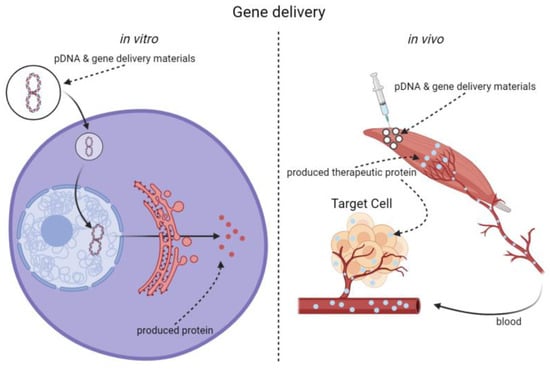
Since Jon A. Wolff found skeletal muscle cells being able to express foreign genes and Russell J. Mumper increased the gene transfection efficiency into the myocytes by adding polymers, skeletal muscles have become a potential gene delivery and expression target. Different methods have been developing to deliver transgene into skeletal muscles. Among them, viral vectors may achieve potent gene delivery efficiency. However, the potential for triggering biosafety risks limited their clinical applications. Therefore, non-viral biomaterial-mediated methods with reliable biocompatibility are promising tools for intramuscular gene delivery in situ. In recent years, a series of advanced non-viral gene delivery materials and related methods have been reported, such as polymers, liposomes, cell penetrating peptides, as well as physical delivery methods. We summarized the research progresses and challenges in non-viral intramuscular gene delivery materials and related methods, focusing on the achievements and future directions of polymers.
- non-viral materials
- gene delivery
- skeletal muscle
1. Introduction
| Polymers | Year | Reporter Genes | Functional Genes | Function |
|---|---|---|---|---|
| Polyvinyl pyrrolidone (PVP) | 1996 [7] | CAT, β-CAT | / |
|
| Pluronic P85® | 2005 [8] | Luc | / |
|
| 2009 [9] | GFP, Luc | / |
|
|
| 2016 [10] | Luc, GFP | pDRIVE5Lucia-mDesmin & pDRIVE5GFP-mDesmin |
|
|
| Multi-block copolymers from Pluronic P85 and di-(ethylene glycol) divinyl ether® | 2009 [11] | Luc | / |
|
| Pluronic SP1017® | 2005 [8] | Luc | / |
|
| Pluronic 25R2® | 2011 [12] | Luc | / |
|
| Pluronic L64® | 2002 [13] | Luc, β-Gal | / |
|
| 2014 [14] | / | HIF-1α |
|
|
| 2019 [15] | lacZ, Luc, E2-Crimson | GHRH |
|
|
| 2021 [1] | E2-Crimson, EGFP | mlFVII Icon |
|
|
| PEG13-PLGA10-PEG13 | 2007 [16] | Luc | VEGF |
|
| PEO–PTHF–PEO | 2008 [17] | Luc, lacZ | / |
|
| LPL | 2014 [18] | lacZ, Luc, E2-Crimson | mGH |
|
| rL2PL2, rL3PL3 | 2016 [19] | lacZ, Luc, E2-Crimson | GHRH |
|
| TBCPs | 2020 [20] | Luc | / |
|
| Lutrol | 2005 [21] | Luc | / |
|
| Poloxamine 304 | ||||
| poly(D, L-lactide-co-glycolide) | 2006 [22] | Luc | / |
|
| PLGA/E100 | 2009 [23] | / | IL-10 |
|
| HCPEI | 2008 [24] | / | VEGF |
|
| TAEI-PEI | 2012 [25] | GFP | / |
|
| PLGA | 2008 [26] | / | VEGF |
|
| PEG-PAMAM | 2009 [3] | EGFP | / |
|
| PAMAM-G5/SMTP/DBP/NLS mixture | 2021 [27] | EGFP | micro-dystrophin (µDys) |
|
2. Advantages and Challenges of the Skeletal Muscle Gene Delivery
2.1. Advantages of the Skeletal Muscle as the Target for Gene Delivery
2.2. Obstacles to Skeletal Muscles Gene Delivery
2.2.1. Obstructions in the Extracellular Matrix
2.2.2. Cytoplasmic Membrane
2.2.3. Endosomal Escaping
2.2.4. Entering the Nucleus
2.2.5. Material Stability
2.2.6. Biosecurity
This entry is adapted from the peer-reviewed paper 10.3390/pharmaceutics14112428
References
- Ma, L.; Wang, G.; Liu, S.; Bi, F.; Liu, M.; Wang, G.; Intramuscular Expression of Plasmid-Encoded FVII-Fc Immunoconjugate for Tumor Immunotherapy by Targeting Tumoral Blood Vessels and Cells. Front. Oncol. 2021, 11, 638591, .
