations of current technology; and constraints of general resources for better medical coverage. Systems healthcare and personalized medicine are connected to the related scientific fields of: ethics and legal status; data integration; taxonomic revisions; policy decisions; and organization of human genomic data.
1. Introduction
The term “constraints” was described in biology in the context of natural selection and organism survival. Biological networks are constrained by a variety of factors such as the biological, environmental and physicochemical. Biological constraints could be self-imposed and produced by regulatory networks while hard constraints are imposed by other factors (e.g., environment)
[1]. Constraint-based analysis methods are being used to study genome-scale models and the biological properties of whole organisms. The constraint-based concept is used widely in systems biology (SB) from genomes to clinical phenotypes, and it is related to personalized medicine and clinical guidelines having an impact on current clinical practice and diseases. Cardiologists following patients with complex cardiovascular diseases (CVDs), such as coronary artery disease (CAD) and heart failure (HF), are complying with current clinical practice, but they are also familiarized with the clinical constraints hypothesis incorporated in medical guidelines directions.
Traditional healthcare systems are using a reductionist approach explaining and managing complex diseases as they reduce—through a very simple manner—the medical problem to an isolated organ-problem or biochemical fault
[2]. In traditional medicine the term “disease” designates that specific molecular systems or organs of the human body are unnaturally functioning and, therefore, subvert human health
[3]. With the reductionist approach significant advances in diagnosis and therapy of CAD were applied in everyday clinical practice, with a decrease of cardiac events and symptoms and an increase in longevity. Despite these successes in clinical management, complex CVDs continue to be the leading cause of mortality and morbidity, while chronic progression of the atherosclerotic process continues. Over the past few years it has become obvious that medical issues from molecular to clinical reasoning necessitate a new scientific approach requiring cooperation between medicine and interrelated sciences. Medicine cannot be considered in isolation from other systemic sciences, and a holistic approach is needed for complex diseases. Chronic complex systemic medical problems should be addressed with the holistic approach as it needs interdisciplinary integration and study of dynamical interactions between organs’ complex networks involving genetic, epigenetic and environmental factors. Many of the medical issues are interconnected with other systemic sciences including SB. The SB approach should be regarded as the science that combines biology with physics, mathematics, medicine and many other sciences like ecology and sociology. Systems healthcare (systems medicine) is the holistic approach to health based on the holistic principle of SB and to current clinical medical practice. Systems healthcare integrates data from molecules to phenotypes and from societies to environment, extending to the disciplines of economics, ethics and law
[4].
2. Constraints in Medicine and Clinical Cardiology
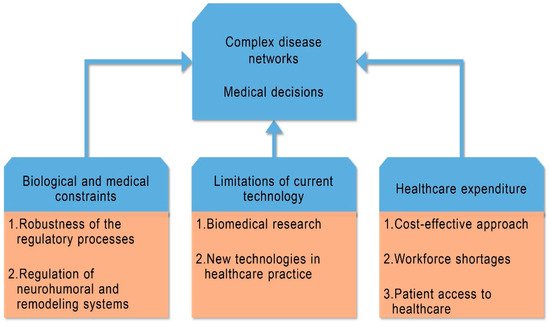
The application of constraints in medicine and clinical cardiology presumes that the constraint-based hypothesis, as it is implemented in biological and medical domains, should reflect a systems’ robustness. Also, the concept of constraints should be considered as a regulatory mechanism not only for molecular or other biological networks but also it is a decisive factor for medical decisions. Based on these assumptions the application of constraints encompasses three fields related to clinical cardiology: biological and medical constraints applied to cardiovascular diseases; constraints due to limitations of current technology; constraints of general resources for better medical coverage ().
Table 1. Constraints and personalized medicine in cardiovascular diseases.
| Constraints |
Personalized Medicine |
Biological and medical constraints
Limitations of current technology
Healthcare expenditure |
Ethics and legal status |
| Data integration |
| Taxonomic revision |
| Policy decisions |
| Organization of human genomic data |
2.1. Biological and Medical Constraints
The constraint-based thinking constitutes a holistic concept with downward causation interpreting many complex features of biology and diseases. In biology the term “constraints” is referred to antagonistic biological processes addressed to every evolutionary change in order to strengthen natural selection. Also, the constraint-based reasoning was applied to experimental biology to strengthen robustness of some experimental biological models
[5].
