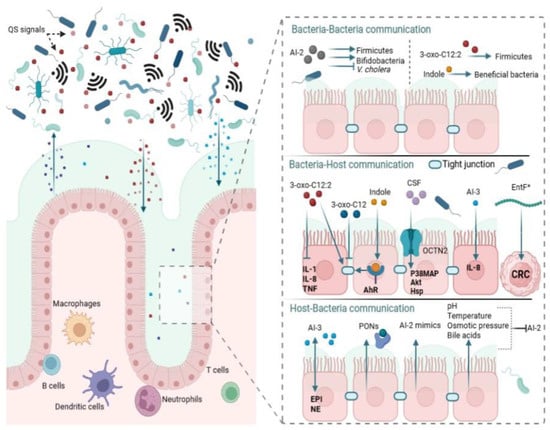
Gut bacteria were initially thought to be eavesdropping on AHLs synthesized within the complex gut microbial ecosystem [
]. However, available evidence, recently obtained using highly sensitive and sophisticated technologies, indicates the existence of AHL signaling among native gut residents [
]. The discovery of AHL signaling in the gut which is linked to normobiosis is one of the significant findings in understanding the human gut. In the human fecal samples from patients with inflammatory bowel disease (IBD) and healthy individuals, 14 distinct AHLs were profiled, one of which was prominent, identified as 3-oxo-C12:2-HSL [
].
A growing body of knowledge shows that interspecies interactions mediated by Al-2 play a vital role in gut bacterial composition and balance. By using engineered
E. coli strains that manipulate AI-2 levels by either raising or lowering the concentration of AI-2 in the gut, Thompson et al. evaluated the effect of the QS molecule on gut microbiota equilibrium [
145]. In their studies, the researchers found that the antibiotic treatment lowered Firmicutes and other members carrying the
luxS gene, indicating that AI-2 levels were reduced. Bacteroidetes were shown to dominate the microbiota following antibiotic-induced dysbiosis, possibly due to competitive advantage acquired from resistance caused by spontaneous mutations [
146].
One of the crucial interspecies QS molecules produced by some bacteria is indole, an amino acid-derived metabolite [
108,
130]. At indole concentrations below 1 mM,
E. coli was reported to only exhibit a repellent response, but switched to an attraction response when the indole concentration was at 1 mM or more [
158]. Yang et al. suggest that indole may prevent pathogen invasion with a repulsion mechanism while bringing beneficial resident bacterial species together and enhancing their proliferation [
158]. Through differential adaptation, gut bacteria in the presence of indole can suppress pathogen expansion. Enteropathogenic
E. coli (EPEC) motility, epithelial cell adhesion, biofilm formation, and virulence gene expression were all reported to be reduced in the presence of indole [
159,
160,
161]. Usually, co-infections with two or more infectious agents, such as
V. cholerae and EPEC, have been observed in diarrheal samples [
162].
5.3. Interkingdom QS and Normobiosis
The most documented inter-kingdom communications (host–bacteria interactions) are the ones driven by AHL molecules from pathogens [
166]. Hosts adjust accordingly by monitoring AHLs within the gut ecosystem and resisting infection by interfering with QS signal transduction [
92]. Enteric bacterial pathogens use host stress hormones to their advantage. Although the exact mechanisms behind microbiota and hormonal signaling are as yet unknown, hormones released during host stress can affect the host–bacteria interactions, bacterial pathogenicity, and vulnerability to infection [
51]. These stress hormones may be exploited by enteric pathogens as signaling molecules to modulate their virulence genes [
52]. The growth and motility of pathogenic bacteria like
Helicobacter pylori [
167],
Vibrio spp. [
128,
168],
Klebsiella pneumoniae,
P. aeruginosa,
E. coli, and
Staphylococcus aureus [
169] have been found to be modulated by catecholamines. Catecholamines have also been reported to increase bacterial virulence [
170]. In
P. aeruginosa PA14, virulence appears to be induced via the
las QS pathway following norepinephrine treatment [
171].Another important finding is the release of AI-2 mimics by human epithelial cells that are recognized by bacterial AI-2 receptors [
95]. Although the mechanism of AI-2 mimic synthesis is still poorly understood, the data currently available indicate that AI-2 mimic activity is stimulated when epithelial cells are exposed to bacteria, either directly or indirectly, suggesting that one or more secreted bacterial components induce AI-2 mimic synthesis [
95]. One can compare pathogen interactions with their host as an “arms race” in which each player continuously responds to the other’s changing tactics [
92]. The human aryl hydrocarbon receptor (AhR), a protein well-known for its function in mediating toxicity [
184], has been shown to interact with several QS molecules (such as 3-oxo-C12-HSL, C4-HSL, and PQS) produced by
P. aeruginosa to keep track of the bacterial infection at various stages [
92]. Such eavesdropping helps the host adapt to changes in the gut flora.
Compared to other QS signaling molecules, much less research has been done on the impact of AIP on gut microbiota balance. However, available data suggest the importance of host–bacteria interaction via AIP in gut microbial balance (
Figure 1). An AIP from
Bacillus subtilis, also known as competence and sporulation factor (CSF), was shown to contribute to normobiosis by mediating inter-kingdom signaling [
193].
B. subtilis was previously thought to be just soil bacteria, however, it is now confirmed to be a member of the human gut and evolved to exist there [
194,
195].
6. Conclusions
Today, QS is broadened to include multimodal communication, encompassing intraspecies, interspecies, and interkingdom signaling. The role of QS in normobiosis is undeniable. Via QS, gut microbiota maintains balance by suppressing pathogen expansion through enhancing biofilm formation and fitness of resident gut bacteria, mobilizing native members to reestablish balance following substance-induced dysbiosis, exerting anti-inflammatory response as well as preserving the tight junction integrity. Although gut microbiota research has just scratched the surface, exciting prospects exist for QS-based therapeutic interventions. With the advancement in technology, more and more tools are made available to further clarify the role of QS in normobiosis and elucidate the connection between QS inhibition and dysbiosis.