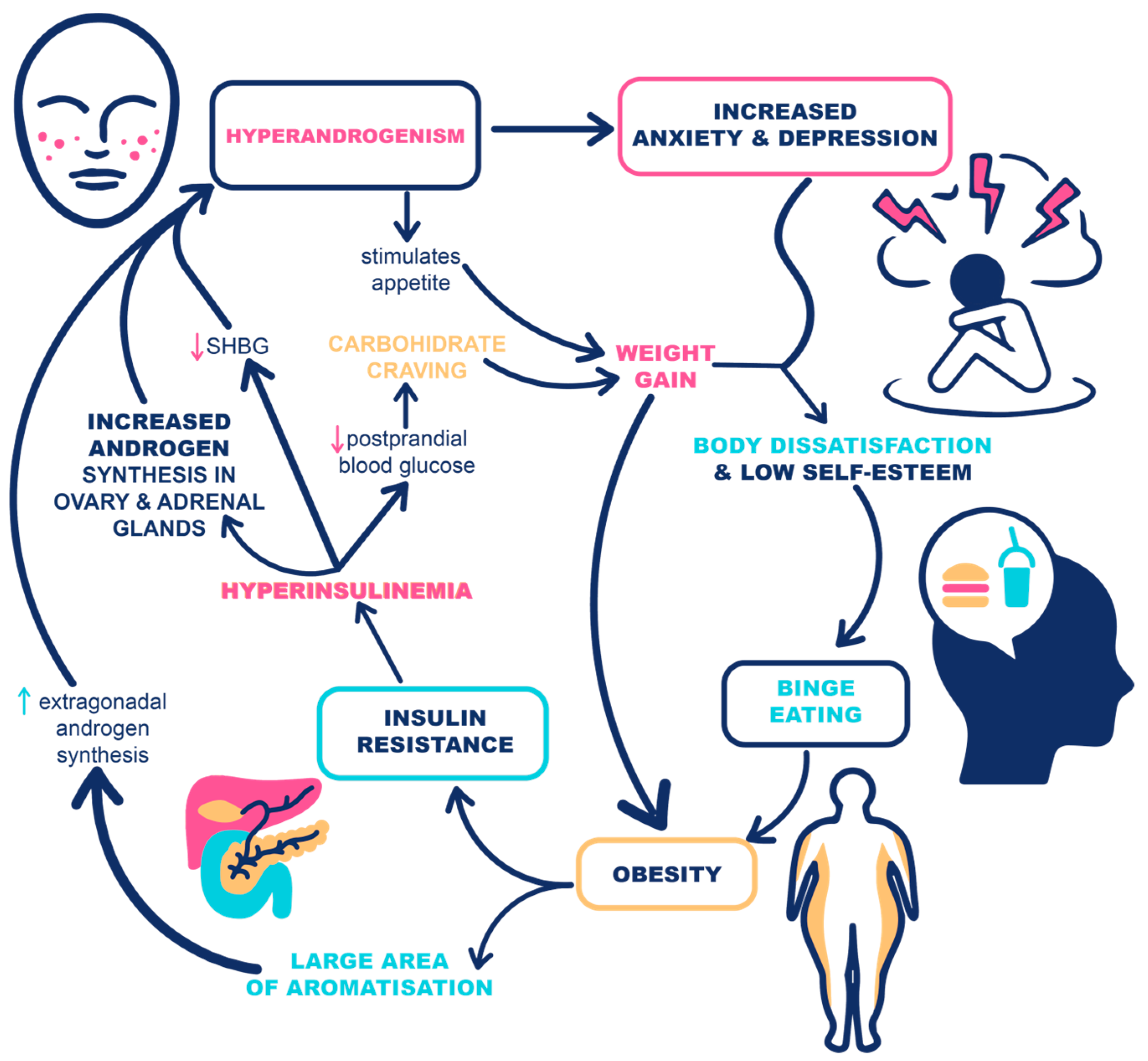
Polycystic ovary syndrome (PCOS) is a heterogeneous, multifactorial disease that leads to menstrual and ovulatory irregularities, infertility, hyperandrogenism, and metabolic disturbances. It is recognized as the most prevalent endocrine/metabolic disorder in reproductive-aged women. Despite continuous research efforts, the unifying pathophysiological mechanisms that could explain the etiology of this complex disorder are still not fully understood. The interplay between genetic and epigenetic factors, mitochondrial dysfunction, altered protein and miRNA profiles, and environmental factors that also contribute to obesity lead to exaggerated gonadotropin-releasing hormone (GnRH) pulsatility with hypersecretion of luteinizing hormone (LH) and insulin resistance (IR). Compensatory hyperinsulinemia adds to increased ovarian androgen production and impaired oocyte development. The essential role of the frequent coexistence of mental disorders and polycystic ovary syndrome (PCOS) is being increasingly recognized in the management of PCOS patients since it influences the success of weight loss interventions. Patients frequently experience disrupted eating behaviors, evidenced by the high prevalence of eating disorders in this