| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Elzbieta Kusmierek | + 1088 word(s) | 1088 | 2020-04-21 10:19:38 | | | |
| 2 | Bruce Ren | + 4 word(s) | 1092 | 2020-07-23 09:37:42 | | | | |
| 3 | Camila Xu | -2 word(s) | 1090 | 2020-11-01 09:45:44 | | |
Video Upload Options
Industrial sources of environmental pollution generate huge amounts of industrial wastewater containing various recalcitrant organic and inorganic pollutants that are hazardous to the environment. On the other hand, industrial wastewater can be regarded as a prospective source of fresh water, energy, and valuable raw materials. Conventional sewage treatment systems are often not efficient enough for the complete degradation of pollutants and they are characterized by high energy consumption. Moreover, the chemical energy that is stored in the wastewater is wasted. A solution to these problems is an application of photoelectrocatalytic treatment methods, especially when they are coupled with energy generation. The paper presents a general overview of the semiconductor materials applied as photoelectrodes in the treatment of various pollutants. The fundamentals of photoelectrocatalytic reactions and the mechanism of pollutants treatment as well as parameters affecting treatment processes are presented. Examples of different semiconductor photoelectrodes that are applied in treatment processes are described in order to present the strengths and weaknesses of the photoelectrocatalytic treatment of industrial wastewater. This overview is an addition to the existing knowledge with a particular focus on the main experimental conditions employed in the photoelectrocatalytic degradation of various pollutants with the application of semiconductor photoelectrodes.
1. Introduction or History
In the XXI century, energy crisis and environmental pollution constitute two main problems in sustainable development, which is defined as the ability of natural systems to supply the natural resources and ecosystems while meeting current human needs, not affecting the ability of the future generations to meet their needs and without deteriorating of the integrity and stability of the natural systems.
Environmental pollution results in serious climate changes and is the most important threat to human population on a global scale nowadays. Thus, it is important to significantly limit the emission of pollutants to the environment. Industrial sources of environmental pollution generate huge amounts of industrial wastewater, which have serious impact on the environment. Although the industrial wastewater contains various recalcitrant organic and inorganic pollutants, it can be regarded as a prospective source of fresh water, energy, and valuable raw materials. Pollutants that are present in wastewater can be treated and simultaneously converted into methane in microbial fuel cells (MFC)[1][2][3][4][5][6][7][8], becoming a potential source of energy. Conventional sewage treatment systems are often not efficient enough for the complete degradation of pollutants present in the industrial wastewater and they are characterized by high energy consumption. On the other hand, the primary energy is still supplied from fossil fuels. Their combustion leads to the greenhouse effect and dangerous climate changes[9][10]. The pollution of the environment and increasing demand for energy supply has resulted in the development of renewable energy sources. Sunlight seems to be the cleanest and most abundant renewable source of energy, which should be applied in the industrial wastewater treatment[11].
Industrial wastewater usually contains different recalcitrant pollutants and its composition is complex. The wastewater pollutants include textile dyes, pharmaceuticals, pesticides, heavy metals, pulp and paper, surfactants, and other chemicals. Thus, an application of advanced treatment technologies might be required in order to remove or utilize them. Conventional sewage treatment systems are not efficient enough to completely degrade pollutants present in the industrial wastewater. Moreover, the chemical energy stored in the wastewater is wasted[12]. Therefore, it is important to apply clean, environmentally friendly, and efficient wastewater treatment technologies that can totally remove pollutants, recover fresh water and raw materials, and simultaneously recover the chemical energy contained in the wastewater. One of the alternatives to commonly applied wastewater treatment is heterogeneous photocatalysis. However, this technology has some drawbacks. Although many semiconductor photocatalysts are easily photoexcited by UV or VIS irradiation, are stable, and non-toxic, they suffer from low photocatalytic efficiency due to the fast recombination of holes and electron[13][14][15]. The photocatalytic treatment of wastewater usually does not degrade pollutants completely but results in a decrease of pollutant concentration and formation of intermediated products. The second alternative to the commonly applied wastewater treatment is electrocatalysis, especially electrochemical advanced oxidation processes (EAOPs) that are applied in the case of organic pollutants treatment[16][17][18][19]. However, total degradation of organic compounds present in the wastewater often requires so-called “deep electrooxidation” due to the necessity of high voltages application in order to degrade the pollutants to inorganic compounds, such as CO2, H2O, N2, etc., or to simple organic compounds that are easily biodegradable. These methods are characterized by high energy consumption, making the treatment process uneconomical. The photoelectrocatalytic treatment of industrial wastewater should overcome this problem, especially when it is coupled with energy generation. However, the selection of proper electrode materials applied in the photoelectrocatalytic degradation is very important for achieving a decrease in electrical energy consumption. Special attention should be paid to possible photoexcitation of electrode material by visible light (solar irradiation) instead of harmful and more energetic ultraviolet light. Moreover, high long-term stability, mechanical strength, high corrosion resistance, high efficiency of degradation processes, and low costs should characterize electrode materials.