Green and Jones
[6] believe that the constraint-based interpretation of biological entities differs from mechanistic thinking of “change-relating causal features”, while the “constraint-based explanations emphasize formal dependencies and generic organizational features that are relatively independent of lower-level changes in causal details”. Furthermore, SB evaluates functional properties of living organisms and explores structured biological entities of genetic regulatory and metabolic networks, and the dynamics of enlarged networks in the form of modules (discrete functional regulatory networks) and phenotypes
[7]. It seems that the term “constraints” implies the presence of scale-dependency and close connection between biological systems functioning at different levels with strong downward causation (top-down effect). The top-down direction is crucial at first for information extraction from the lower levels and secondly for the capacity of the higher level to enforce constraints to lower levels with decreased degrees of freedom. The downward causation is interpreted as a regulatory constraining process that modifies and, in the end, determines behavior of lower level variables. The applied constraints limit some behaviors at the lower stage and simultaneously allow or “authorize” alternative behaviors to be released
[8][9][10].
For example, the boundary of cardiac cell geometrical structure generates both cellular membrane potential and cardiac rhythm. The imposed constraints of cellular membrane boundary structure and the triggered cardiac action potential through downward causation are responsible for the appearance and maintenance of cardiac rhythm
[9]. The above example indicates the limits of the reductionist position as cellular membrane, action potential and cardiac rhythm do not related directly to the genetic scale. The whole cellular membrane construction and function belong to a higher level of cardiac cellular construction.
Multileveled complex CVDs progression can be translated as a staged (leveled) structure with downward causation and constraints application from the top (phenotype) to the lower step (genome) of the disease. The top-down constraints’ application actually represents downward causation “imposing” some “biological behavior” or “order” to the lower level of the disease (pathological, diagnostic or therapeutic). The concept of constraint-based reasoning is proposed as a significant scientific tool for cardiovascular questioning and clinical research organization. Implementation of constraints in clinical cardiology has an impact to explain some properties of disease complexity and, also, to elucidate the unrelenting progression towards final disease stages. In fact, medical guidelines are founded on the constraint-based concept having downward causation. The cardiologist has decreased degrees of freedom, as only specific diagnostic tests and therapeutic procedures are available. In a way, the cardiologist “selects” the appropriate methodology according to current clinical guidelines using diagnostic and therapeutic constraints in a downward direction. In the realm of SB thinking, the applied constraints increase robustness of the regulatory processes for the stability of the unsteady metabolic networks and, also, for the variability of clinical complex entities
[11][12][13].
In this paper, it is proposed that the constraint-based thinking could be used not only as a concept for metabolic networks, but also as a fundamental clinical tool deciphering progressiveness of a disease’s clinical course. Both cardiac atherosclerotic process and CVDs are considered complex entities that follow a downward direction and causation in pathogenesis or in clinical management
[14]. For example, the size of a myocardial infarction and particularly the location and the importance of the myocardial area involved would induce a post-infarction myocardial dysfunction alongside of some compensatory mechanisms such as myocardial remodeling and growth of local coronary collaterals. According to SB approach, the size of myocardial infarction (phenotype) will impose constraints with downward direction on the degree of myocardial compensation and will dictate the most suitable medical and/or coronary invasive therapy, percutaneous coronary intervention (PCI) or bypass surgery.
In another example, patients with cardiac ischemic high-risk features are recommended for clinically indicated PCI and treated with dual antiplatelet therapy (DAPT) in the post-PCI period according to 2017 DAPT guidelines
[15]. Some patients demonstrated an increased number of ischemic and bleeding episodes following PCI which influenced the decision for intensity and duration of the DAPT in the post-PCI period. Therefore, pre-PCI constraints should be used in some of these patients with high-risk features if the invasive procedure is unavoidable or the DAPT regimen should be revised. A more personal approach with constraints in medical decisions and DAPT application is obligatory to some of these patients.
