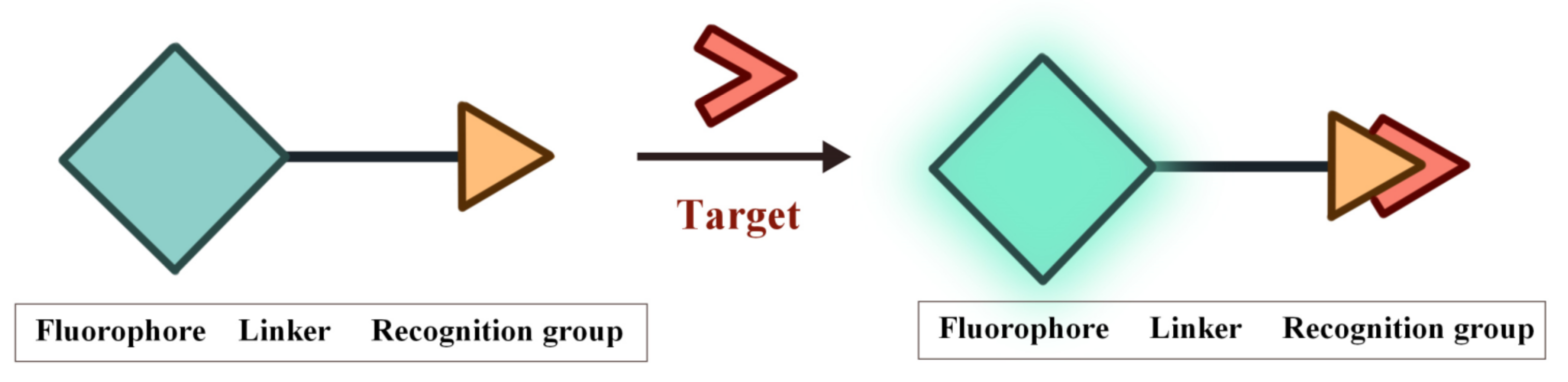
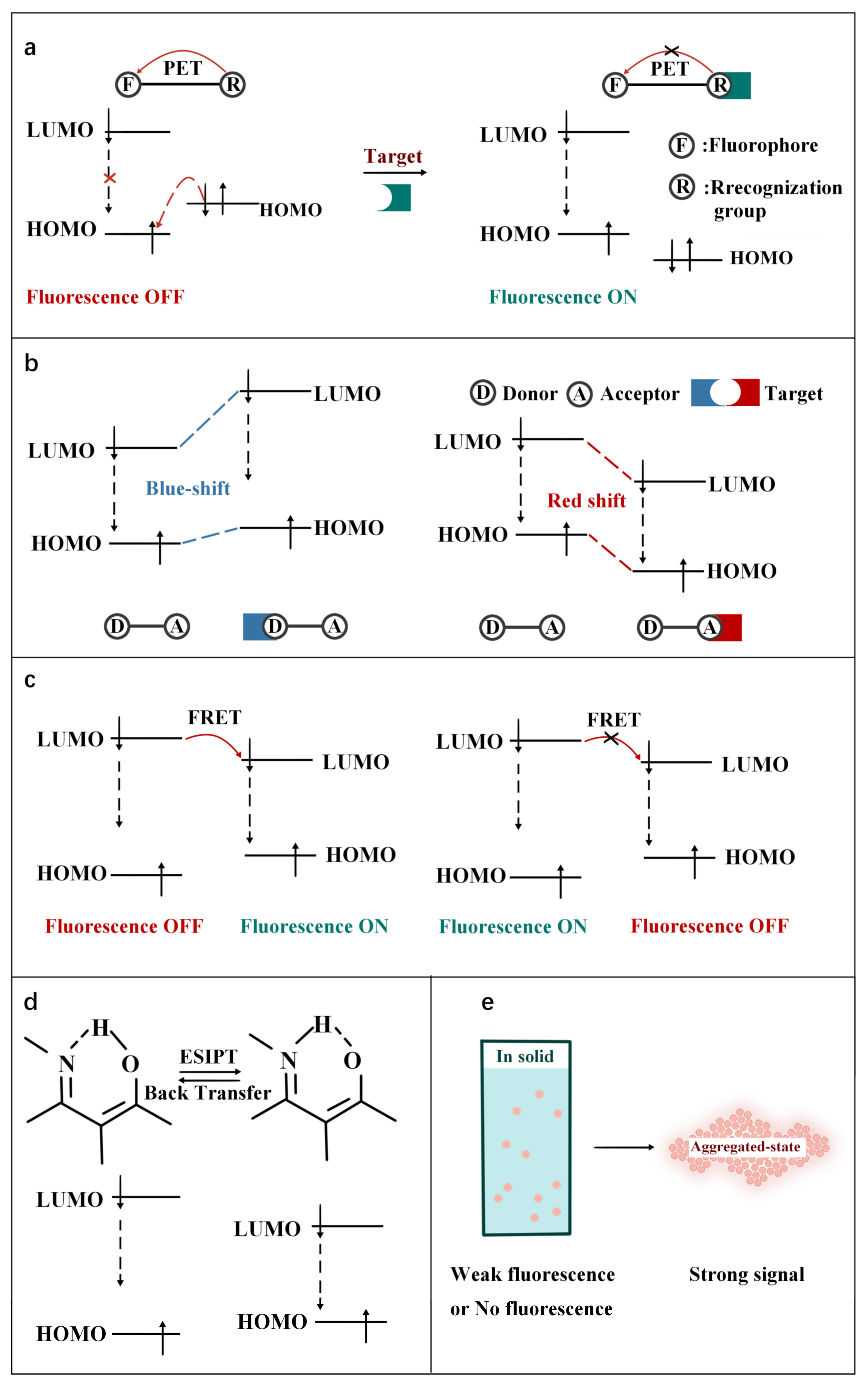
Fluorescence imaging technology provides a visual tool for medicine, showing great potential in the fields of molecular biology, cellular immunology and oncology. In recent years, organic fluorescent probes have attracted much attention in the bioanalytical field. Among various organic fluorescent probes, fluorescent organic small molecule probes (FOSMPs) have become a research hotspot due to their excellent physicochemical properties, such as good photostability, high spatial and temporal resolution, as well as excellent biocompatibility. FOSMPs have proved to be suitable for in vivo bioimaging and detection.
- fluorescent organic small molecules
- bioimaging
- detection
- recognition mechanisms
- fluorescent organic nanoparticles
1. Introduction
2. FOSMPs for Bioimaging and Detection
| Probe | λex/λem (nm) | Mechanism | Analyte | Solvent System | Linear Detection Range | Limit of Detection (LOD) | Target | Ref. |
|---|---|---|---|---|---|---|---|---|
| Probe | 500/525 | PET | Cys | CH3CN-Water-HEPES buffer | 0–100 μM | 3.7 × 10−2 μM | A375 cells | [59] |
| NP-S | 455/551 | PET | H2S | PBS buffer-EtOH | 30–300 μM | 0.376 μM | Mouse liver slices | [60] |
| Tyro1, Tyro2 | 455/560 | PET | Tyrosinase | Potassium phosphate buffer | - | - | B16F10 cells | [61] |
| NFP-G, NFP-A | 440/541 | PET | Formaldehyde (FA) | DMSO-PBS buffer | 0–30 μM, 0–15 μM | 1.2 μM, 0.18 μM | HepG-2 cells | [62] |
| Naphthalimide chromophore | 400/502 | ICT | CN− | HEPES buffer -CH3OH | 0–15 μM | 0.066 μM | HepG2 cells | [63] |
| DCM-β-gal | 535/685 | ICT | β-galactosidase (β-gal) | PBS buffer-DMSO | 0–12 U/L | 0.17 U/L | 293T cells | [64] |
| DCDHF-Glu | 510/613 | ICT | γ-Glutamyltranspeptidase (GGT) | PBS buffer | 0–40 U/L | 0.0379 U/L | HepG2 cells, LO2 cells | [65] |