population. Therefore, assessment and potential modification of eating disorders and eating-related behavior might be especially relevant to improve obesity treatment outcomes in this population, which remains the most efficient causal treatment in PCOS patients with high metabolic risk.
- polycystic ovary syndrome
- feeding and eating disorders
- eating behavior
- obesity
1. Eating Disorders in Polycystic Ovary Syndrome (PCOS)
1.1. Classification of Eating Disorders
1.2. Prevalence of Eating Disorders in PCOS
1.3. The Casual Link between Eating Disorders and PCOS
1.4. The Vicious Cycle of PCOS, Eating Disorders, and Obesity
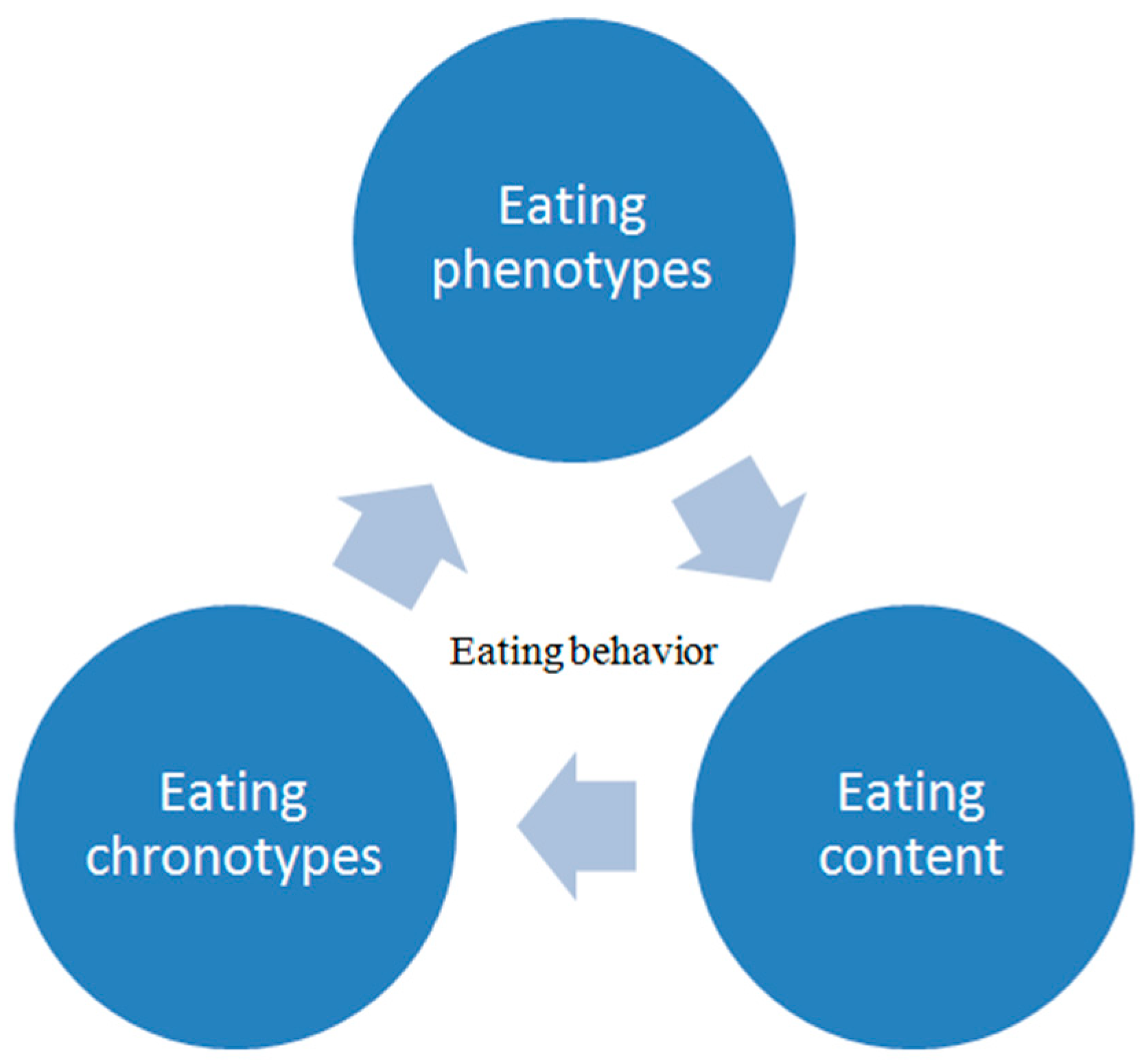
2. Eating Behavior
2.1. Regulation of Eating Behavior
2.2. Assessment of Eating Behavior
This entry is adapted from the peer-reviewed paper 10.3390/life12111906
References
- Treasure, J.; Duarte, T.A.; Schmidt, U. Eating disorders. Lancet 2020, 395, 899–911.
- Hollinrake, E.; Abreu, A.; Maifeld, M.; Van Voorhis, B.J.; Dokras, A. Increased risk of depressive disorders in women with polycystic ovary syndrome. Fertil. Steril. 2007, 87, 1369–1376.
- Månsson, M.; Holte, J.; Landin-Wilhelmsen, K.; Dahlgren, E.; Johansson, A.; Landén, M. Women with polycystic ovary syndrome are often depressed or anxious--a case control study. Psychoneuroendocrinology 2008, 33, 1132–1138.