Human HF is a complex cardiac disease characterized by chronic clinical progression that involves participation of intrinsic compensatory or regulatory mechanisms
[7]. The SB methodology, to unravel potential causes of HF progression from early stages of myocardial dysfunction to more advanced phases of myocardial failure, integrates genes, epigenetic mechanisms and molecules, deciphers molecular networks or modular functional elements, and clarifies the interconnection of myocardial mechanical dysfunction with cardiac remodeling and other compensatory mechanisms
[16]. Heart failure should be addressed as a biological complex entity that is unstable, adaptive and self organized through its regulatory mechanisms ().
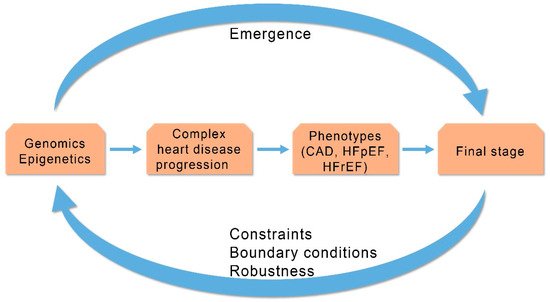
figure 1. Progression of complex heart diseases: Relationship between emergent properties and constraints outlines progression of complex heart diseases. CAD (Coronary Artery Disease), HFpEF (Heart Failure with Preserved Ejection Fraction), HFrEF (Heart Failure with Reduced Ejection Fraction). (Revised from:
[11]).
The regulatory mechanisms incorporate neurohumoral and remodeling systems that intend to compensate failing myocardium and change the unstable clinical equilibrium to a more stable clinical equilibrium status. The size of myocardial dysfunction in HF patients is related to the degree of compensation by the regulatory mechanisms. Thus, activation of the compensatory mechanisms (neurohumoral and remodeling) represents a constraint-based control (downward causation) of the degree of the compensation from the higher level (HF phenotype) to lower level (regulatory mechanisms). However, the activation of the regulatory mechanisms is often the cause of unwanted effects (symptoms) and clinical deterioration needing specific treatment to improve clinical status. The degree of clinical deterioration (by the regulatory mechanisms) in each stage of a disease’s progression dictates appropriate personalized treatment. Information and communication technologies could help through collection of related clinical data mined from published papers to improve management of HF patients. This could be achieved by the identification of the related biological networks that connect data in each level of HF progression and, after monitoring, the activated compensatory regulatory mechanisms from genome to clinical phenotypes can be clarified ().
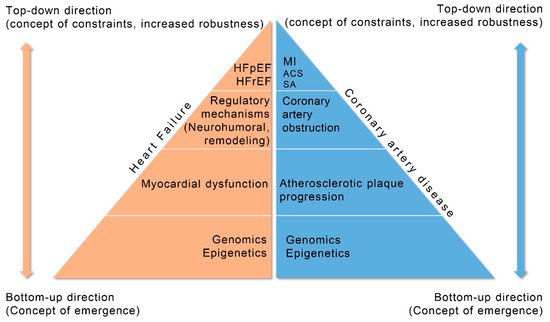
figure 2. Progression of coronary artery disease (CAD) and heart failure (HF) explained by systems biology approach. HFpEF (Heart Failure with Preserved Ejection Fraction), HFrEF (Heart Failure with Reduced Ejection Fraction), MI (Myocardial Infarction), ACS (Acute Coronary Syndromes), SA (Stable Angina).
2.2. Constraints Due to Limitations of Current Technology
In clinical practice, constraints may arise for diagnostic and therapeutic interventions due to limited facilities of current technological status.
New technologies require “cost effectiveness studies in the presence of health care input constraints” and crucial adjustments of conventional incremental cost effectiveness ratios (ICERs), because “without such adjustments the cost effectiveness analysis might lead to health losses”
[17] ().
figure 3. Constraints in three fields related to clinical cardiology; they behave as regulatory mechanisms for complex disease networks and medical decisions.