| SHC | 370/540 | ICT | hNQO1 | PBS buffer | 0–0.8 μM | 0.0146 μM | HT-29 cells, MDA-MB-468 cells | [66] |
| AI | 370/495 | ICT | Hypochlorous acid (HOCl) |
DMSO-PBS buffer | 0–50 μM | 0.84 μM | HeLa cells, Nude mice | [67] |
| P-ONOO− | 365/480 | ICT | ONOO− | DMSO-PBS buffer | 0.429–3.0 µM | 0.0104 µM | HeLa cells | [68] |
| SR400, SR550 | 400/525, 550/675 | FRET | H2S | PBS buffer | - | - | HEK293 cells | [69] |
| CF | 415/517 | FRET | HNO | PBS buffer | 0–100 μM | 1.4 μM | HeLa cells | [70] |
| FIP-1 | 515/556 | FRET | Fe(II) | HEPES buffer | - | - | HEK293 cells, MDAMB-231 cells | [71] |
| PNCy3Cy5 | 530/660 | FRET | OONO– | Phosphate buffer-DMF | 0–0.7 μM | 6.5 × 10−4 μM | RAW264.7 | [72] |
| FTR-βgal | 450/540 | FRET | β-gal | PBS buffer-EtOH | 0–5 U/L | 4.11 × 10−8 U/L | Hek293 cells | [73] |
| PPA | 400/511 | ESIPT | Palladium | CH3CN-PBS buffer | 0–180 μM | 0.028 μM | A549 cells | [74] |
| Py-GSH | 488/545 | ESIPT | GGT | PBS buffer | 1–30 U/L | 1 × 10−2 U/L | SKOV3 cells, HOSEpiC cells, Tumor-bearing mice, Human specimens |
[75] |
| NIR-TS | 550/836 | ESIPT | SO2 | Water | 0.5–40 μM | 0.067 μM | HeLa cells, Mice | [76] |
| TPE-Gal | 344/512 | AIE | β-gal | PBS buffer | 8 × 10−4–4.8 × 10−3 U/L | 3.3 × 10−4 U/L | HeLa cells, OVCAR-3 cells | [77] |
| TT | 320/440–550 | AIE | H2O2 | DMSO-Water | - | - | RAW264.7 cells, HLF cells | [78] |
| QP-DNP | 482/582 | AIE | Hydrazine | DMSO-HEPES buffer | 0–0.8 μM | 0.055 μM | HeLa cells, Kunming mouse | [79] |
| AIE-Lyso-1 | 356/532 | AIE/ESIPT | Esterase | DMSO–Water | 100–500 U/L | 2.4 U/L | MCF-7 cells | [80] |
| Probe 1 | 314/446 | PET/ICT | Cys | PBS buffer | 0.2–2.0 μM | 1.4 × 10−3 μM | HeLa cells | [81] |
| NCQ | 423/490, 544 423/490423/490, 624 |
Triple-emission | Cys/Hcy, GSH/H2S, thiopheno(PhSH) |
PBS buffer-acetonitrile | 0–10 μM/10 μM, 0–6 μM/8 μM, 0–70 μM |