- Tay, C.T.; Teede, H.J.; Hill, B.; Loxton, D.; Joham, A.E. Increased prevalence of eating disorders, low self-esteem, and psychological distress in women with polycystic ovary syndrome: A community-based cohort study. Fertil. Steril. 2019, 112, 353–361.
- Lee, I.; Cooney, L.G.; Saini, S.; Smith, M.E.; Sammel, M.D.; Allison, K.C.; Dokras, A. Increased risk of disordered eating in polycystic ovary syndrome. Fertil. Steril. 2017, 107, 796–802.
- Asdaq, S.M.B.; Jomah, S.; Hasan, R.; Al-Baroudi, D.; Alharbi, M.; Alsubaie, S.; Buhamad, M.H.; Alyahya, B.; Al-Yamani, M.J. Impact of polycystic ovary syndrome on eating behavior, depression and health related quality of life: A cross-sectional study in Riyadh. Saudi J. Biol. Sci. 2020, 27, 3342–3347.
- Thannickal, A.; Brutocao, C.; Alsawas, M.; Morrow, A.; Zaiem, F.; Murad, M.H.; Chattha, A.J. Eating, sleeping and sexual function disorders in women with polycystic ovary syndrome (PCOS): A systematic review and meta-analysis. Clin. Endocrinol. 2020, 92, 338–349.
- Morgan, J.; Scholtz, S.; Lacey, H.; Conway, G. The prevalence of eating disorders in women with facial hirsutism: An epidemiological cohort study. Int. J. Eat. Disord. 2008, 41, 427–431.
