Presoaking seeds in water (hydropriming) or in a solution, usually of polyethylene glycol (PEG) or various salts at low water potential (osmopriming), has been demonstrated to improve the germination of seeds of numerous species including vegetables (carrot, celery, leek, lettuce, tomato), floral plants (cyclamen, primrose, pansy) and others (sugar beet, rape, soybean, sunflower). This treatment allows the germination stricto sensu to occur but prevents the radicle protrusion. Germination of primed seeds is more rapid and uniform than that of unprimed ones. Primed seeds germinate in a wider range of temperatures and are less sensitive to oxygen deprivation. Interestingly, priming also improves the germination of aged seeds.
- seed quality
- hydropriming
- osmopriming
1. Introduction
2. Main Conventional Seed Priming Techniques
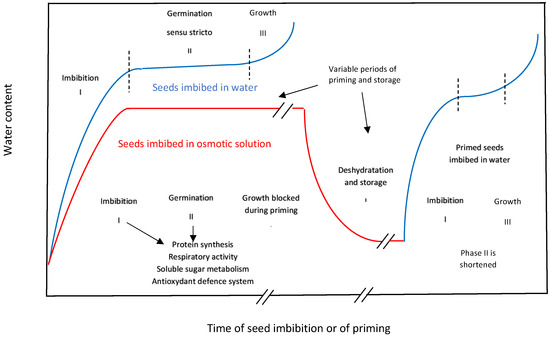
3. Beneficial Effects of Priming
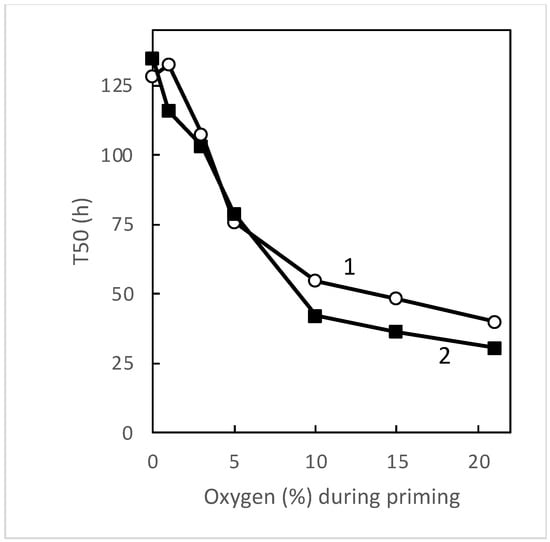
3.1. Seed Sensitivity to Temperature and Oxygen
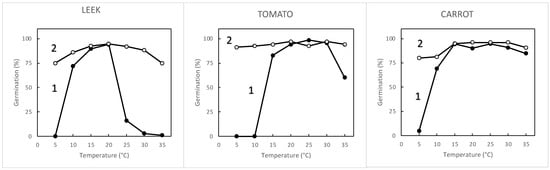
| Species | Seed Treatment | Germination (%) in Atmosphere Containing 1 to 21% Oxygen | |||||
|---|---|---|---|---|---|---|---|
| 1% | 3% | 5% | 10% | 15% | 21% | ||
| Carrot [36] | Non-primed Primed |
0 0 |
0 5.5 |
0 34.6 |
13.1 78.5 |
23.0 84.6 |
70.0 94.1 |
| Lamb’s lettuce [17][33] | Non-primed Primed |
0 0 |
0 4.4 |
4.2 23.0 |
5.3 92.2 |
10.5 97.5 |
51.1 98.1 |
| Leek [19][33] | Non-primed Primed |
0 0 |
0 11.4 |
3.1 34.4 |
20.2 85.2 |
52.2 93.3 |
53.2 95.2 |
| Sunflower [34][35] | Non-primed Primed |
4.6 10.2 |
40.7 75.6 |
55.6 95.3 |
79.6 100 |
92.5 100 |
100 100 |
| Tomato [31][33][36] | Non-primed Primed |
0 2.2 |
0 10.1 |
0 50.1 |
0 76.7 |
20.7 92.4 |
48.4 95.5 |
3.2. Germination of Aged Seeds
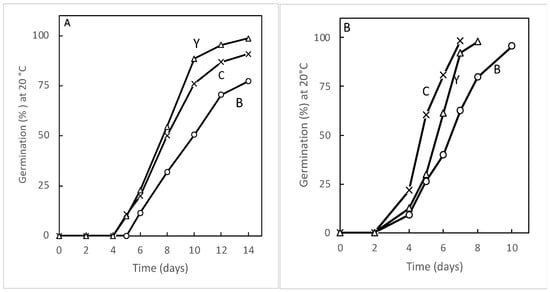
3.3. Examples of Priming Beneficial Effects on Several Species
This entry is adapted from the peer-reviewed paper 10.3390/seeds2010008
References
- Halmer, P. Methods to improve seed performance in the field. In Handbook of Seed Physiology: Applications to Agriculture; Benech-Arnold, R.L., Sanchez, R.A., Eds.; The Haworth Reference Press: New York, NY, USA; London, UK; Oxford, UK, 2004; pp. 125–166.
