
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Ronen Borenstein | + 839 word(s) | 839 | 2021-05-12 11:38:51 | | | |
| 2 | Rita Xu | Meta information modification | 839 | 2021-05-24 05:22:41 | | |
Video Upload Options
Viral pathogens often exploit host cell regulatory and signaling pathways to ensure an optimal environment for growth and survival. Several studies have suggested that 5′-adenosine monophosphate-activated protein kinase (AMPK), an intracellular serine/threonine kinase, plays a significant role in the modulation of infection.
1. Introduction
5′-adenosine monophosphate-activated protein kinase (AMPK) is an intracellular serine/threonine kinase that acts as a key metabolic regulator in maintaining cellular energy [1]. AMPK is activated by metabolic stress and acts to restore energy balance by changing cellular metabolism to generate energy through catabolic pathways such as involving glucose uptake, and by the inhibition of non-essential anabolic processes including lipid, protein and carbohydrate biosynthesis [2]. AMPK is also involved in autophagy, mitochondrial homeostasis, and mitophagy [3].
Due to its important role in the cell’s homeostasis, AMPK is an important cellular factor that many viruses utilize for replication, as it involves an energy-dependent process requiring high cellular ATP levels [4][5][6]. Viruses use AMPK to manipulate autophagy [7][8], fatty acid and lipid metabolism [5][9], glucose metabolism [10][11] and many other cellular processes.
2. AMPK Structure
AMPK is a heterotrimeric complex composed of a catalytic α subunit (two isoforms: α1/2), regulatory β subunit (two isoforms: β1/2) and γ subunit (three isoforms: γ1/2/3), which allows for the expression of 12 distinct complexes (Figure 1). The catalytic α subunit is characterized by serine/threonine kinase domains on the n-terminus and regulatory domain interactions occurring on the C-terminus [12][13][14]. The α1 isoform is widely expressed in all cells and accounts for 94% of the enzyme’s activity. Whereas, the α2 isoform is highly expressed in skeletal muscle, cardiac muscle, and liver [15]. In mammalian systems, activation of catalytic kinase domains occurs by the phosphorylation of a conserved threonine residue, Thr172, located in the activation loop [12]. In response to a decrease in cellular adenosine triphosphate (ATP) levels, the AMPK pathway is activated by three distinct mechanisms:
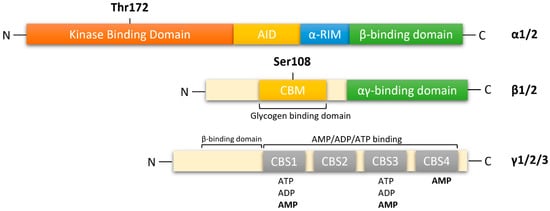
Figure 1. Functional domain of AMPK subunits. AMPK is a heterotrimeric complex composed of a catalytic α subunit(α1/2), regulatory β subunit (β1/2), and γ (γ1/2/3) subunit. AMPKα: kinase domain (KD) at the N-terminal contains Thr172, which is phosphorylated by upstream kinases; AID, auto-inhibitory domain; α-RIM: regulatory subunit interacting motif triggering conformational changes; β-subunit binding domain at the C-terminal. AMPKβ subunit: carbohydrate-binding module (CBM) near the N-terminal contains Ser108, which is important for the mechanism of action of some direct activators of AMPK; C-terminal domain containing the α-subunit-binding site and immediately followed by the domain for the γ-subunit interaction. αγ-binding domain: α-subunit-binding and γ-subunit interaction site at the C-terminal. AMPKγ subunit: cystathione-β-synthases (CBS) domain, which forms two Bateman domains containing four ATP/ADP/AMP-binding sites (CBS1–4).
- (1)
- (2)
-
Inhibition of Thr172 dephosphorylation by protein phosphatases, PP2a or PP2c (mechanisms 1 and 2 require the presence of adenosine monophosphate (AMP) and adenosine diphosphate (ADP) [1];
- (3)
-
Allosteric activation of AMPK by AMP [19].
In response to decreasing AMP levels, auto inhibitory domain (AID) and regulatory subunit interacting motifs (α-RIM) on the C-terminal of the α subunit, maintain the α-kinase domains (α-KD) in an inactive conformation [13].