- American Psychiatric Association Division of Research. Highlights of Changes from DSM-IV to DSM-5. FOCUS 2013, 11, 525–527.
- Jeanes, Y.M.; Reeves, S.; Gibson, E.L.; Piggott, C.; May, V.A.; Hart, K.H. Binge eating behaviours and food cravings in women with Polycystic Ovary Syndrome. Appetite 2017, 109, 24–32.
- Farrell, K.; Antoni, M.H. Insulin resistance, obesity, inflammation, and depression in polycystic ovary syndrome: Biobehavioral mechanisms and interventions. Fertil. Steril. 2010, 94, 1565–1574.
- Setji, T.L.; Brown, A.J. Comprehensive clinical management of polycystic ovary syndrome. Minerva Med. 2007, 98, 175–189.
- Teede, H.J.; Misso, M.L.; Costello, M.F.; Dokras, A.; Laven, J.; Moran, L.; Piltonen, T.; Norman, R.J.; International PCOS Network. Recommendations from the international evidence-based guideline for the assessment and management of polycystic ovary syndrome. Hum. Reprod. 2018, 33, 1602–1618.
- Moghetti, P.; Tosi, F. Insulin resistance and PCOS: Chicken or egg? J. Endocrinol. Investig. 2021, 44, 233–244.
- Sadeeqa, S.; Mustafa, T.; Latif, S. Polycystic Ovarian Syndrome-Related Depression in Adolescent Girls: A Review. J. Pharm. Bioallied. Sci. 2018, 10, 55–59.
- Baker, J.H.; Girdler, S.S.; Bulik, C.M. The role of reproductive hormones in the development and maintenance of eating disorders. Expert Rev. Obstet. Gynecol. 2012, 7, 573–583.
- Sundblad, C.; Bergman, L.; Eriksson, E. High levels of free testosterone in women with bulimia nervosa. Acta Psychiatr. Scand. 1994, 90, 397–398.
- Sundblad, C.; Landén, M.; Eriksson, T.; Bergman, L.; Eriksson, E. Effects of the Androgen Antagonist Flutamide and the Serotonin Reuptake Inhibitor Citalopram in Bulimia Nervosa. J. Clin. Psychopharmacol. 2005, 25, 85–88.
- Krug, I.; Giles, S.; Paganini, C. Binge eating in patients with polycystic ovary syndrome: Prevalence, causes, and management strategies. Neuropsychiatr. Dis. Treat. 2019, 15, 1273–1285.
- Garvey, W.T. Is Obesity or Adiposity-Based Chronic Disease Curable: The Set Point Theory, the Environment, and Second-Generation Medications. Endocr. Pract. Off. J. Am. Coll. Endocrinol. Am. Assoc. Clin. Endocrinol. 2022, 28, 214–222.
- Karacan, E.; Caglar, G.S.; Gürsoy, A.Y.; Yilmaz, M.B. Body satisfaction and eating attitudes among girls and young women with and without polycystic ovary syndrome. J. Pediatr. Adolesc. Gynecol. 2014, 27, 72–77.
- Frisch, R.E.; Revelle, R. Height and weight at menarche and a hypothesis of critical body weights and adolescent events. Science 1970, 169, 397–399.
- Biro, F.M.; Galvez, M.P.; Greenspan, L.C.; Succop, P.A.; Vangeepuram, N.; Pinney, S.M.; Teitelbaum, S.; Windham, G.C.; Kushi, L.H.; Wolff, M.S. Pubertal assessment method and baseline characteristics in a mixed longitudinal study of girls. Pediatrics 2010, 126, e583–e590.
- Durda-Masny, M.; Hanć, T.; Czapla, Z.; Szwed, A. BMI at menarche and timing of growth spurt and puberty in Polish girls-longitudinal study. Anthropol. Anz. Ber. Uber Biol.-Anthropol. Lit. 2019, 76, 37–47.
- de Siqueira Barros, B.; Kuschnir, M.C.M.C.; Bloch, K.V.; da Silva, T.L.N. ERICA: Age at menarche and its association with nutritional status. J. Pediatr. 2019, 95, 106–111.
