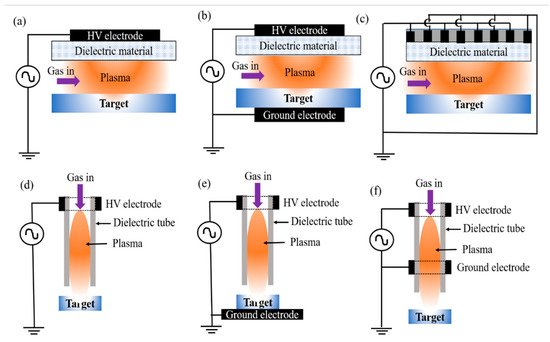
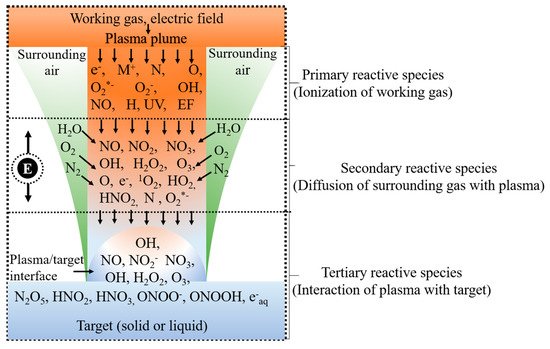
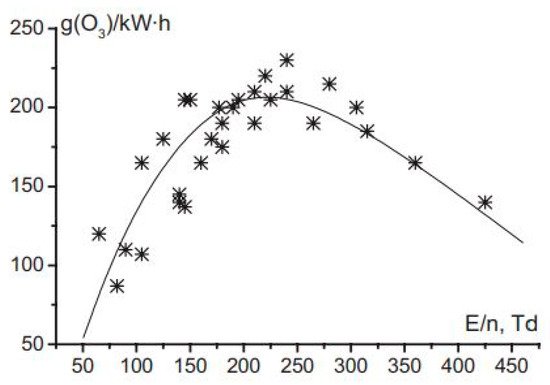
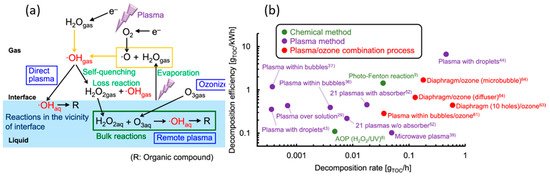
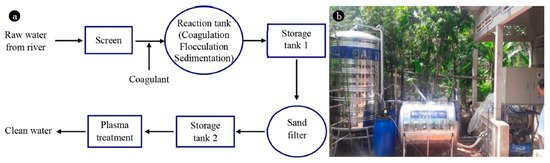
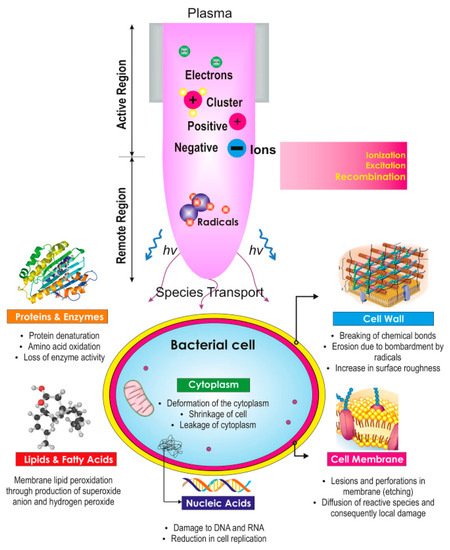
As mentioned earlier, conventional water treatment methods cannot satisfactorily remove all contaminants. For instance, species that are resistant to Cl (e.g., bacteria spores and protozoa) are not eliminated by conventional methods, although AOPs can be used with traditional techniques such as filtration to decompose these contaminants. Furthermore, NTAPP species (charged species, reactive species, and UV photons) have significant effects on sterilization and microorganism disinfection. Previous studies have demonstrated that the interaction between plasma and water can produce H
2O
2, which has wide scope to disinfect the various microorganisms such as viruses, molds, and bacteria. It is worth noting that some chemical and physical parameters of the liquid (e.g., overall hardness and total dissolved solid, and conductivity) change slightly due to interaction with the plasma [
44]. Substantial research has been conducted in this area in recent years. The present article reviews numerous research papers concerning the interaction of plasma with wastewater to inactivate bacteria.