- Deng, L.; Yang, P.; Li, C.; Xie, L.; Lu, W.; Zhang, Y.; Liu, M.; Wang, G.; Prolonged control of insulin-dependent diabetes via intramuscular expression of plasmid-encoded single-strand insulin analogue. . Genes Dis. 2022, /, /, 10.1016/j.gendis.2022.05.009.
- Qi, R.; Gao, Y.; Tang, Y.; He, R.R.; Liu, T.L.; He, Y.; Sun, S.; Li, B.Y.; Li, Y.B.; Liu, G.; et al. PEG-conjugated PAMAM dendrimers mediate efficient intramuscular gene expression. Aaps j 2009, 11, 395-405, 10.1208/s12248-009-9116-1.
- Wolff, J.A.; Malone, R.W.; Williams, P.; Chong, W.; Acsadi, G.; Jani, A.; Felgner, P.L.; Direct gene transfer into mouse muscle in vivo. Science 1990, 247, 1465-1468, .
- Gao, X.; Kim, K.S.; Liu, D.; Nonviral gene delivery: what we know and what is next.. Aaps j 2007, 9, E92-104, 10.1208/aapsj0901009..
- Aied, A.; Greiser, U.; Pandit, A.; Wang, W.; Polymer gene delivery: overcoming the obstacles.. Drug Discov. Today 2013, 18, 1090-1098, 10.1016/j.drudis.2013.06.014.
- Mumper, R.J.; Duguid, J.G.; Anwer, K.; Barron, M.K.; Nitta, H.; Rolland, A.P.; Polyvinyl derivatives as novel interactive polymers for controlled gene delivery to muscle.. Pharm Res 1996, 13, 701-709, 10.1023/a:1016039330870.
- Yang, Z.; Zhu, J.; Sriadibhatla, S.; Gebhart, C.; Alakhov, V.; Kabanov, A.; Yang, Z.; Zhu, J.; Sriadibhatla, S.; Gebhart, C.; Alakhov, V.; Kabanov, A.. J Control Release 2005, 108, 496-512, .
- Gaymalov, Z.Z.; Yang, Z.; Pisarev, V.M.; Alakhov, V.Y.; Kabanov, A.V.; The effect of the nonionic block copolymer pluronic P85 on gene expression in mouse muscle and antigen-presenting cells.. Biomaterials 2009, 30, 1232-1245, .
- Mahajan, V.; Gaymalov, Z.; Alakhova, D.; Gupta, R.; Zucker, I.H.; Kabanov, A.V.; Horizontal gene transfer from macrophages to ischemic muscles upon delivery of naked DNA with Pluronic block copolymers.. Biomaterials 2016, 75, 58-70, .
- Namgung, R.; Nam, S.; Kim, S.K.; Son, S.; Singha, K.; Kwon, J.S.; Ahn, Y.; Jeong, M.H.; Park, I.K.; Garripelli, V.K.; et al. An acid-labile temperature-responsive sol-gel reversible polymer for enhanced gene delivery to the myocardium and skeletal muscle cells. Biomaterials 2009, 30, 5225-5233, 10.1016/j.biomaterials.2009.05.073..
- Guiraud, S.; Alimi-Guez, D.; van Wittenberghe, L.; Scherman, D.; Kichler, A.; The reverse block copolymer Pluronic 25R2 promotes DNA transfection of skeletal muscle. Macromol Biosci 2011, 11, 590-594, 10.1002/mabi.201000463.