The β-subunits have myristoylated carbohydrate-binding molecules (CBM) in the N-terminus and interaction domains on the C-terminus [13][14]. In mammalian cells, modifications of AMPK occur cotranslationally by myristoylation of glycine residue 2 (Gly2) in the N-terminal of the β1-subunit [20][21]. This association allows β-subunits to form core complexes by linking the α C-terminus and γ N-terminus, forming an ST loop that contains phosphorylation sites for cAMP-dependent protein kinase (PKA) and serine/threonine protein kinases such as protein kinase B and glycogen synthase kinase (GSK) [14][22]. Hence, in response to AMP, myristoylation of the β-subunit is an essential requirement for AMPK signaling initiation [21]. The β1 isoform is more highly expressed in the liver than in skeletal muscle, whereas the β2 isoform is highly expressed in skeletal muscle [23].
The γ subunit contains four cystathionine-β-synthase (CBS) domains that create binding sites for adenine nucleotides, AMP, ADP and ATP (Figure 1). Thus, the γ-subunit plays an important role in AMPK regulation in response to cellular energy levels. CBS isoforms CBS1/CBS2 and CBS3/CBS4 assemble into complexes that generate four potential ligand-binding clefts in the center, which can be activated by AMP [13][14]. Regulatory site 3 in the CBS complex appears to be critical, as it binds to AMP at a greater affinity than ATP. AMP bound on regulatory site 3 facilitates interactions between α and γ subunits, leading to a compact conformational change in structure [14][24]. Conserved α-regulatory subunit interacting motif (α-RIM) within the α-linker interacts with the AMPK-γ subunit when AMP is bound at CBS3 [13]. Interestingly, phosphorylation of Thr172 by upstream kinases and simultaneous allosteric activation of AMPK (by binding of AMP to CBS domains) increases kinetic activity by ≥1000-fold [12][14][25]. As ATP displaces AMP on site 3, α-linkers are released from γ-subunits, reversing the conformational change. The regulatory γ subunit plays a vital role in activating the catalytic α subunit and the formation of heterotrimeric complexes [13][25]. The γ isoforms have the greatest variability in structure. The γ1 isoform (331 residues) is widely expressed in cells and tissues, and it is activated up to 3-fold [26]. Expression of γ2 (569 residues) is restricted to the brain, placenta, skeletal and cardiac muscle, where activation by AMP leads to a 3-fold increase. The γ3 isoform (489 residues) is restricted to skeletal muscle [26].
References
- Oakhill, J.S.; Chen, Z.-P.; Scott, J.W.; Steel, R.; Castelli, L.A.; Ling, N.; Macaulay, S.L.; Kemp, B.E. β-Subunit myristoylation is the gatekeeper for initiating metabolic stress sensing by AMP-activated protein kinase (AMPK). Proc. Natl. Acad. Sci. USA 2010, 107, 19237–19241.
- Matos, A.L.D.S.A.; Oakhill, J.S.; Moreira, J.; Loh, K.; Galic, S.; Scott, J.W. Allosteric regulation of AMP-activated protein kinase by adenylate nucleotides and small-molecule drugs. Biochem. Soc. Trans. 2019, 47, 733–741.
- Herzig, S.; Shaw, R.J. AMPK: Guardian of metabolism and mitochondrial homeostasis. Nat. Rev. Mol. Cell Biol. 2018, 19, 121–135.
- Silwal, P.; Kim, J.K.; Yuk, J.-M.; Jo, E.-K. AMP-Activated Protein Kinase and Host Defense against Infection. Int. J. Mol. Sci. 2018, 19, 3495.
- Mankouri, J.; Tedbury, P.R.; Gretton, S.; Hughes, M.E.; Griffin, S.D.C.; Dallas, M.L.; Green, K.A.; Hardie, D.G.; Peers, C.; Harris, M. Enhanced hepatitis C virus genome replication and lipid accumulation mediated by inhibition of AMP-activated protein kinase. Proc. Natl. Acad. Sci. USA 2010, 107, 11549–11554.
- Brunton, J.; Steele, S.; Ziehr, B.; Moorman, N.; Kawula, T. Feeding Uninvited Guests: mTOR and AMPK Set the Table for Intracellular Pathogens. PLoS Pathog. 2013, 9, e1003552.
- Xie, N.; Yuan, K.; Zhou, L.; Wang, K.; Chen, H.-N.; Lei, Y.; Lan, J.; Pu, Q.; Gao, W.; Zhang, L.; et al. PRKAA/AMPK restricts HBV replication through promotion of autophagic degradation. Autophagy 2016, 12, 1507–1520.
- König, P.; Svrlanska, A.; Read, C.; Feichtinger, S.; Stamminger, T. The Autophagy-Initiating Protein Kinase ULK1 Phosphorylates Human Cytomegalovirus Tegument Protein pp28 and Regulates Efficient Virus Release. J. Virol. 2020, 95, 02346-20.