0.57 μM/0.65 μM, 0.49 μM/0.52 μM, 0.34 μM |
HeLa cells | [82] |
3. FOSMPs-Based FONPs for Bioimaging and Detection
This entry is adapted from the peer-reviewed paper 10.3390/molecules27238421
References
- Hao, J.; Yan, B. A water-stable lanthanide-functionalized MOF as a highly selective and sensitive fluorescent probe for Cd2+. Chem. Commun. 2015, 51, 7737–7740.
- Donia, M.S.; Fischbach, M.A. Small molecules from the human microbiota. Science 2015, 349, 1254766.
- Fukuto, J.M.; Carrington, S.J.; Tantillo, D.J.; Harrison, J.G.; Ignarro, L.J.; Freeman, B.A.; Chen, A.; Wink, D.A. Small Molecule Signaling Agents: The Integrated Chemistry and Biochemistry of Nitrogen Oxides, Oxides of Carbon, Dioxygen, Hydrogen Sulfide, and Their Derived Species. Chem. Res. Toxicol. 2012, 25, 769–793.
- Griendling, K.K.; Touyz, R.M.; Zweier, J.L.; Dikalov, S.; Chilian, W.; Chen, Y.; Harrison, D.G.; Bhatnagar, A.; Amer, H.A.C.B. Measurement of Reactive Oxygen Species, Reactive Nitrogen Species, and Redox-Dependent Signaling in the Cardiovascular System: A Scientific Statement From the American Heart Association. Circ. Res. 2016, 119, E39–E75.
- Jiao, X.; Li, Y.; Niu, J.; Xie, X.; Wang, X.; Tang, B. Small-Molecule Fluorescent Probes for Imaging and Detection of Reactive Oxygen, Nitrogen, and Sulfur Species in Biological Systems. Anal. Chem. 2018, 90, 533–555.
- Kalyanaraman, B. Teaching the basics of redox biology to medical and graduate students: Oxidants, antioxidants and disease mechanisms. Redox Biol. 2013, 1, 244–257.
- Figueira, T.R.; Barros, M.H.; Camargo, A.A.; Castilho, R.F.; Ferreira, J.C.B.; Kowaltowski, A.J.; Sluse, F.E.; Souza-Pinto, N.C.; Vercesi, A.E. Mitochondria as a Source of Reactive Oxygen and Nitrogen Species: From Molecular Mechanisms to Human Health. Antioxid. Redox Sign. 2013, 18, 2029–2074.