- Quennell, J.H.; Mulligan, A.C.; Tups, A.; Liu, X.; Phipps, S.J.; Kemp, C.J.; Herbison, A.; Grattan, D.; Anderson, G.M. Leptin Indirectly Regulates Gonadotropin-Releasing Hormone Neuronal Function. Endocrinology 2009, 150, 2805–2812.
- Ahmed, M.L.; Ong, K.K.; Dunger, D.B. Childhood obesity and the timing of puberty. Trends Endocrinol. Metab. 2009, 20, 237–242.
- Heras, V.; Castellano, J.M.; Fernandois, D.; Velasco, I.; Rodríguez-Vazquez, E.; Roa, J.; Vazquez, M.J.; Ruiz-Pino, F.; Rubio, M.; Pineda, R.; et al. Central Ceramide Signaling Mediates Obesity-Induced Precocious Puberty. Cell Metab. 2020, 32, 951–966.e8.
- Chen, J.; Shen, S.; Tan, Y.; Xia, D.; Xia, Y.; Cao, Y.; Wang, W.; Wu, X.; Wang, H.; Yi, L.; et al. The correlation of aromatase activity and obesity in women with or without polycystic ovary syndrome. J. Ovarian Res. 2015, 8, 11.
- Apter, D.; Bützow, T.; Laughlin, G.A.; Yen, S.S. Metabolic features of polycystic ovary syndrome are found in adolescent girls with hyperandrogenism. J. Clin. Endocrinol. Metab. 1995, 80, 2966–2973.
- Wei, S.; Schmidt, M.D.; Dwyer, T.; Norman, R.J.; Venn, A.J. Obesity and menstrual irregularity: Associations with SHBG, testosterone, and insulin. Obes. Silver Spring Md. 2009, 17, 1070–1076.
- Diamanti-Kandarakis, E.; Papavassiliou, A.G. Molecular mechanisms of insulin resistance in polycystic ovary syndrome. Trends Mol. Med. 2006, 12, 324–332.
- Tosi, F.; Bonora, E.; Moghetti, P. Insulin resistance in a large cohort of women with polycystic ovary syndrome: A comparison between euglycaemic-hyperinsulinaemic clamp and surrogate indexes. Hum. Reprod. 2017, 32, 2515–2521.
- Itriyeva, K. The effects of obesity on the menstrual cycle. Curr. Probl. Pediatr. Adolesc. Health Care 2022, 52, 101241.
- Grimm, E.R.; Steinle, N.I. Genetics of eating behavior: Established and emerging concepts. Nutr Rev. 2011, 69, 52–60.
- Annagür, B.B.; Kerimoglu, Ö.S.; Tazegül, A.; Gündüz, Ş.; Gençoglu, B.B. Psychiatric comorbidity in women with polycystic ovary syndrome. J. Obstet. Gynaecol. Res. 2015, 41, 1229–1233.
- Badman, M.K.; Flier, J.S. The gut and energy balance: Visceral allies in the obesity wars. Science 2005, 307, 1909–1914.
- Klok, M.D.; Jakobsdottir, S.; Drent, M.L. The role of leptin and ghrelin in the regulation of food intake and body weight in humans: A review. Obes. Rev. Off. J. Int. Assoc. Study Obes. 2007, 8, 21–34.
- Berridge, K.C.; Ho, C.Y.; Richard, J.M.; DiFeliceantonio, A.G. The tempted brain eats: Pleasure and desire circuits in obesity and eating disorders. Brain Res. 2010, 1350, 43–64.
- Vallis, M. Sustained behaviour change in healthy eating to improve obesity outcomes: It is time to abandon willpower to appreciate wanting. Clin. Obes. 2019, 9, e12299.
- Brzechffa, P.R.; Jakimiuk, A.J.; Agarwal, S.K.; Weitsman, S.R.; Buyalos, R.P.; Magoffin, D.A. Serum immunoreactive leptin concentrations in women with polycystic ovary syndrome. J. Clin. Endocrinol. Metab. 1996, 81, 4166–4169.