For example, novel techniques based on computer simulation, a process of mathematical modeling, are designed to predict atherosclerotic changes in coronary arteries. Such techniques are: positron emission tomography (PET), with the use of 18F-fluorodeoxyglucose, can label metabolically active areas in the myocardium and arteries; magnetic resonance imaging (MRI) can produce a molecular imaging of the cardiovascular system; cardiac computed tomography (CT) or CT angiography can be used for coronary calcium scoring and to measure subclinical or asymptomatic obstructive CAD; optical coherence tomography (OCT) for atherosclerotic plaque composition and stability.
In coronary artery areas with bifurcations or curbs there seems to exist a strong connection between concentrations of circulating plasma low density lipoprotein and turbulent flow with the development of atherosclerosis. Digitized images of coronary arterial post-mortem segments were analyzed with a computational fluid dynamic analysis, and the critical role of the local low wall static pressure was underlined for coronary wall thickening as a precipitating factor in the pathogenesis of coronary atherosclerosis
[18]. Another example of new technology is the Heart Flow Analysis, a system based on cloud services that offers non-invasively detailed information of coronary arteries and is used instead of an invasive cardiac procedure. The Heart Flow Analysis is scheduled to support the functional evaluation of CAD. It produces a personalized 3D model of coronary arteries using computed tomography (CT) images constructing a fluid dynamic model of the coronary blood flow. It identifies and calculates the size of coronary obstruction and advises cardiologists for further management. All new technologies have a positive cost-effective value for health care systems as they diminish current constraints for medical risks, and also reduce the high cost on health spending.
Digital health technologies aim to increase health care decision-making and improve health management. They represent actually “a broad spectrum of measurement technologies that include personal wearable devices and internal devices as well as sensors…but the current state of technology development and deployment requires… a cautionary note”
[19]. Also, digital health technology can “identify health risks and assist with diagnosis, treatment, and monitoring of health and disease conditions”
[20]. To some of the patients, digital health applications can offer new diagnoses and chances for novel treatment, but worldwide use of new digital health devices will need clinical trials to prove their usefulness
[20]. Therefore, the existent constraints for the full value of digital health technologies will be retracted when the new technologies become standardized and interoperable during clinical trials, and when new clinical guidelines have incorporated digital health devices
[19].
Gaveikaite et al.
[21] suggest that telehealth services can increase the “quality of health services for chronic obstructive pulmonary disease (COPD) management” and in those patients “complex interactions between multiple variables influence the adoption of telehealth services” for COPD by different healthcare professionals. Moreover, some constraints remain as “key variables were identified that require attention to ensure success of telehealth services” but “there is no consensus where self-management services should be positioned in the COPD care pathway”
[21]. Thus, medical practitioners or researchers from other fields, such as pulmonary diseases’ practitioners, computer science and network science researchers, can contribute their expertise to a common cause to explore further complex and interrelated human disease conditions.
Significant constraints are raised when the genetic base of chronic atherosclerotic disease is explored with modern genetic technologies. In complex cardiovascular atherosclerotic disorder the importance of genetics is elusive, as the disease is multifaceted and is not explained by single-gene mutations. In reality, the diverse phenotypes of CAD represent integrated clinical wholes with clinical behavior continuously changing due to the progressive nature of atherosclerotic process. Current understanding of genetics and genomics, as well as genome-wide association studies (GWAS), are inadequate alone to explain the natural course of cardiac atherosclerosis. It seems that “the genetic risk variants of atherosclerosis are activated concurrently with functionally active specific environmental risk factors” and that cardiac atherosclerosis could be studied only as a unified complex entity
[14]. The GWAS approach was expected to trace statistically significant interrelationships between single nucleotide polymorphisms (SNPs) and atherosclerosis. It was expected that SNPs related to atherosclerosis to be more frequently present in CAD patients than in control individuals, but, in contrast, a variety of genomic DNA markers were detected in individuals without CAD. Also, it was found that genomic technologies as SNP array, gene expression microarray and micro-RNA array were unable to demonstrate accurately the genetic atherosclerotic profile of CAD people. With the GWAS approach, it was found that only 10.6% of individuals with atherosclerosis possessed a probable heritable genetic factor. More important was that large-scale association analysis identified new risk loci for CAD, and that DNA methylation-mediated epigenetic downregulations and histone modifications triggered by lifestyle features play a vital role in atherosclerosis
[22][23][24]. The GWAS approach, based only on genetic variability, does not identify and clarify the vessel wall pathological changes or the clinical progressive nature of CAD phenotypes
[12].