- Fransen, M.; Nordgren, M.; Wang, B.; Apanasets, O. Role of peroxisomes in ROS/RNS-metabolism: Implications for human disease. Bba.-Mol. Basis Dis. 2012, 1822, 1363–1373.
- Mehta, V.N.; Desai, M.L.; Basu, H.; Kumar Singhal, R.; Kailasa, S.K. Recent developments on fluorescent hybrid nanomaterials for metal ions sensing and bioimaging applications: A review. J. Mol. Liq. 2021, 333, 115950.
- Huang, J.; Jones, A.; Waite, T.D.; Chen, Y.; Huang, X.; Rosso, K.M.; Kappler, A.; Mansor, M.; Tratnyek, P.G.; Zhang, H. Fe(II) Redox Chemistry in the Environment. Chem. Rev. 2021, 121, 8161–8233.
- Kim, S.; Noh, J.Y.; Kim, K.Y.; Kim, J.H.; Kang, H.K.; Nam, S.; Kim, S.H.; Park, S.; Kim, C.; Kim, J. Salicylimine-Based Fluorescent Chemosensor for Aluminum Ions and Application to Bioimaging. Inorg. Chem. 2012, 51, 3597–3602.
- Kumar, A.; Palfrey, H.A.; Pathak, R.; Kadowitz, P.J.; Gettys, T.W.; Murthy, S.N. The metabolism and significance of homocysteine in nutrition and health. Nutr. Metab. 2017, 14, 78.
- Skovierova, H.; Vidomanova, E.; Mahmood, S.; Sopkova, J.; Drgova, A.; Cervenova, T.; Halasova, E.; Lehotsky, J. The Molecular and Cellular Effect of Homocysteine Metabolism Imbalance on Human Health. Int. J. Mol. Sci. 2016, 17, 1733.
- Poole, L.B. The basics of thiols and cysteines in redox biology and chemistry. Free Radical Bio. Med. 2015, 80, 148–157.
- Dao, N.V.; Ercole, F.; Urquhart, M.C.; Kaminskas, L.M.; Nowell, C.J.; Davis, T.P.; Sloan, E.K.; Whittaker, M.R.; Quinn, J.F. Trisulfide linked cholesteryl PEG conjugate attenuates intracellular ROS and collagen-1 production in a breast cancer co-culture model. Biomater. Sci. UK 2021, 9, 835–846.
- Chen, X.; Wang, F.; Hyun, J.Y.; Wei, T.; Qiang, J.; Ren, X.; Shin, I.; Yoon, J. Recent progress in the development of fluorescent, luminescent and colorimetric probes for detection of reactive oxygen and nitrogen species. Chem. Soc. Rev. 2016, 45, 2976–3016.
- Graves, D.B. The emerging role of reactive oxygen and nitrogen species in redox biology and some implications for plasma applications to medicine and biology. J. Phys. D Appl. Phys. 2012, 45, 263001.
- Van-Nghia, N.; Ha, J.; Cho, M.; Li, H.; Swamy, K.M.K.; Yoon, J. Recent developments of BODIPY-based colorimetric and fluorescent probes for the detection of reactive oxygen/nitrogen species and cancer diagnosis. Coordin. Chem. Rev. 2021, 439, 213936.
- Ogawa, M.; Takakura, H. In Vivo Molecular Imaging for Biomedical Analysis and Therapies. Anal. Sci. 2018, 34, 273–281.
- Peng, F. Recent Advances in Cancer Imaging with (CuCl2)-Cu-64 PET/CT. Nucl. Med. Mol. Imaging. 2022, 56, 80–85.
- Yan, Y.; Wang, H.; Zhao, Y. Radiolabeled peptide probe for tumor imaging. Chinese Chem. Lett. 2022, 33, 3361–3370.
- Seetharam Bhat, K.R.; Samavedi, S.; Moschovas, M.C.; Onol, F.F.; Roof, S.; Rogers, T.; Patel, V.R.; Sivaraman, A. Magnetic resonance imaging-guided prostate biopsy—A review of literature. Asian J. Urol. 2021, 8, 105–116.
- Kitada, N.; Maki, S.; Kim, S. Visible Light Bioluminescence Imaging Platform for Animal Cell Imaging. Methods Mol. Biol. 2022, 2524, 37–51.
- Komatsu, H.; Kobayashi, E.; Gonzalez, N.; Rawson, J.; Ortiz, J.; Donohue, C.; Ku, H.T.; Kandeel, F.; Mullen, Y. Critical Considerations in Bioluminescence Imaging of Transplanted Islets Dynamic Signal Change in Early Posttransplant Phase and Signal Absorption by Tissues. Pancreas 2022, 51, 234–242.