- Blüher, S.; Mantzoros, C.S. Leptin in humans: Lessons from translational research. Am. J. Clin. Nutr. 2009, 89, 991S–997S.
- Kieffer, T.J.; Heller, R.S.; Leech, C.A.; Holz, G.G.; Habener, J.F. Leptin suppression of insulin secretion by
- Casabiell, X.; Piñeiro, V.; Vega, F.; De La Cruz, L.F.; Diéguez, C.; Casanueva, F.F. Leptin, reproduction and sex steroids. Pituitary 2001, 4, 93–99.
- Erturk, E.; Kuru, N.; Savcı, V.; Tuncel, E.; Ersoy, C.; Imamoglu, S. Serum leptin levels correlate with obesity parameters but not with hyperinsulinism in women with polycystic ovary syndrome. Fertil. Steril. 2004, 82, 1364–1368.
- Morin-Papunen, L.C.; Koivunen, R.M.; Tomás, C.; Ruokonen, A.; Martikainen, H.K. Decreased serum leptin concentrations during metformin therapy in obese women with polycystic ovary syndrome. J. Clin. Endocrinol. Metab. 1998, 83, 2566–2568.
- Romualdi, D.; Campagna, G.; Selvaggi LJr Cento, R.; Proto, C.; Lanzone, A.; Guido, M. Metformin treatment does not affect total leptin levels and free leptin index in obese patients with polycystic ovary syndrome. Fertil. Steril. 2008, 89, 1273–1276.
- Romualdi, D.; Immediata, V.; De Cicco, S.; Tagliaferri, V.; Lanzone, A. Neuroendocrine Regulation of Food Intake in Polycystic Ovary Syndrome. Reprod. Sci. 2018, 25, 644–653. [
- Acosta, A.; Camilleri, M.; Abu Dayyeh, B.; Calderon, G.; Gonzalez, D.; McRae, A.; Rossini, W.; Singh, S.; Burton, D.; Clark, M.M. Selection of Antiobesity Medications Based on Phenotypes Enhances Weight Loss: A Pragmatic Trial in an Obesity Clinic. Obes. Silver Spring Md. 2021, 29, 662–671.
- Barrea, L.; Verde, L.; Vetrani, C.; Savastano, S.; Colao, A.; Muscogiuri, G. Chronotype: A Tool to Screen Eating Habits in Polycystic Ovary Syndrome? Nutrients 2022, 14, 955.
- Eleftheriadou, M.; Stefanidis, K.; Lykeridou, K.; Iliadis, I.; Michala, L. Dietary habits in adolescent girls with polycystic ovarian syndrome. Gynecol. Endocrinol. Off. J. Int. Soc. Gynecol. Endocrinol. 2015, 31, 269–271.
- Kulshreshtha, B.; Sharma, N.; Pant, S.; Sharma, L.; Pahuja, B.; Singh, P. PCOS patients differ in meal timings rather than total caloric or macronutrient intake in comparison to weight matched controls. Eur. J. Obstet. Gynecol. Reprod. Biol. 2022, 270, 11–16.
- De Giuseppe, R.; Braschi, V.; Bosoni, D.; Biino, G.; Stanford, F.C.; Nappi, R.E.; Cena, H. Dietary underreporting in women affected by polycystic ovary syndrome: A pilot study. Nutr. Diet J. Dietit. Assoc. Aust. 2019, 76, 560–566.
- Cetik, S.; Acikgoz, A.; Yildiz, B.O. Investigation of taste function and eating behavior in women with polycystic ovary syndrome. Appetite 2022, 168, 105776.
- Jensterle, M.; Ferjan, S.; Battelino, T.; Kovač, J.; Battelino, S.; Šuput, D.; Vovk, A.; Janež, A. Does intervention with GLP-1 receptor agonist semaglutide modulate perception of sweet taste in women with obesity: Study protocol of a randomized, single-blinded, placebo-controlled clinical trial. Trials 2021, 22, 464.