The World Health Organization determines that a biomarker is “any substance, structure, or process that can be measured in the body or its products and influence or predict the incidence of outcome or disease”
[25]. The role of genetic biomarkers is limited and the genetic variation is of uncertain significance in clinical “whole exome sequencing” (WES), requiring continuous revision when clinical interpretation is demanded
[26][27]. Moreover, WES technologies are evolving using new diagnostic tools and medical clinicians and laboratory scientists can increase further their knowledge for complex diseases and tailor unique therapies for individual patients. Timmerman
[26] stresses the importance of standardization in laboratory research and argues that “the match between phenotype and genotype is circumscribed by the team’s reliance on specific standards”. As an example, he describes how a “clinical exome sequencing team” elects the time when to trust standards and a clinical exome sequencing technology will make “the transition from a laboratory research tool to a routine clinical technique used to diagnose patients”.
Emerging technologies are developing for “multi-omics studies and an increasing shift toward proteomics-going straight to the heart of biology that represents actual disease state and progression” and “to gain insight into the pathophysiology of disease and to identify proteins that are causally associated with disease, providing new targets for effective drug development”
[28]. Regardless of the advances in metabolomic methodologies that succeeded to produce thousands of molecules or biomarkers—some of those related to cardiology—many more are needed to give a new description of clinical phenotypes. It seems that more imaginative holistic approaches and new methodologies are needed to define novel clinical cardiac phenotypes. New multimodal systems of “omics”, metabolic pathways, environmental impacts and sophisticated disease-related networks are required to be integrated and provide a new holistic and realistic picture.
2.3. Constraints on Healthcare Budget
Health economic evaluations and the results of cost-effectiveness analyses studies are helpful for decision makers to confront the main economic constraint, the health care budget. In reality, besides the health care budget, there are “multiple other resource constraints that are involved relating, for instance, to health care inputs such as a shortage of skilled labor”
[17]. There are, also, other constraints involved, “consisting of supply-side (e.g., workforce shortages), demand-side (e.g., obstacles of access to healthcare) and healthcare system constraints (e.g., regulatory constraints)”
[17]. Complex CVDs swiftly increase their complexity changing pathology and course while clinical stages are overlapping. Moreover, alterations of the clinical course are approached differently in each phase of the disease. Clinical approach is modified in each step of the disease guided by current clinical, diagnostic and therapeutic constraints. Both, health system and practicing cardiologists are responsible for the wise use of the available resources and, also, to increase patients’ longevity. In view of the chronic and progressive course of the CVDs and in order to eliminate health disparities between underserved communities health authorities should define specific medical strategies and remove imposed constraints
[29].
The pre-hospital management strategy for patients with acute coronary syndromes (ACS) is a strong example from a medical or ethical point of view that requires coherent relations among patients and cardiologists. Patients with chest pain need immediate medical attention in an emergency cardiology department for further management that includes probable admission in acute coronary unit and PCI. However, there are worldwide limitations to further management of these patients due to restricted resources and insufficient organization (constraints). Patient’s transfer to the nearer medical center should be imperative following standard procedures and avoiding unnecessary bureaucratic retardations. Invasive procedures such as PCI and bypass surgery vary on their outcome from medical institution to another due to differences in expertise and resources. Thus, local medical circumstances can limit (constrain) medical decisions and practices diverging from current medical guidelines.