- Sato, K. Bioluminescence Imaging for Evaluation of Antitumor Effect In Vitro and In Vivo in Mice Xenografted Tumor Models. Methods Mol. Biol. 2022, 2524, 307–315.
- Pan, X.; Gao, A.; Lin, Z. Fluorescence imaging of tumor immune contexture in immune checkpoint blockade therapy. Int. Immunopharmacol. 2022, 106, 108617.
- Wu, X.; Li, H.; Lee, E.; Yoon, J. Sensors for In Situ Real-Time Fluorescence Imaging of Enzymes. Chem 2020, 6, 2893–2901.
- Zhu, H.; Hamachi, I. Fluorescence imaging of drug target proteins using chemical probes. J. Pharm. Sci. 2020, 10, 426–433.
- van Manen, L.; Handgraaf, H.J.M.; Diana, M.; Dijkstra, J.; Ishizawa, T.; Vahrmeijer, A.L.; Mieog, J.S.D. A practical guide for the use of indocyanine green and methylene blue in fluorescence-guided abdominal surgery. J. Surg. Oncol. 2018, 118, 283–300.
- Wang, J.; Li, C.; Jiang, G. Progress in Fluorescent Probes for Aminopeptidase N. Chem. Eur. J. 2018, 81, 972–980.
- Xu, Y.; Liu, R.; Xu, K.; Dai, Z. Fluorescent Probes for Intraoperative Navigation. Prog. Chem. 2021, 33, 52–65.
- Yagishita, A.; Ueno, T.; Tsuchihara, K.; Urano, Y. Amino BODIPY-Based Blue Fluorescent Probes for Aldehyde Dehydrogenase 1-Expressing Cells. Bioconjugate Chem. 2021, 32, 234–238.
- Han, H.; Zhong, Y.; He, C.; Fu, L.; Huang, Q.; Kuang, Y.; Yi, X.; Zeng, W.; Zhong, H.; Yang, M. Recent advances in organic fluorescent probes for tumor related enzyme detection. Dyes Pigm. 2022, 204, 110386.
- Pak, Y.L.; Swamy, K.; Yoon, J. Recent Progress in Fluorescent Imaging Probes. Sensors 2015, 15, 24374–24396.
- Shaya, J.; Corridon, P.R.; Al-Omari, B.; Aoudi, A.; Shunnar, A.; Mohideen, M.I.H.; Qurashi, A.; Michel, B.Y.; Burger, A. Design, photophysical properties, and applications of fluorene-based fluorophores in two-photon fluorescence bioimaging: A review. J. Photoch. Photobio. C. 2022, 52, 100529.
- Terai, T.; Nagano, T. Fluorescent probes for bioimaging applications. Curr. Opin. Chem. Biol. 2008, 12, 515–521.
- Balleza, E.; Kim, J.M.; Cluzel, P. Systematic characterization of maturation time of fluorescent proteins in living cells. Nat. Methods 2018, 15, 47.
- Han, Y.; Wang, X.; Li, S. Biocompatible Europium Doped Hydroxyapatite Nanoparticles as a Biological Fluorescent Probe. Curr. Nanosci. 2010, 6, 178–183.
- Shore, A. A review on fluorescent inorganic nanoparticles for optical sensing applications (vol 6, pg 21624, 2016). RSC Adv. 2019, 9, 16565.
- Wu, C.; Chiu, D.T. Highly Fluorescent Semiconducting Polymer Dots for Biology and Medicine. Angew. Chem. Int. Edit. 2013, 52, 3086–3109.
- Zhang, J.; Huang, Y.; Wang, D.; Pollard, A.C.; Chen, Z.G.; Egap, E. Triblock near-infrared fluorescent polymer semiconductor nanoparticles for targeted imaging. J. Mater. Chem. C. 2017, 5, 5685–5692.
- Chan, J.; Dodani, S.C.; Chang, C.J. Reaction-based small-molecule fluorescent probes for chemoselective bioimaging. Nat. Chem. 2012, 4, 973–984.
- Gu, B.; Zhang, Q. Recent Advances on Functionalized Upconversion Nanoparticles for Detection of Small Molecules and Ions in Biosystems. Adv. Sci. 2018, 5, 1700609.
- Berhanu, A.L.; Gaurav; Mohiuddin, I.; Malik, A.K.; Aulakh, J.S.; Kumar, V.; Kim, K. A review of the applications of Schiff bases as optical chemical sensors. TrAC, Trends Anal. Chem. 2019, 116, 74–91.