- De Oya, N.J.; Blázquez, A.-B.; Casas, J.; Saiz, J.-C.; Martín-Acebes, M.A. Direct Activation of Adenosine Monophosphate-Activated Protein Kinase (AMPK) by PF-06409577 Inhibits Flavivirus Infection through Modification of Host Cell Lipid Metabolism. Antimicrob. Agents Chemother. 2018, 62, 00360-18.
- Nakashima, K.; Takeuchi, K.; Chihara, K.; Hotta, H.; Sada, K. Inhibition of hepatitis C virus replication through adenosine monophosphate-activated protein kinase-dependent and -independent pathways. Microbiol. Immunol. 2011, 55, 774–782.
- Noch, E.; Sariyer, I.K.; Gordon, J.; Khalili, K. JC Virus T-Antigen Regulates Glucose Metabolic Pathways in Brain Tumor Cells. PLoS ONE 2012, 7, e35054.
- Hardie, D.G. AMP-activated protein kinase: An energy sensor that regulates all aspects of cell function. Genes Dev. 2011, 25, 1895–1908.
- Hardie, D.G.; Schaffer, B.E.; Brunet, A. AMPK: An Energy-Sensing Pathway with Multiple Inputs and Outputs. Trends Cell Biol. 2016, 26, 190–201.
- Hardie, D.G.; Lin, S.-C. AMP-activated protein kinase—not just an energy sensor. F1000Research 2017, 6, 1724.
- Stapleton, D.; Mitchelhill, K.I.; Gao, G.; Widmer, J.; Michell, B.J.; Teh, T.; House, C.M.; Fernandez, C.S.; Cox, T.; Witters, L.A.; et al. Mammalian AMP-activated Protein Kinase Subfamily. J. Biol. Chem. 1996, 271, 611–614.
- Neumann, D. Is TAK1 a Direct Upstream Kinase of AMPK? Int. J. Mol. Sci. 2018, 19, 2412.
- Luo, L.; Jiang, S.; Huang, D.; Lu, N.; Luo, Z. MLK3 Phophorylates AMPK Independently of LKB1. PLoS ONE 2015, 10, e0123927.
- Ke, R.; Xu, Q.; Li, C.; Luo, L.; Huang, D. Mechanisms of AMPK in the maintenance of ATP balance during energy metabolism. Cell Biol. Int. 2018, 42, 384–392.
- Ross, F.A.; Jensen, T.E.; Hardie, D.G. Differential regulation by AMP and ADP of AMPK complexes containing different γ subunit isoforms. Biochem. J. 2016, 473, 189–199.
- Oakhill, J.S.; Scott, J.W.; Kemp, B.E. AMPK functions as an adenylate charge-regulated protein kinase. Trends Endocrinol. Metab. 2012, 23, 125–132.
- Ali, N.; Ling, N.; Krishnamurthy, S.; Oakhill, J.S.; Scott, J.W.; Stapleton, D.I.; Kemp, B.E.; Anand, G.S.; Gooley, P.R. β-subunit myristoylation functions as an energy sensor by modulating the dynamics of AMP-activated Protein Kinase. Sci. Rep. 2016, 6, 39417.
- Yan, Y.; Zhou, X.E.; Xu, H.E.; Melcher, K. Structure and Physiological Regulation of AMPK. Int. J. Mol. Sci. 2018, 19, 3534.
- Thornton, C.; Snowden, M.A.; Carling, D. Identification of a Novel AMP-activated Protein Kinase β Subunit Isoform That Is Highly Expressed in Skeletal Muscle. J. Biol. Chem. 1998, 273, 12443–12450.
- Xiao, B.; Sanders, M.J.; Carmena, D.; Bright, N.J.; Haire, L.F.; Underwood, E.; Patel, B.R.; Heath, R.B.; Walker, P.A.; Hallen, S.; et al. Structural basis of AMPK regulation by small molecule activators. Nat. Commun. 2013, 4, 3017.
- An, H.; Wang, Y.; Qin, C.; Li, M.; Maheshwari, A.; He, L. The importance of the AMPK gamma 1 subunit in metformin suppression of liver glucose production. Sci. Rep. 2020, 10, 1–10.
- Moffat, C.; Harper, M.E. Metabolic functions of AMPK: Aspects of structure and of natural mutations in the regulatory gamma subunits. IUBMB Life 2010, 62, 739–745.




