Distinct isoforms of FAD-containing ferredoxin-NADP+ oxidoreductase (FNR) and ferredoxin (Fd) are involved in photosynthetic and non-photosynthetic electron transfer systems. The FNR (FAD)-Fd [2Fe-2S] redox pair complex switches between one- and two-electron transfer reactions in steps involving FAD semiquinone intermediates. In cyanobacteria and some algae, one-electron carrier Fd serves as a substitute for low-potential FMN-containing flavodoxin (Fld) during growth under low-iron conditions. This complex evolves into the covalent FNR (FAD)-Fld (FMN) pair, which participates in a wide variety of NAD(P)H-dependent metabolic pathways as an electron donor, including bacterial sulfite reductase, cytochrome P450 BM3, plant or mammalian cytochrome P450 reductase and nitric oxide synthase isoforms. These electron transfer systems share the conserved Ser-Glu/Asp pair in the active site of the FAD module. In addition to physiological electron acceptors, the NAD(P)H-dependent diflavin reductase family catalyzes a one-electron reduction of artificial electron acceptors such as quinone-containing anticancer drugs. Conversely, NAD(P)H: quinone oxidoreductase (NQO1), which shares a Fld-like active site, functions as a typical two-electron transfer antioxidant enzyme, and the NQO1 and UDP-glucuronosyltransfease/sulfotransferase pairs function as an antioxidant detoxification system.
- ferredoxin-NADP+ oxidoreductase (FNR)
- ferredoxin (Fd)
- flavodoxin (Fld)
- diflavin reductase family
- catalytic cycle
- electron transfer
- redox potentials
- evolutionary aspects
1. Introduction
2. Structure and Properties of the FNR, Fd and Fld
| Enzyme Specicies | Eox/sq (mV) | Esq/red (mV) | pH |
|---|---|---|---|
| FNR (FAD) | |||
| Spinach (FAD) | −350 | −335 | 7 |
| −402 | −358 | 8 | |
| Spinach (FAD) | −306 | −386 | 8 |
| (in the presence of NADP+) | |||
| Anabaena (FAD) | −385 | −371 | 8 |
| Anabaena Flavodoxin (FMN) | −212 | −436 | 7 |
| Cyt P450 reductase (FAD-FMN) | |||
| Rabbit (FAD) | −290 | −365 | 7 |
| Rabbit (FMN) | −110 | −270 | 7 |
| Human (FAD) | −283 | −382 | 7 |
| Human (FMN) | −56 | −274 | 7 |
| nNOS reductase domain (FAD-FMN) | |||
| Rat (FAD) | −260 | −280 | 7.6 |
| Rat (FMN) | −50 | −276 | 7.6 |