- Pitard, B.; Pollard, H.; Agbulut, O.; Lambert, O.; Vilquin, J.T.; Cherel, Y.; Abadie, J.; Samuel, J.L.; Rigaud, J.L.; Menoret, S.; et al. A nonionic amphiphile agent promotes gene delivery in vivo to skeletal and cardiac muscles. Hum Gene Ther 2002, 13, 1767-1775, 10.1089/104303402760293592.
- Song, H.; Liu, S.; Li, C.; Geng, Y.; Wang, G.; Gu, Z.; Pluronic L64-mediated stable HIF-1α expression in muscle for therapeutic angiogenesis in mouse hindlimb ischemia. Int J Nanomedicine 2014, 9, 3439-3452, 10.2147/ijn.S65353.
- He, Y.; Liu, Y.; Sun, Z.; Han, F.; Tang, J.Z.; Gao, R.; Wang, G.; The proper strategy to compress and protect plasmid DNA in the Pluronic L64-electropulse system for enhanced intramuscular gene delivery. Regen Biomater 2019, 6, 289-298, 10.1093/rb/rby028.
- Chang, C.W.; Choi, D.; Kim, W.J.; Yockman, J.W.; Christensen, L.V.; Kim, Y.H.; Kim, S.W.; Non-ionic amphiphilic biodegradable PEG-PLGA-PEG copolymer enhances gene delivery efficiency in rat skeletal muscle. J Control Release 2007, 118, 245-253, 10.1016/j.jconrel.2006.11.025.
- Pomel, C.; Leborgne, C.; Cheradame, H.; Scherman, D.; Kichler, A.; Guegan, P.; Synthesis and evaluation of amphiphilic poly(tetrahydrofuran-b-ethylene oxide) copolymers for DNA delivery into skeletal muscle. Pharm Res 2008, 25, 2963-2971, 10.1007/s11095-008-9698-9.
- Pu, L.; Geng, Y.; Liu, S.; Chen, J.; Luo, K.; Wang, G.; Gu, Z.; .Electroneutralized amphiphilic triblock copolymer with a peptide dendron for efficient muscular gene delivery. ACS Appl Mater Interfaces 2014, 6, 15344-15351, 10.1021/am503808b.
- Pu, L.; Wang, J.; Li, N.; Chai, Q.; Irache, J.M.; Wang, G.; Tang, J.Z.; Gu, Z.; Synthesis of Electroneutralized Amphiphilic Copolymers with Peptide Dendrons for Intramuscular Gene Delivery. ACS Appl Mater Interfaces 2016, 8, 13724-13734, 10.1021/acsami.6b02592.
- Rasolonjatovo, B.; Illy, N.; Bennevault, V.; Mathé, J.; Midoux, P.; Le Gall, T.; Haudebourg, T.; Montier, T.; Lehn, P.; Pitard, B.; et al. Temperature-Sensitive Amphiphilic Non-Ionic Triblock Copolymers for Enhanced In Vivo Skeletal Muscle Transfection. Macromol Biosci 2020, 20, e1900276, 10.1002/mabi.201900276.
- Richard, P.; Bossard, F.; Desigaux, L.; Lanctin, C.; Bello-Roufai, M.; Pitard, B.; Amphiphilic block copolymers promote gene delivery in vivo to pathological skeletal muscles. Hum Gene Ther 2005, 16, 1318-1324, 10.1089/hum.2005.16.1318.
- Jang, J.H.; Shea, L.D.; Intramuscular delivery of DNA releasing microspheres: microsphere properties and transgene expression. J Control Release 2006, 112, 120-128, 10.1016/j.jconrel.2006.01.013.
- Basarkar, A.; Singh, J.; Poly (lactide-co-glycolide)-polymethacrylate nanoparticles for intramuscular delivery of plasmid encoding interleukin-10 to prevent autoimmune diabetes in mice.. Pharm Res 2009, 26, 72-81, 10.1007/s11095-008-9710-4.
- Jeon, O.; Yang, H.S.; Lee, T.J.; Kim, B.S.; Heparin-conjugated polyethylenimine for gene delivery. J Control Release 2008, 132, 236-242, 10.1016/j.jconrel.2008.05.017.