- Immanuel David, C.; Prabakaran, G.; Nandhakumar, R. Recent approaches of 2HN derived fluorophores on recognition of Al3+ ions: A review for future outlook. Microchem. J. 2021, 169, 106590.
- Zoubi, W.A. Biological Activities of Schiff Bases and Their Complexes: A Review of Recent Works. Int. J. Org. Chem. 2013, 3, 24.
- Han, X.; Wang, Y.; Huang, Y.; Wang, X.; Choo, J.; Chen, L. Fluorescent probes for biomolecule detection under environmental stress. J. Hazard. Mater. 2022, 431, 128527.
- Zhang, Y.; Hao, Y.; Ma, X.; Chen, S.; Xu, M. A dicyanoisophorone-based highly sensitive and selective near-infrared fluorescent probe for sensing thiophenol in water samples and living cells. Environ. Pollut. 2020, 265, 114958.
- Lai, X.; Qiu, G.; Ye, Q.; Wang, R.; Liu, J. A reaction-based fluorescent probe for detecting o-phenylenediamine in water and lateritic soil samples. J. Photochem. Photobiol. A. 2020, 386, 112101.
- Suratsawadee, A.; Wangmo, L.; Ratvijitvech, T.; Siripinyanond, A. A spoilage indicator card based on distance-based color change of paper impregnated with acid-base indicator for freshness monitoring of shrimp. Microchem. J. 2022, 175, 107110.
- Zhong, K.; Zhou, S.; Yan, X.; Li, X.; Hou, S.; Cheng, L.; Gao, X.; Li, Y.; Tang, L. A simple H2S fluorescent probe with long wavelength emission: Application in water, wine, living cells and detection of H2S gas. Dyes Pigment. 2020, 174, 108049.
- Choudhury, P.; Das, P.K. Progress and trends in self-assembly driven fluorescent organic nanoparticles: A brief overview. J. Indian Chem. Soc. 2021, 98, 100123.
- Li, J.; Yim, D.; Jang, W.; Yoon, J. Recent progress in the design and applications of fluorescence probes containing crown ethers. Chem. Soc. Rev. 2017, 46, 2437–2458.
- Liu, Z.; Zhou, X.; Miao, Y.; Hu, Y.; Kwon, N.; Wu, X.; Yoon, J. A Reversible Fluorescent Probe for Real-Time Quantitative Monitoring of Cellular Glutathione. Angew. Chem. Int. Edit. 2017, 56, 5812–5816.
- Niu, L.; Chen, Y.; Zheng, H.; Wu, L.; Tung, C.; Yang, Q. Design strategies of fluorescent probes for selective detection among biothiols. Chem. Soc. Rev. 2015, 44, 6143–6160.
- Park, S.; Kwon, N.; Lee, J.; Yoon, J.; Shin, I. Synthetic ratiometric fluorescent probes for detection of ions. Chem. Soc. Rev. 2020, 49, 143–179.
- Wang, Y.; Zhang, L.; Han, X.; Zhang, L.; Wang, X.; Chen, L. Fluorescent probe for mercury ion imaging analysis: Strategies and applications. Chem. Eng. J. 2021, 406, 127166.
- Zhang, J.; Wang, N.; Ji, X.; Tao, Y.; Wang, J.; Zhao, W. BODIPY-Based Fluorescent Probes for Biothiols. Chem.-Eur. J. 2020, 26, 4172–4192.
- Fan, W.; Huang, X.; Shi, X.; Wang, Z.; Lu, Z.; Fan, C.; Bo, Q. A simple fluorescent probe for sensing cysteine over homocysteine and glutathione based on PET. Spectrochim. Acta A Mol. Biomol. Spectrosc. 2017, 173, 918–923.
- Xu, K.; He, L.; Yang, Y.; Lin, W. A PET-based turn-on fluorescent probe for sensitive detection of thiols and H2S and its bioimaging application in living cells, tissues and zebrafish. New J. Chem. 2019, 43, 2865–2869.
- Kumar, P.; Biswas, S.; Koner, A.L. Fast tyrosinase detection in early-stage melanoma with nanomolar sensitivity using a naphthalimide-based fluorescent read-out probe. New J. Chem. 2020, 44, 10771–10775.
- Zhou, L.; Cui, J.; Yu, Z.; Zou, D.; Zhang, W.; Qian, J. A β-d-galactose-guided fluorescent probe for selectively bioimaging endogenous formaldehyde in living HepG-2 cells. Sens. Actuators B Chem. 2021, 332, 129494.