2. Applications
Solar irradiation is regarded as a clean, safe and limitless source of photons. However, the photoexcitation of electrode materials by low energy photons requires the application of special semiconductors or coupled semiconductors or doped semiconductors that are capable of absorbing sunlight in order to degrade organic pollutants present in the industrial wastewater. Thus, three factors should be considered in the selection of semiconductors applied as electrode materials[20]:
(1) absorption of UV and VIS irradiation;
(2) charge-carrier transport within the semiconductor; and,
(3) interfacial charge transfer.
Besides these parameters, the semiconductor should also be characterized by appropriate position of valence and conduction bands related to oxidation and reduction potentials of pollutants, good electrical conductivity and sufficient stability to be applied in treatment processes implemented in practical applications on a large scale. The above-mentioned parameters are important in both wastewater treatment and energy generation.
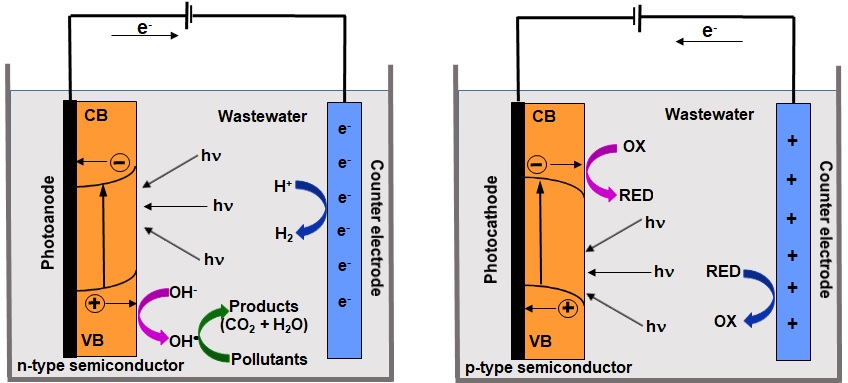
Figure 1. The overall mechanism of photoelectrocatalytic degradation of pollutants in wastewater with the application of n-type and p-type semiconductor electrodes.
The aim of this paper is to present a general overview on the semiconductor materials that are applied as photoelectrodes in the photoelectrocatalytic treatment of pollutants present in the industrial wastewater. The fundamentals of photoelectrocatalytic reactions and mechanism of pollutants treatment (Figure 1) as well as parameters affecting the treatment process are presented first. They are followed by examples of various semiconductor materials applied as photoelectrodes in the treatment of different kinds of pollutants in order to prove the advantages of photoelectrocatalytic treatment processes. This overview is an addition to the existing knowledge with a particular focus on the main experimental conditions employed in the photoelectrocatalytic degradation of various pollutants.
References
- Gude, V.G.; Wastewater treatment of microbial fuel cells - An overview. J. Clean. Prod. 2016, 24, 2095-2111.
- Chonde, S.G.; Microbial fuel cell: A new approach of wastewater treatment with power generation. Int. J. Chem. Environ. Pharmaceut. Res. 2014, 5, 8-12.
- Chaturvedi, V: Verma, P.; Microbial fuel cell: A green approach for the utilization of waste for the generation of bioelectricity. Bioresour. Bioprocess. 2016, 3, 38.