- Wang, M.; Tucker, J.D.; Lu, P.; Wu, B.; Cloer, C.; Lu, Q.; Tris[2-(acryloyloxy)ethyl]isocyanurate cross-linked low-molecular-weight polyethylenimine as gene delivery carriers in cell culture and dystrophic mdx mice. Bioconjug Chem 2012, 23, 837-845, 10.1021/bc200674e.
- Kang, S.W.; Lim, H.W.; Seo, S.W.; Jeon, O.; Lee, M.; Nanosphere-mediated delivery of vascular endothelial growth factor gene for therapeutic angiogenesis in mouse ischemic limbs. Biomaterials 2008, 29, 1109-1117, 10.1016/j.biomaterials.2007.11.004.
- Hersh, J.; Condor Capcha, J.M.; Iansen Irion, C.; Lambert, G.; Noguera, M.; Singh, M.; Kaur, A.; Dikici, E.; Jiménez, J.J.; Shehadeh, L.A.; et al. Peptide-Functionalized Dendrimer Nanocarriers for Targeted Microdystrophin Gene Delivery. Pharmaceutics 2021, 13, 2159-2175, 10.3390/pharmaceutics13122159.
- Lu, Q.L.; Bou-Gharios, G.; Partridge, T.A.; Non-viral gene delivery in skeletal muscle: a protein factory. Gene Ther 2003, 10, 131-142, 10.1038/sj.gt.3301874.
- Liu, J.; Song, L.; Liu, S.; Jiang, Q.; Liu, Q.; Li, N.; Wang, Z.G.; Ding, B.; A DNA-Based Nanocarrier for Efficient Gene Delivery and Combined Cancer Therapy. Nano Lett 2018, 18, 3328-3334 Nano Lett 2018, 18, 3328-3334, Nano Lett 2018, 18, 3328-3334, Nano Lett 2018, 18, 3328-3334, .
- Zhou, X.; Xu, L.; Xu, J.; Wu, J.; Kirk, T.B.; Ma, D.; Xue, W.; Construction of a High-Efficiency Drug and Gene Co-Delivery System for Cancer Therapy from a pH-Sensitive Supramolecular Inclusion between Oligoethylenimine- graft-β-cyclodextrin and Hyperbranched Polyglycerol Derivative. ACS Appl Mater Interfaces 2018, 10, 35812-35829, 10.1021/acsami.8b14517.
- Mahendra, G.; Kumar, S.; Isayeva, T.; Mahasreshti, P.J.; Curiel, D.T.; Stockardt, C.R.; Grizzle, W.E.; Alapati, V.; Singh, R.; Siegal, G.P.; et al. Antiangiogenic cancer gene therapy by adeno-associated virus 2-mediated stable expression of the soluble FMS-like tyrosine kinase-1 receptor. Cancer Gene Ther 2005, 12, 26-34, 10.1038/sj.cgt.7700754.
- Piekarowicz, K.; Bertrand, A.T.; Azibani, F.; Beuvin, M.; Julien, L.; Machowska, M.; Bonne, G.; Rzepecki, R.; A Muscle Hybrid Promoter as a Novel Tool for Gene Therapy. Mol Ther Methods Clin Dev 2019, 15, 157-169, 10.1016/j.omtm.2019.09.001.
- Le Bihan, M.C.; Bigot, A.; Jensen, S.S.; Dennis, J.L.; Rogowska-Wrzesinska, A.; Laine, J.; Gache, V.; Furling, D.; Jensen, O.N.; Voit, T.; et al. In-depth analysis of the secretome identifies three major independent secretory pathways in differentiating human myoblasts. J Proteomics 2012, 77, 344-356, 10.1016/j.jprot.2012.09.008.
- Ost, M.; Coleman, V.; Kasch, J.; Klaus, S.; Regulation of myokine expression: Role of exercise and cellular stress. Free Radic Biol Med 2016, 98, 78-89, 10.1016/j.freeradbiomed.2016.02.018.
- Riu, E.; Mas, A.; Ferre, T.; Pujol, A.; Gros, L.; Otaegui, P.; Montoliu, L.; Bosch, F.; Counteraction of type 1 diabetic alterations by engineering skeletal muscle to produce insulin: insights from transgenic mice. Diabetes 2002, 51, 704-711, 10.2337/diabetes.51.3.704.
