This review focused on current challenges and future directions on the treatment and prevention of ovarian failure or infertility by chemotherapy in young reproductive women with cancer. We also address current knowledge on chemotherapy-induced ovarian toxicity and its mechanisms. We hope this review will help clinicians to prevent and treat girls and young women with cancer who desire to preserve their ovarian endocrine function and fertility.
- gonadotoxicity
- fertility preservation
- embryo cryopreservation
- oocyte cryopreservation
- ovarian tissue cryopreservation
- oocyte in vitro maturation
- artificial ovaries
- stem cell technologies
- ovarian suppression
- oncofertility
1. Introduction
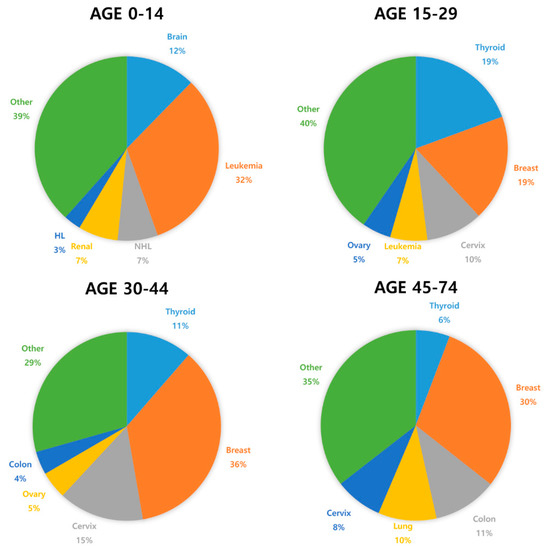
Cancer incidence is rapidly growing worldwide. In 2018, 8.6 million women were diagnosed with cancer globally [1]. Most women diagnosed with cancer are older, but 10% are <45 years of age [2]. Due to advances in cancer diagnosis and treatment, the survival rate for prepubertal and young women with cancer has significantly improved. In Europe, the five-year-survival rate is 79.1% in children diagnosed with cancer [3]. However, aggressive chemotherapy can cause impairment of reproductive functions and even fertility loss [4,5,6,7]. Although depletion of ovarian function is associated with improved survival outcomes in breast cancer patients of reproductive age, it has several side effects, such as hot flashes, osteoporosis, and sexual dysfunction [8]. Cardiovascular disease is the main cause of shortened life expectancy in women with premature ovarian insufficiency (POI) [9]. Moreover, chemotherapy-related POI and infertility may be associated with increased risk of neuro-degenerating disease and psychosocial distress [9].
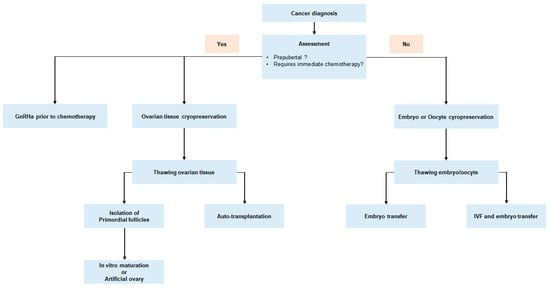
In recent years, interest in fertility preservation has increased significantly among female cancer patients [10]. Despite the huge interest cancer patients have with respect to preserving fertility, there is an unmet need in children and young cancer survivors [11]. Oncofertility is a relatively innovative concept that describes a multidisciplinary network of experts focused on developing and providing the option of fertility preservation to young cancer patients. Currently, embryo and oocyte cryopreservation are the only established methods for fertility preservation [12]. However, there is accumulating evidence for other experimental techniques including ovarian tissue cryopreservation, artificial ovaries, and in vitro maturation [13].
2. The Effect of Chemotherapy on Ovarian Function
2.1. Risk of Ovarian Toxicity Due to Chemotherapy Agents
| Diagnosis | Chemotherapy Protocol | Risk of Iatrogenic POI |
|---|---|---|
| Non-Hodgkin lymphoma |
Cyclophosphamide, hydroxydaunorubicin, oncovin, and prednisone (CHOP) (four to six cycles) Rituximab, cyclophosphamide, hydroxydaunorubicin, oncovin, and prednisone (R-CHOP) (four to six cycles) |
<20% [3,28] |
| Hodgkin lymphoma |
Adriamycin, bleomycin, vinblastine, and dacarbazine (ABVD) |
<20% [3] |
| Mustargen, oncovin, prednisone, and procarbazine (MOPP) |
10–50% [29,30,31,32] | |
| Bleomycin, etoposide, adriamycin, cyclophosphamide, oncovin, procarbazine, and prednisone (BEACOPP) (eight cycles) |
50–95% (age dependent) [33] | |
| Acute lympho- cytic leukemia |
Most standard chemotherapy protocols do not include a gonadotoxic multi-agent | <20% [3,13,14] |
| Acute myeloid leukemia |
Most standard chemotherapy protocols do not include gonadotoxic anthracycline/cytarabine | <20% [3,13,14] |
| Breast cancer | Cyclophosphamide, methotrexate, fluorouracil (CMF) (six cycles) Cyclophosphamide, epirubicin, fluorouracil (CEF) (six cycles) Cyclophosphamide, eoxorubicin (adriamycin), fluorouracil (CAF) (six cycles) |
>80% [3] (≥age 40) |
| 30–70% [3] (age 30–39) |
||
| Doxorubicin (adriamycin), cyclophosphamide (AC) (four cycles) |
30–70% [3] (≥age 40) |
|
| >20 [3] (age 30–39) |
||
| Others | Cyclophosphamide ≥ 7 g/m2 in females < 20 years Cyclophosphamide ≥ 5 g/m2 in females > 40 years Any alkylating agent (e.g., cyclophosphamide, ifosfamide, busulfan, carmustine, lomustine) |
>80% [34,35] |
| Cyclophosphamide ≥ 5 g/m2 in females 30–40 years | 30–70% [34,35] | |
| Taxanes Oxaliplatin Irinotecan Monoclonal antibodies (trastuzumab, bevacizumab, cetuximab) Tyrosine kinase inhibitors (erlotinib, imatinib) |
Unknown |
2.2. Mechanisms of Ovarian Toxicity
Gonadotoxic chemotherapy leads to primordial follicle loss, resulting in POI and infertility. Both direct acute and indirect delayed mechanisms have been reported for the effects of anticancer agents that cause a decrease in ovarian reserve. The main mechanism is that anticancer drugs directly induce DNA double-strand breaks (DSBs), which activate apoptosis and/or autophagy-related pathways [36,37,38,39,40,41,42]. The second mechanism is that anticancer drugs can indirectly cause primordial follicle depletion by microvascular and stromal injury through ischemia, necrosis, or inflammation [38,42,43,44,45]. There is third hypothesis called the “burnout” effect. A few studies have shown that anticancer drugs induce activation of the phosphoinositide 3-kinase/protein kinase B/forkhead box protein O3a (PI3K/AKT/FOXO3a) pathway, which leads to follicle reduction by massive activation of primordial follicles in mice and cultured human ovarian tissue [36,46,47,48,49]. However, there is some question of methodology and biological mechanism of follicle loss based on studies supporting “burnout theory”. It has not been proven that primordial follicle growth is the main cause of chemotherapy-induced primordial follicle loss. Thus, the “burnout” theory of chemotherapy-induced follicle depletion is still lacking evidence and is under debate [36,46]. The main cause of the follicle depletion induced by chemotherapy seems to be DNA double-strand breaks and apoptosis.
3. Improving Oncofertility Care
This entry is adapted from the peer-reviewed paper 10.3390/ijms21207792