- Araneda, I.; Tapia, N.F.; Allende, K.L.; Vargas, I.T.; Constructed wetland-microbial fuel cells for sustainable greywater treatment. Water 2018, 10, 940.
- Tatinclaux, M.; Gregoire, K.; Leininger, A.; Biffinger, J.C.; Tender, L.; Ramirez, M.; Torrents, A.; Kjellerup, B.V.; Electricity generation from wastewater using a floating air cathode microbial fuel cell. Water Energy Nexus 2018, 1, 97-103.
- Guadarrama-Perez, O.; Gutiererez-Macias, T.; Garcia-Sanchez, L.; Guadarrama-Perez, V.H.; Estrada- Arriaga, E.B.; Recent advances in constructed wetland-microbial fuel cells for simultaneous bioelectricity production and wastewater treatment: A review. Int. J. Energ. Res. 2019, 43, 5106-5127.
- Antolini, E.; Photoelectrocatalytic fuel cells and photoelectrode microbial fuel cells for wastewater treatment and power generation. J. Environ. Chem. Eng 2019, 7, 103241.
- Wlodarczyk, P.P.; Wlodarczyk, B.; Preparation and analysis of Ni-Co catalyst used for electricity production and COD reduction in microbial fuel cells. Catalysts 2019, 9, 1042.
- Osterloh, F.E.; Parkinson, B.A.; Recent developments in solar water-splitting photocatalysis. MRS Bull. 2011, 36, 17-22.
- Habisreutinger, S.N.; Schmidt-Mende, L.; Stolarczyk, J.K.; Photocatalytic reduction of CO2 on TiO2 and other semiconductors. Angew. Chem. Int. Ed. 2013, 52, 7372-7408.
- Li, J.; Wu, N.; Semiconductor-based photocatalysts and photoelectrochemical cells for solar fuel generation: A review. Catal. Sci. Technol. 2015, 5, 1360-1384.
- Chen, X.; Chen, R.; Zhu, X.; Liao, Q.; Zhang, Y.; Ye, D.; Zhang, B.; Yu, Y.; Li, J.; A solar responsive cubic nanosized CuS/Cu2O/Cu photocathode with enhanced photoelectrochemical activity. J. Catal. 2019, 372, 182-192.
- Liang, H.; Jia, Z.; Zhang, H.; Wang, X.; Wang, J.; Photocatalysis oxidation activity regulation of Ag/TiO2 composites evaluated by the selective oxidation of Rhodamine B. Appl. Surf. Sci. 2017, 422, 1-10.
- Hu, Y.; Chen, W.; Fu, J.; Ba, M.; Sun, F.; Zhang, P.; Zou, J.; Hydrothermal synthesis of BiVO4/TiO2 composites and their application for degradation of gaseous benzene under visible light irradiation. Appl. Surf. Sci. 2018, 436, 319-322.
- Di Paola, A.; Garcia-Lopez, E.; Marci, G.; Palmisano, L.; A survey of photocatalytic materials for environmental remediation. J. Hazard. Mater. 2012, 211, 3-29.
- Brillas, E.; Martinez-Huitle, C.A.; Decontamination of wastewaters containing synthetic organic dyes by electrochemical methods. An updated review. Appl. Catal. B Environ. 2015, 166-167, 603-643.
- Oturan, M.A.; Aaron, J.J.; Advanced oxidation processes in water/wastewater treatment: Principles and applications. A review. Crit. Rev. Environ. Sci. Technol. 2014, 44, 2577-2641.
- Sires, I.; Brillas, E.; Oturan, M.A.; Rodrigo, M.A.; Panizza, M.; Electrochemical advanced oxidation processes: Today and tomorrow. A review. Environ. Sci. Pollut. Res. 2014, 21, 8336-8367.
- Chaplin, B.P.; Critical review of electrochemical advanced oxidation processes for water treatment applications. Environ. Sci. Processes Impacts 2014, 16, 1182-1203.
- Reyes-Gil, K.R.; Robinson, D.B.; WO3-enhanced TiO2 Nanotube photoanodes for solar water splitting with simultaneous wastewater treatment. Appl. Mater. Interfaces 2013, 5, 12400-12410.