Zhang et al. [
87] used an AC-driven microplasma jet array under atmospheric pressure with a repetition frequency of several kilohertz to inactivate resistant
Pseudomonas sp. HB1 cells in water. The results showed that the species with short lifetimes (such as charged particles and OH radicals) in microplasma jets could effectively inactivate resistant
Pseudomonas sp. cells. With 108 colony-forming units (CFUs) in an 80 mL suspension, all
Pseudomonas sp. cells were killed within 6 min of plasma remedy. Various studies have been conducted on the elimination of
Escherichia coli in wastewater using NTAPP. Similarly, Ziuzina et al. [
88] considered the effects of cold plasma produced in a closed chamber on
Escherichia coli inactivation under atmospheric pressure. All samples in this research were directly and indirectly treated with plasma, and the impacts of treatment and post-treatment time were investigated. The results demonstrated that this plasma configuration can eliminate high concentrations of
Escherichia coli in water in several seconds. An important benefit of this approach is the absence of post-processing pollution. Plasma treatment directly leads to the complete inactivation of bacteria. Meanwhile, Zheng et al. [
89] used water-pulsed NTAPP for water disinfection (
Escherichia coli inactivation). Their results indicated that the liquid conductivity had a significant role in the creation of plasma containing reactive species and UV emission and revealed its effect on disinfection. The disinfection effect was dramatic when the conductivity of water was up to 1.5 mS/cm. This study indicated that during water sterilization, OH radicals has strong chemical sterilizing power. However, because of the short lifetime of OH radicals, which is only a few milliseconds, it is difficult to use OH radicals to disinfect pathogenic microbes directly. Hangbo et al. [
43] investigated the effects of various ROS with short lifetimes on yeast cell inactivation in a liquid and showed that
1O
2, OH radicals, and O
2− are the three main particles created in a plasma–liquid system. Based on the experimental results, OH radicals has the least contribution to plasma inactivation among the plasma species. In contrast, O
2− and plasma acid play major roles in inactivation. It is worth noting that in the plasma–liquid interaction,
1O
2 is a more significant antimicrobial factor than the species mentioned earlier (i.e., OH radicals and O
2−). It should be noted that despite the strong oxidizing properties of OH radicals, it does not have the capacity for significant diffusion [
43]. Because of its short life and high reactivity, it can only react with nearby particles. On the contrary,
1O
2 can effectively diffuse into the cell layer and initiate peroxidation of lipid due to its lifetime being longer than that of OH. Moreover, the results illustrated that in plasma devices (e.g., DBD plasma and plasma micro jets), the
1O
2 concentration is often greater than the OH radicals concentration. Nevertheless, O
2− requires a greater oxidation ability to remove H
2 from the phospholipids and consequently can only enter the cell hydrophobic area with significant effort. However, by the Haber–Weiss reaction [
90], O
2− can be changed into OH radicals and converted into hydroperoxyl (HOO) radicals by protonation when the pH is less than 4.7. Hence, these species can easily enter the cell hydrophobic area. Moreover, it has been demonstrated that plasma acid is a significant factor in the inactivation of plasma–liquid media. It is a vital parameter in enabling plasma species to pass into cell walls and in decreasing the resistance of cells versus acidic media [
91,
92,
93,
94]. To enhance the effects of sterilization, it is important to develop a device in which most components in the liquid (microorganisms) could come into direct contact with the plasma. The results showed that NTAPP performs sterilization much more effectively, because the concentrations of RONS causing sterilization are quite high in NTAPP, and the probability of reaction between
Escherichia coli and the plasma-generated RONS increases [
50]. Both nitrous and nitric acid are formed through the interaction of NO and NO
2 with water by hydrolysis. Meanwhile, NO
x is produced by reaction with the air, but when plasma interacts with liquid and thus does not have the opportunity to contact air, NO
x generation barely occurs. Furthermore, air plasma jet have a greater sterilization effect than nitrogen plasma jet due to the generally huge number of reactive species causing bacterial inactivation [
50]. Moreover, the radical concentration affects the sterilization in proportion to the plasma length (e.g., it increases with increasing gas flow rate). In another study [
86], the behavior of the surface potential of bacteria was investigated as a function of the applied energy by analyzing the molecular levels of DNA and proteins. The applied pulse voltage (23 kV) and frequency (25 Hz) in the experiments enabled rapid disinfection. In a treatment time of 6 min, complete disinfection was achieved. This result indicated that plasma could increase the protein leakage via the membrane of bacteria. It is interesting that in this case, approximately 70% of all proteins were leaked during the first 8 min of treatment.
The plasma spark method is another important approach for water disinfection. Rashmei et al. [
95] investigated water disinfection (
Escherichia coli and
Enterococcus faecalis inactivation) using the plasma spark technique and compared the results with those of ordinary methods. In this research, some of the chemical and physical parameters of the water were evaluated. The results demonstrated that
Escherichia faecalis and
Escherichia coli decrease by 8 log CFU after 12 and 15 min, respectively. Moreover, this research showed that major bacteria inactivation in water is achievable using electric fields and H
2O
2 molecules produced using plasma. NTAPP also has a powerful ability to remove bacteria. A concentration reduction of 8 log CFU was observed in all samples. In this method, some plasma species (e.g., H
2O
2, OH radicals, O
3) are strong oxidizers with the necessary ingredients of bacterial invasions, including lipids, proteins, and DNA, as summarized in .
After applying plasma to drinking or wastewater, the effects of different types of organic and inorganic matter on the disinfection process have been investigated. The results have shown that the concentrations of inorganic and organic compounds in tap water can change after treatment [
70]. It is worth noting that these changes are ascribed to the material interaction of the plasma species in tap water. It is advantageous that NTAAP can dramatically change the organoleptic characteristics of the water, such as smell, while increasing the amount of soluble O and decreasing the required concentration. However, plasma dramatically increases the concentrations of NO
2 and NO
3 and can significantly enhance the conductivity and decrease the pH [
44]. On the other hand, cold plasma also has a negative effect on turbidity. Thus, the turbidity increases after treatment, because by using O
3 dispersion with a porous plate, large particles can be broken into many small ones [
44]. Furthermore, plasma significantly enhances the concentrations of NO
2 and NO
3 due to the existence of N in air in the plasma formation zone. Thus, plasma technology could solve the existing drawback in conventional water sterilization and disinfection method using the exchange resin of the anion. To design a plasma device, parameters such as the device output, device lifetime, operation volume, known toxicities of the ingredients, and pre- and post-remedy demands should be considered [
45]. One of the major benefits of increasing the scale of plasma devices from the laboratory scale to the industrial scale is that the size of the of plasma–liquid interface can be increased [
96]. The most effective devices for water treatment are those that expose thin layers of the liquid to plasma because the plasma–liquid interface determines the treatable throughput. These configurations increase the plasma induced in the water as much as possible. Reactors with high throughput are convenient for practical applications.