- Callejas, D.; Mann, C.J.; Ayuso, E.; Lage, R.; Grifoll, I.; Roca, C.; Andaluz, A.; Ruiz-de Gopegui, R.; Montané, J.; Muñoz, S.; et al. Treatment of diabetes and long-term survival after insulin and glucokinase gene therapy. Diabetes 2013, 62, 1718-1729, 10.2337/db12-1113.
- Blaveri, K.; Heslop, L.; Yu, D.S.; Rosenblatt, J.D.; Gross, J.G.; Partridge, T.A.; Morgan, J.E.; Patterns of repair of dystrophic mouse muscle: studies on isolated fibersPatterns of repair of dystrophic mouse muscle: studies on isolated fibers. Dev Dyn 1999, 216, 244-256, 10.1002/(sici)1097-0177(199911)216:3<244::Aid-dvdy3>3.0.Co;2-9.
- Csapo, R.; Gumpenberger, M.; Wessner, B.; Skeletal Muscle Extracellular Matrix - What Do We Know About Its Composition, Regulation, and Physiological Roles? A Narrative Review. Front Physiol 2020, 11, 253, 10.3389/fphys.2020.00253.
- Gillies, A.R.; Lieber, R.L.; Structure and function of the skeletal muscle extracellular matrix. Muscle Nerve 2011, 44, 318-331, 10.1002/mus.22094.
- Ruponen, M.; Rönkkö, S.; Honkakoski, P.; Pelkonen, J.; Tammi, M.; Urtti, A.; Extracellular glycosaminoglycans modify cellular trafficking of lipoplexes and polyplexes. J Biol Chem 2001, 276, 33875-33880, 10.1074/jbc.M011553200.
- Caron, N.J.; Torrente, Y.; Camirand, G.; Bujold, M.; Chapdelaine, P.; Leriche, K.; Bresolin, N.; Tremblay, J.P.; Intracellular delivery of a Tat-eGFP fusion protein into muscle cells. Mol Ther 2001, 3, 310-318, 10.1006/mthe.2001.0279.
- Ruponen, M.; Honkakoski, P.; Rönkkö, S.; Pelkonen, J.; Tammi, M.; Urtti, A.; Extracellular and intracellular barriers in non-viral gene delivery. J Control Release 2003, 93, 213-217, 10.1016/j.jconrel.2003.08.004.
- Itaka, K.; Osada, K.; Morii, K.; Kim, P.; Yun, S.H.; Kataoka, K.; Polyplex nanomicelle promotes hydrodynamic gene introduction to skeletal muscle. J Control Release 2010, 143, 112-119, 10.1016/j.jconrel.2009.12.014.
- Walther, W.; Stein, U.; Siegel, R.; Fichtner, I.; Schlag, P.M.; Use of the nuclease inhibitor aurintricarboxylic acid (ATA) for improved non-viral intratumoral in vivo gene transfer by jet-injection. J Gene Med 2005, 7, 477-485, 10.1002/jgm.690.
- Sato, Y.; Yamauchi, N.; Takahashi, M.; Sasaki, K.; Fukaura, J.; Neda, H.; Fujii, S.; Hirayama, M.; Itoh, Y.; Koshita, Y.; et al. In vivo gene delivery to tumor cells by transferrin-streptavidin-DNA conjugate. Faseb j 2000, 14, 2108-2118, 10.1096/fj.99-1052com.
- Chen, J.; Luo, J.; Zhao, Y.; Pu, L.; Lu, X.; Gao, R.; Wang, G.; Gu, Z.; Increase in transgene expression by pluronic L64-mediated endosomal/lysosomal escape through its membrane-disturbing action. ACS Appl Mater Interfaces 2015, 7, 7282-7293, 10.1021/acsami.5b00486.
- Yang, N.S.; Burkholder, J.; Roberts, B.; Martinell, B.; McCabe, D.; In vivo and in vitro gene transfer to mammalian somatic cells by particle bombardment. Proc Natl Acad Sci U S A 1990, 87, 9568-9572, 10.1073/pnas.87.24.9568.