- Xiong, K.; Huo, F.; Yin, C.; Yang, Y.; Chao, J.; Zhang, Y.; Xu, M. A off–on green fluorescent chemosensor for cyanide based on a hybrid coumarin–hemicyanine dye and its bioimaging. Sens. Actuators B Chem. 2015, 220, 822–828.
- Gu, K.; Xu, Y.; Li, H.; Guo, Z.; Zhu, S.; Zhu, S.; Shi, P.; James, T.D.; Tian, H.; Zhu, W. Real-Time Tracking and In Vivo Visualization of β-Galactosidase Activity in Colorectal Tumor with a Ratiometric Near-Infrared Fluorescent Probe. J. Am. Chem. Soc. 2016, 138, 5334–5340.
- Liu, F.; Wang, Z.; Wang, W.; Luo, J.; Kong, L. Red-Emitting Fluorescent Probe for Detection of γ-Glutamyltranspeptidase and Its Application of Real-Time Imaging under Oxidative Stress in Cells and in Vivo. Anal. Chem. 2018, 90, 7467–7473.
- Cho, M.K.; Lim, C.S.; Sarkar, A.R.; Lee, H.W.; Choi, H.J.; Noh, C.; Shin, S.J.; Kim, H.M. A two-photon ratiometric probe for detection of hNQO1 enzyme activity in human colon tissue. Sens. Actuators Chem. 2018, 272, 203–210.
- Shi, Y.; Huo, F.; Yin, C. Malononitrile as the ‘double-edged sword’ of passivation-activation regulating two ICT to highly sensitive and accurate ratiometric fluorescent detection for hypochlorous acid in biological system. Sens. Actuators Chem. 2020, 325, 128793.
- Yu, H.; Fang, Y.; Wang, J.; Zhang, Q.; Chen, S.; Wang, K.; Hu, Z. Enhancing probe’s sensitivity for peroxynitrite through alkoxy modification of dicyanovinylchromene. Anal. Bioanal. Chem. 2022, 414, 6779–6789.
- Wei, L.; Yi, L.; Song, F.; Wei, C.; Wang, B.; Xi, Z. FRET ratiometric probes reveal the chiral-sensitive cysteine-dependent H2S production and regulation in living cells. Sci. Rep. UK 2014, 4, 4521.
- Zhang, H.; Liu, R.; Tan, Y.; Xie, W.H.; Lei, H.; Cheung, H.; Sun, H. A FRET-based Ratiometric Fluorescent Probe for Nitroxyl Detection in Living Cells. ACS Appl. Mater. Inter. 2015, 7, 5438–5443.
- Aron, A.T.; Loehr, M.O.; Bogena, J.; Chang, C.J. An Endoperoxide Reactivity-Based FRET Probe for Ratiometric Fluorescence Imaging of Labile Iron Pools in Living Cells. J. Am. Chem. Soc. 2016, 138, 14338–14346.
- Jia, X.; Chen, Q.; Yang, Y.; Tang, Y.; Wang, R.; Xu, Y.; Zhu, W.; Qian, X. FRET-Based Mito-Specific Fluorescent Probe for Ratiometric Detection and Imaging of Endogenous Peroxynitrite: Dyad of Cy3 and Cy5. J. Am. Chem. Soc. 2016, 138, 10778–10781.
- Wei, X.; Hu, X.; Zhang, L.; Li, J.; Wang, J.; Wang, P.; Song, Z.; Zhang, J.; Yan, M.; Yu, J. Highly selective and sensitive FRET based ratiometric two-photon fluorescent probe for endogenous β-galactosidase detection in living cells and tissues. Microchem. J. 2020, 157, 105046.
- Shen, Y.; Zhang, X.; Wu, Y.; Zhang, Y.; Liu, X.; Chen, Y.; Li, H.; Zhong, Y. A lysosome targetable fluorescent probe for palladium species detection base on an ESIPT phthalimide derivative. Spectrochim. Acta A Mol. Biomol. Spectrosc. 2018, 205, 66–71.
- Zhou, X.; Liu, Y.; Liu, Q.; Yan, L.; Xue, M.; Yuan, W.; Shi, M.; Feng, W.; Xu, C.; Li, F. Point-of-care Ratiometric Fluorescence Imaging of Tissue for the Diagnosis of Ovarian Cancer. Theranostics 2019, 9, 4597–4607.
- Ren, H.; Huo, F.; Wu, X.; Liu, X.; Yin, C. An ESIPT-induced NIR fluorescent probe to visualize mitochondrial sulfur dioxide during oxidative stress in vivo. Chem. Commun. 2021, 57, 655–658.