- Corbineau, F.; Côme, D. Priming: A technique for improving seed quality. Seed Test. Inter. 2006, 132, 38–40.
- Waqas, M.; Korres, N.E.; Khan, M.D.; Nizami, A.S.; Deeba, F.; Ali, I.; Hussain, H. Advances in the concept and methods of seed priming. In Priming and Pretreatment of Seeds and Seedlings; Hasanuzzaman, M., Fotopoulos, V., Eds.; Springer Nature Singapore Pte Ltd.: Singapore, 2019; pp. 11–41.
- Kumar, P. A review on seed priming techniques in field crops. Inter. J. Progress. Res. Sci. Eng. 2020, 1, 86–91.
- Garcia, D.; Zhao, S.; Arif, S.; Zhao, Y.; Ming, L.C.; Huang, D. Seed priming technology as a key strategy to increase crop plant production under adverse environmental conditions. J. Agric. Hortic. Res. 2022, 5, 27–35.
- Harada, J.J. Seed maturation and control of germination. In Cellular and Molecular Biology of Plant Seed Development; Larkins, B., Vasil, I., Eds.; Kluwer Academic Publishers: Dordrecht, The Netherlands, 1997; pp. 545–592.
- Al-Chaarani, G.R.; Gentzbittel, L.; Wedzony, M.; Sarrafi, A. Identification of QTLs for germination and seedling development in sunflower (Helianthus annuus L.). Plant Sci. 2005, 169, 221–227.
- Finch-Savage, W.E.; Clay, H.A.; Lynn, J.R.; Morris, K. Towards a genetic understanding of seed vigour in small-seeded crops using natural variation in Brassica oleracea. Plant Sci. 2010, 179, 582–589.
- Vandecasteele, C.; Teulat-Merah, B.; Morère-Le Paven, M.C.; Leprince, O.; Ly Vu, B.; Viau, L.; Ledroit, L.; Pelletier, S.; Payer, N.; Satour, P.; et al. Quantitative trait loci analysis reveals a correlation between the ratio of sucrose/raffinose family oligosaccharides and seed vigour in Medicago truncatula. Plant Cell Environ. 2011, 34, 1473–1487.
- Dias, P.; Brunel-Muguet, S.; Dürr, C.; Huguet, T.; Demilly, D.; Wagner, M.-H.; Teulat-Merah, B. QTL analysis of seed germination and pre-emergence growth at extreme temperatures in Medicago truncatula. Theor. Appl. Genet. 2011, 122, 429–444.
- McDonald, M.B. Seed quality assessment. Seed Sci. Res. 1998, 8, 265–275.
- McDonald, M.B. Seed Enhancements. In Seed Science and Technology; Copeland, L.O., McDonald, M.B., Eds.; Kluwer Academic Publishers: Dordrecht, The Netherlands, 2001; pp. 277–296.
- Ligterink, W.; Joosen, R.V.L.; Hilhorst, H.W.M. Unravelling the complex trait of seed quality: Using natural variation through a combination of physiology, genetics and –omics technologies. Seed Sci. Res. 2012, 22, S45–S52.
- Guedes, A.C.; Cantliffe, D.J. Germination of lettuce seeds at high temperature after seed priming. J. Am. Soc. Hortic. Sci. 1980, 105, 777–781.
- Brocklehurst, P.A.; Dearman, J. Interactions between seed priming treatments and nine seedlots of carrot, celery and onion. I. Laboratory germination. Ann. Appl. Biol. 1983, 102, 577–584.
- Brocklehurst, P.A.; Dearman, J. Interactions between seed priming treatments and nine seedlots of carrot, celery and onion. II. Seedling emergence and plant growth. Ann. Appl. Biol. 1983, 102, 585–593.
- Corbineau, F.; Côme, D. Effects of priming on the germination of Valerianella olitoria seeds in relation with temperature and oxygen. Acta Hortic. 1990, 267, 191–197.
- Corbineau, F.; Picard, M.A.; Côme, D. Germinability of some vegetable seeds in relation to temperature and oxygen. In Fourth International Worshop on Seeds. Basic and Applied Aspects of Seed Biology; Côme, D., Corbineau, F., Eds.; ASFIS: Paris, France, 1993; Volume 3, pp. 1027–1032.
- Corbineau, F.; Picard, M.A.; Côme, D. Germinability of leek seeds and its improvement by osmopriming. Acta Hortic. 1994, 371, 45–52.
- Capron, I.; Corbineau, F.; Dacher, F.; Job, C.; Côme, D.; Job, D. Sugar seed priming: Effects of priming conditions on germination, solubilization of 11-S globulin and accumulation of LEA proteins. Seed Sci. Res. 2000, 10, 243–254.
- Taylor, A.G.; Allen, P.S.; Bennett, M.A.; Bradford, K.J.; Burris, J.S.; Misra, M.K. Seed enhancements. Seed Sci. Res. 1998, 8, 245–256.