- Mir, L.M.; Banoun, H.; Paoletti, C.; Introduction of definite amounts of nonpermeant molecules into living cells after electropermeabilization: direct access to the cytosol. Exp Cell Res 1988, 175, 15-25, 10.1016/0014-4827(88)90251-0.
- Mitragotri, S.; Healing sound: the use of ultrasound in drug delivery and other therapeutic applications. Nat Rev Drug Discov 2005, 4, 255-260, 10.1038/nrd1662.
- Liu, F.; Song, Y.; Liu, D.; Hydrodynamics-based transfection in animals by systemic administration of plasmid DNA. Gene Ther 1999, 6, 1258-1266, 10.1038/sj.gt.3300947.
- Akinc, A.; Thomas, M.; Klibanov, A.M.; Langer, R.; Exploring polyethylenimine-mediated DNA transfection and the proton sponge hypothesis. J Gene Med 2005, 7, 657-663, 10.1002/jgm.696.
- Wightman, L.; Kircheis, R.; Rössler, V.; Carotta, S.; Ruzicka, R.; Kursa, M.; Wagner, E.; Different behavior of branched and linear polyethylenimine for gene delivery in vitro and in vivo. J Gene Med 2001, 3, 362-372, .
- Hu, Q.; Wang, J.; Shen, J.; Liu, M.; Jin, X.; Tang, G.; Chu, P.K.; Intracellular pathways and nuclear localization signal peptide-mediated gene transfection by cationic polymeric nanovectors. Biomaterials 2012, 33, 1135-1145, 10.1016/j.biomaterials.2011.10.023.
- Yi, W.J.; Yang, J.; Li, C.; Wang, H.Y.; Liu, C.W.; Tao, L.; Cheng, S.X.; Zhuo, R.X.; Zhang, X.Z.; Enhanced nuclear import and transfection efficiency of TAT peptide-based gene delivery systems modified by additional nuclear localization signals. Bioconjug Chem 2012, 23, 125-134, 10.1021/bc2005472.
- Ketola, T.M.; Hanzlíková, M.; Urtti, A.; Lemmetyinen, H.; Yliperttula, M.; Vuorimaa, E.; Role of polyplex intermediate species on gene transfer efficiency: polyethylenimine-DNA complexes and time-resolved fluorescence spectroscopy. J Phys Chem B 2011, 115, 1895-1902, 10.1021/jp109984c.
- Cai, J.; Yue, Y.; Rui, D.; Zhang, Y.; Liu, S.; Wu, C.; Effect of Chain Length on Cytotoxicity and Endocytosis of Cationic Polymers. Macromolecules 2011, 44, 2050-2057, 10.1021/ma102498g.
- Kunath, K.; von Harpe, A.; Fischer, D.; Petersen, H.; Bickel, U.; Voigt, K.; Kissel, T.; Low-molecular-weight polyethylenimine as a non-viral vector for DNA delivery: comparison of physicochemical properties, transfection efficiency and in vivo distribution with high-molecular-weight polyethylenimine. J Control Release 2003, 89, 113-125, 10.1016/s0168-3659(03)00076-2.
- Wirth, T.; Parker, N.; Ylä-Herttuala, S.; History of gene therapy. Gene 2013, 525, 162-169, 10.1016/j.gene.2013.03.137.
- Lai, T.C.; Kataoka, K.; Kwon, G.S.; Bioreducible polyether-based pDNA ternary polyplexes: balancing particle stability and transfection efficiency. Colloids Surf B Biointerfaces 2012, 99, 27-37, 10.1016/j.colsurfb.2011.09.026.
- Grandinetti, G.; Smith, A.E.; Reineke, T.M.; Membrane and nuclear permeabilization by polymeric pDNA vehicles: efficient method for gene delivery or mechanism of cytotoxicity?. Mol Pharm 2012, 9, 523-538, 10.1021/mp200368p.
- Roques, C.; Bouchemal, K.; Ponchel, G.; Fromes, Y.; Fattal, E.; Parameters affecting organization and transfection efficiency of amphiphilic copolymers/DNA carriers. J Control Release 2009, 138, 71-77, 10.1016/j.jconrel.2009.04.030.