- Jiang, G.; Zeng, G.; Zhu, W.; Li, Y.; Dong, X.; Zhang, G.; Fan, X.; Wang, J.; Wu, Y.; Tang, B.Z. A selective and light-up fluorescent probe for β-galactosidase activity detection and imaging in living cells based on an AIE tetraphenylethylene derivative. Chem. Commun. 2017, 53, 4505–4508.
- Cheng, Y.; Dai, J.; Sun, C.; Liu, R.; Zhai, T.; Lou, X.; Xia, F. An Intracellular H2O2-Responsive AIEgen for the Peroxidase-Mediated Selective Imaging and Inhibition of Inflammatory Cells. Angew. Chem. Int. Ed. 2018, 57, 3123–3127.
- Muthusamy, S.; Yin, S.; Rajalakshmi, K.; Meng, S.; Zhu, D.; Xie, M.; Xie, J.; Lodi, R.S.; Xu, Y. Development of a quinoline-derived turn-on fluorescent probe for real time detection of hydrazine and its applications in environment and bioimaging. Dyes Pigm. 2022, 206, 110618.
- Gao, M.; Hu, Q.; Feng, G.; Tang, B.Z.; Liu, B. A fluorescent light-up probe with “AIE + ESIPT” characteristics for specific detection of lysosomal esterase. J. Mater. Chem. B. 2014, 2, 3438–3442.
- Chen, X.; Xu, H.; Ma, S.; Tong, H.; Lou, K.; Wang, W. A simple two-photon turn-on fluorescent probe for the selective detection of cysteine based on a dual PeT/ICT mechanism. RSC Adv. 2018, 8, 13388–13392.
- Yang, L.; Su, Y.; Geng, Y.; Zhang, Y.; Ren, X.; He, L.; Song, X. A Triple-Emission Fluorescent Probe for Discriminatory Detection of Cysteine/Homocysteine, Glutathione/Hydrogen Sulfide, and Thiophenol in Living Cells. ACS Sens. 2018, 3, 1863–1869.
- Ning, Z.; Wu, S.; Liu, G.; Ji, Y.; Jia, L.; Niu, X.; Ma, R.; Zhang, Y.; Xing, G. Water-soluble AIE-Active Fluorescent Organic Nanoparticles: Design, Preparation and Application for Specific Detection of Cysteine over Homocysteine and Glutathione in Living Cells. Chem.-Asian J. 2019, 14, 2220–2224.
- Yan, L.; Zhang, Y.; Xu, B.; Tian, W. Fluorescent nanoparticles based on AIE fluorogens for bioimaging. Nanoscale 2016, 8, 2471–2487.
- Zhang, X.; Zhang, X.; Yang, B.; Zhang, Y.; Liu, M.; Liu, W.; Chen, Y.; Wei, Y. Fabrication of water-dispersible and biocompatible red fluorescent organic nanoparticles via PEGylation of aggregate induced emission enhancement dye and their cell imaging applications. Colloids Surf. B. 2014, 113, 435–441.
- Zhang, Y.; Chang, K.; Xu, B.; Chen, J.; Yan, L.; Ma, S.; Wu, C.; Tian, W. Highly efficient near-infrared organic dots based on novel AEE fluorogen for specific cancer cell imaging. RSC Adv. 2015, 5, 36837–36844.
- Zhao, Q.; Li, K.; Chen, S.; Qin, A.; Ding, D.; Zhang, S.; Liu, Y.; Liu, B.; Sun, J.Z.; Tang, B.Z. Aggregation-induced red-NIR emission organic nanoparticles as effective and photostable fluorescent probes for bioimaging. J. Mater. Chem. 2012, 22, 15128–15135.
- Li, K.; Qin, W.; Ding, D.; Tomczak, N.; Geng, J.; Liu, R.; Liu, J.; Zhang, X.; Liu, H.; Liu, B. Photostable fluorescent organic dots with aggregation-induced emission (AIE dots) for noninvasive long-term cell tracing. Sci. Rep. UK. 2013, 24, 635–643.
- Geng, J.; Zhu, Z.; Qin, W.; Ma, L.; Hu, Y.; Gurzadyan, G.G.; Tang, B.Z.; Liu, B. Near-infrared fluorescence amplified organic nanoparticles with aggregation-induced emission characteristics for in vivo imaging. Nanoscale 2014, 6, 939–945.
- Feng, G.; Tay, C.Y.; Chui, Q.X.; Liu, R.; Tomczak, N.; Liu, J.; Tang, B.Z.; Leong, D.T.; Liu, B. Ultrabright organic dots with aggregation-induced emission characteristics for cell tracking. Biomaterials 2014, 35, 8669–8677.
