3. Fabry Cardiomyopathy
Cardiac manifestations of FD include increased left ventricular (LV) wall thickness, conduction abnormalities, arrhythmias, valvular disease, and aortic dilatation, which result from glycolipid deposition and subsequent fibrosis of contractile cardiomyocytes, conductive cardiomyocytes, valvular interstitial cells, and smooth muscle cells of the cardiovascular system (). Eventually, complications such as hypertension, myocardial infarction, and cardiac death may occur, with heart failure being the most common first cardiovascular event in FD
[12]. Compared to other organs, the heart appears to be the most susceptible to low levels of α-Gal A. The FD-related cardiovascular injury is thought to be due to a combination of Gb
3 accumulation, the accumulation of trophic factors, and microcirculatory ischemia, which contribute to inflammation and ultimately result in myocardial fibrosis
[13]. Patients with FD-related cardiac involvement tend to be asymptomatic from a cardiac perspective during the first four decades of life, then present with non-specific cardiac symptoms such as angina, dyspnea, palpitations, or syncope. Since there is no pathognomonic cardiac manifestation of FD, the non-specific findings often make FD-related cardiac involvement difficult to diagnose.
Table 1. Cardiac manifestations of Fabry disease.
|
Structural abnormalities detected by cardiac imaging
|
-
Increased LV wall thickness. Morphologies include concentric hypertrophy (most common), asymmetric septal hypertrophy, eccentric hypertrophy, and apical hypertrophy. Associated LV outflow tract obstruction may be present but often not [4]
-
LV ejection fraction often preserved but may be reduced with advanced disease [4]
-
Biatrial enlargement due to chronic diastolic dysfunction or underlying atrial myopathy [4]
-
Prominent papillary muscles [4]
-
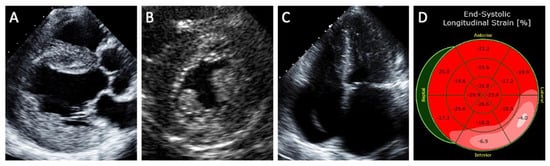
Reduced LV longitudinal strain on echocardiography and T2 elevation (suggesting inflammation) or late gadolinium enhancement (suggesting fibrosis) on MRI in the basal inferolateral segment [4]
-
Reduced native T1 values on MRI [4]
-
Abnormal LV diastolic function [4]
-
Binary sign (no longer considered sensitive or specific for Fabry cardiomyopathy) [4]
-
RV wall thickness may be increased [4]
-
Thickening and redundancy of the valves with some degree of valvular regurgitation, although often not significant enough to warrant intervention [4]
-
Aortic dilatation [4]
|
|
Electrophysiologic abnormalities detected by ECG or prolonged rhythm monitoring
|
-
Short PR interval in younger patients, prolonged PR interval in older patients [14]
-
Bradycardia from chronotropic incompetence [15]
-
Sinus node dysfunction [15]
-
Varying degrees of AV block [15]
-
Atrial arrhythmias including atrial tachycardia, atrial flutter, or atrial fibrillation [15]
-
Ventricular arrhythmias including non-sustained VT and sustained VT [15]
|
Abbreviations: AV, atrioventricular; ECG, electrocardiogram; LV, left ventricular; MRI, magnetic resonance imaging; RV, right ventricular; VT, ventricular tachycardia.
The hallmark feature of FD cardiomyopathy is increased LV wall thickness
[6][9]. Increased right ventricular wall thickness and impaired right ventricular function have also been reported
[16]. Increased LV wall thickness is rarely present in children with FD, tends to be more severe in male FD patients, and is usually not evident until the third or fourth decade in classical FD patients
[4][17]. However, the finding of increased LV wall thickness is not specific for Fabry cardiomyopathy, and it is important for clinicians to consider the differential diagnoses of other causes of increased LV wall thickness ().
Table 2. Differential diagnosis of increased LV wall thickness and common findings on patient history, ECG, echocardiography, and CMR.
| |
Patient History
|
ECG
|
Echocardiography
|
CMR
|
|
Fabry Cardiomyopathy
|
-
Angiokeratoma corporis
-
Acroparesthesia
-
Diarrhea
-
Stroke
-
Chest pain
-
Heart failure
|
-
Short PR interval
-
Prolonged QRS
-
High voltage QRS
|
|
|
|
Hypertension
|
|
|
-
Concentric LVH
-
Diastolic dysfunction
-
Reduced LV GLS
|
|
|
Athlete’s Heart
|
|
|
|
-
Absence of LGE
-
Normal LV SVI
|
|
Aortic Stenosis
|
-
Chest pain
-
Dyspnea
-
Syncope
|
-
High voltage QRS
-
Left atrial enlargement
|
-
Aortic stenosis
-
Concentric LVH
|
|
|
Hypertrophic Cardiomyopathy
|
|
-
High voltage QRS
-
Left atrial enlargement
-
Atrial fibrillation
|
|
|
|
Cardiac Amyloidosis
|
-
Heart failure
-
Bilateral carpal tunnel
-
Nephrotic syndrome
-
Macroglossia
-
Peripheral neuropathy
|
-
Low voltage QRS
-
Atrial fibrillation
-
Pseudo-infarct
|
|
|
Abbreviations: CMR, cardiovascular magnetic resonance imaging; ECG, electrocardiogram; GLS, global longitudinal strain; LGE, late gadolinium enhancement; LV, left ventricular; LVH, left ventricular hypertrophy, LVOT, left ventricular outflow tract; PM, papillary muscle; SVI, stroke volume index.
Electrophysiologic abnormalities represent other common cardiac manifestations of FD
[9]. Advanced conduction disease is thought to be caused by glycolipid accumulation in cardiomyocytes of the atrioventricular (AV) node, bundle of His, and the left and right bundle branches
[18]. In contrast, accelerated AV conduction is common in younger FD patients and is reflected as shortened PR intervals on the electrocardiogram (ECG), while prolonged PR interval may be observed in older FD patients
[18]. Atrial and ventricular arrhythmias are also relatively common and may be due to atrial myopathy, atrial dilatation from longstanding diastolic dysfunction, and atrial and ventricular fibrosis. Atrial arrhythmias such as atrial fibrillation are more common than ventricular arrhythmias.
In addition, valvular diseases such as aortic, mitral, and tricuspid regurgitation are common in patients with FD due to mild thickening of the valves, although valvular regurgitation significant enough to require intervention is uncommon and stenotic lesions attributed to Fabry disease alone are rare
[19]. Thickening of papillary muscles in FD patients has been proposed as a mechanism of mitral regurgitation in FD. Fabry disease can also lead to aortic dilatation, especially in males, where its prevalence increases with age. Aortic dilatation in FD has been shown to be independent of elevated blood pressure
[20] and has been attributed to degenerative changes in the aortic media due to excessive glycolipid substrate deposition
[21]. Significant aortic dilatation due to Fabry disease resulting in acute aortic events has yet to be reported.
Fabry cardiomyopathy may not be as rare as we once thought as it has been shown to be responsible for up to 4% of unexplained hypertrophic cardiomyopathy (HCM) cases
[22][23] and up to 12% of unexplained increased LV wall thickness in other selected cohorts
[9]. When assessing for pathogenic mutations only, the prevalence of
GLA mutation in LVH or HCM clinics is 0.94% in males and 0.90% in females
[24]. In fact, the cardiac variant is the most common form of FD in some countries such as Taiwan
[25]. This is of particular concern as cardiovascular complications represent the predominant source of FD-related mortality and morbidity
[8][26].