- Rowse, H.R. Methods of Priming Seeds. In Priming and Pretreatment of Seeds and Seedlings; Hasanuzzaman, M., Fotopoulos, V., Eds.; Springer: Singapore, 1991.
- Warren, J.E.; Bennett, M.A. Seed hydration using the drum priming system. Hortic. Sci. 1997, 31, 1220–1221.
- Heydecker, W.; Higgins, J.; Gulliver, R.L. Accelerated germination by osmotic seed treatment. Nature 1973, 246, 42.
- Bray, C.M. Biochemical processes during osmopriming of seeds. In Seed Development and Germination; Kigel, J., Galili, G., Eds.; Marcel Dekker, Inc.: New York, NY, USA, 1995; pp. 767–789.
- Lutts, S.; Benincasa, P.; Wojtyla, L.; Kubala, S.; Pace, R.; Lechowska, K.; Quinet, M.; Garnczarska, M. Seed priming: New comprehensive approaches for an old empirical technique. In New Challenges in Seed Biology—Basic and Translational Research Driving Seed Technology; Ajaujo, S., Balestrazzi, A., Eds.; IntechOpen: London, UK, 2016; pp. 1–46.
- Rajjou, L.; Duval, M.; Gallardo, K.; Catusse, J.; Bally, J.; Job, C.; Job, D. Seed germination and vigor. Annu. Rev. Plant Biol. 2012, 63, 507–533.
- Kalal, P.R.; Jajoo, A. Priming with Zinc oxide nanoparticles improve germination and photosynthetic performance in wheat. Plant Physiol. Biochem. 2021, 160, 341–351.
- Reddy, P.P. (Ed.) Biopriming of seeds. In Recent Advances in Crop Protection India; Springer Science & Business Media: New Delhi, India, 2013; pp. 83–90.
- Dearman, J.; Brocklehurst, P.A.; Drew, R.L.K. Effects of osmotic priming and ageing on the germination and emergence of carrot and leek seed. Ann. Appl. Biol. 1987, 111, 717–722.
- Özbingöl, N.; Corbineau, F.; Côme, D. Responses of tomato seeds to osmoconditioning as related to temperature and oxygen. Seed Sci. Res. 1998, 8, 377–384.
- Özbingöl, N.; Corbineau, F.; Groot, S.P.C.; Bino, R.J.; Côme, D. Activation of the cell cycle in tomato (Lycopersicon esculentum Mill.) seeds during osmoconditioning as related to temperature and oxygen. Ann. Bot. 1999, 84, 245–251.
- Côme, D.; Özbingöl, N.; Picard, M.A.; Corbineau, F. Beneficial effects of priming on seed quality. In Progress in Seed Research. Conference Proceedings of the Second International Conference on Seed Science and Technology; Taylor, A.G., Huang, X.L., Eds.; Agricultural Experimental Station: Geneva, Switzerland, 1998; pp. 257–263.
- Smok, M.A.; Chojnowski, M.; Corbineau, F.; Côme, D. Effect of osmotic treatment on sunflower seed germination in relation with temperature and oxygen. In Fourth International Workshop on Seeds: Basic and Applied Aspects of Seed Biology; Côme, D., Corbineau, F., Eds.; ASFIS: Paris, France, 1993; Volume 3, pp. 1033–1038.
- Chojnowski, M.; Corbineau, F.; Côme, D. Physiological and biochemical changes induced in sunflower seeds by osmopriming and subsequent drying, storage and aging. Seed Sci. Res. 1997, 7, 323–331.
- Bradford, K.J.; Côme, D.; Corbineau, F. Quantifying the oxygen sensitivity of seed germination using a population-based threshold model. Seed Sci. Res. 2007, 17, 33–43.
- Khan, A.A.; Peck, N.H.; Samimy, C. Seed osmoconditioning: Physiological and biochemical changes. Isr. J. Bot. 1980, 29, 133–144.
- Dell’Aquila, A.; Taranto, G. Cell division and DNA-synthesis during osmopriming treatment and following germination in aged wheat embryos. Seed Sci. Technol. 1986, 14, 333–341.
- Fujikura, Y.; Karssen, C.M. Effects of controlled deterioration and osmopriming on protein synthesis of cauliflower during early germination. Seed Sci. Sci. 1992, 2, 23–31.
- Van Pijlen, J.G.; Kraak, H.L.; Bino, R.J.; De Vos, C.H.R. Effects of ageing and osmopriming on germination characteristics and chromosome aberrations of tomato. Seed Sci. Technol. 1995, 29, 823–830.
- Bailly, C.; Benamar, A.; Corbineau, F.; Côme, D. Free radical scavenging as affected by accelerated ageing and subsequent priming in sunflower seeds. Physiol. Plant. 1998, 104, 646–652.
- Georghiou, K.; Thanos, C.A.; Passam, H.C. Osmoconditioning as a means of counteracting the ageing of pepper seeds during high temperature storage. Ann. Bot. 1987, 60, 279–285.
- Côme, D.; Corbineau, F. Dictionnaire de la Biologie des Semences et des Plantules; Lavoisier: Paris, France, 2006; p. 226.