| iNOSreductase domain (FAD-FMN) | |||
| Human (FAD) | −240 | −270 | 7 |
| Human (FMN) | −105 | −245 | 7 |
| FNR Species | KFdcat (s−1) | KFdm (µM) | KFdcat/KFdm (µM s−1) | KFldcat (s−1) | KFldm (µM) | KFldcat/KFldm (µM s−1) |
|---|---|---|---|---|---|---|
| Pea FNR | 139 | 6.5 | 21.3 | 30.6 | 16.7 | 1.8 |
| Anabaena FNR | 200 | 11 | 18.2 | 23.3 | 33 | 0.7 |
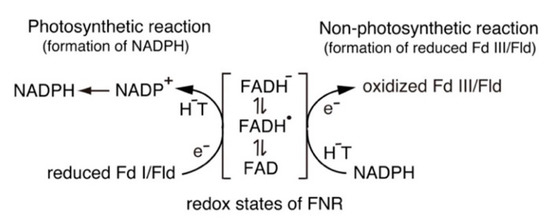
3. Catalytic Cycle of the FNR-Fd and FNR-Fld Systems
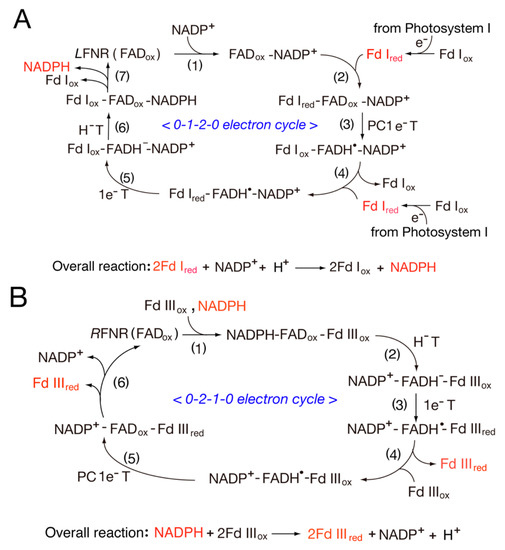
3.1. Catalytic Cycle of the FNR-Fd System
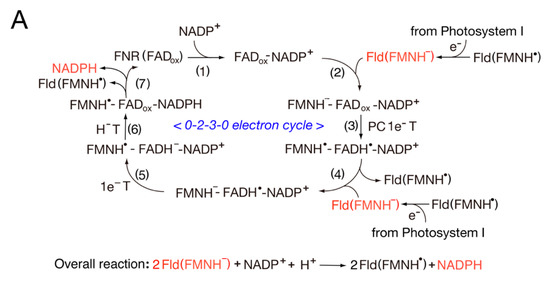
3.2. Catalytic Cycle of the FNR-Fld Systems
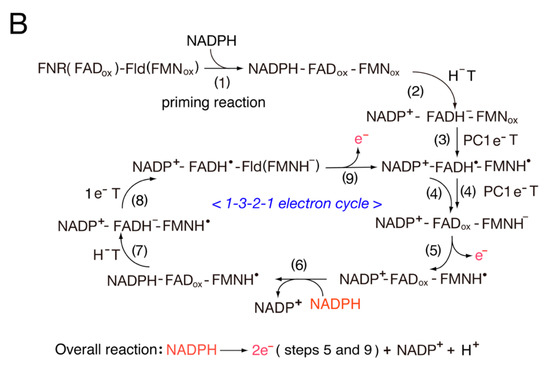
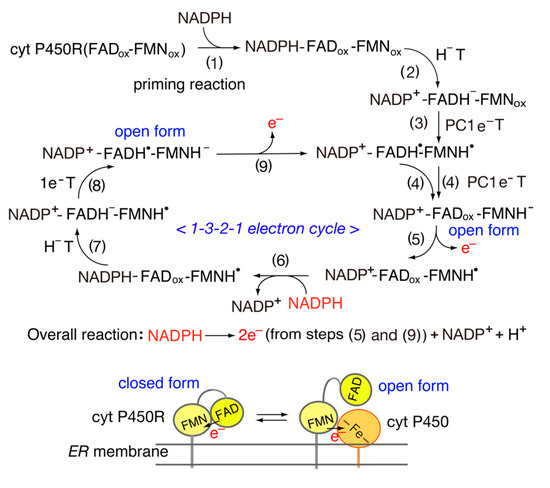
4. Structure and Properties of Diflavin Reductase Family

This entry is adapted from the peer-reviewed paper 10.3390/antiox11112143
References
- Shinohara, F.; Kurisu, G.; Hanke, G.; Bowsher, C.; Hase, T.; Kimata-Ariga, Y. Structural basis for the isotype-specific interactions of ferredoxin and ferredoxin: NADP+ oxidoreductase: An evolutionary switch between photosynthetic and heterotrophic assimilation. Photosynth. Res. 2017, 134, 281–289.
- Hanke, G.T.; Kurisu, G.; Kusunoki, M.; Hase, T. Fd: FNR electron transfer complexes: Evolutionary refinement of structural interactions. Photosynth. Res. 2004, 81, 317–327.
- Ceccarelli, E.A.; Arakaki, A.K.; Cortez, N.; Carrillo, N. Functional plasticity and catalytic efficiency in plant and bacterial ferredoxin-NADP (H) reductases. Biochim. Biophys. Acta Proteins Proteom. 2004, 1698, 155–165.
- Aliverti, A.; Pandini, V.; Pennati, A.; de Rosa, M.; Zanetti, G. Structural and functional diversity of ferredoxin-NADP+ reductases. Arch. Biochem. Biophys. 2008, 474, 283–291.
- Karplus, P.A.; Daniels, M.J. Atomic structure of ferredoxin-NADP+ reductase: Prototype for a structurally novel flavoenzyme family. Science 1991, 251, 60–66.
- Sánchez-Azqueta, A.; Catalano-Dupuy, D.L.; López-Rivero, A.; Tondo, M.L.; Orellano, E.G.; Ceccarelli, E.A.; Medina, M. Dynamics of the active site architecture in plant-type ferredoxin-NADP+ reductases catalytic complexes. Biochim. Biophys. Acta Bioenerg. 2014, 1837, 1730–1738.
- Onda, Y.; Matsumura, T.; Kimata-Ariga, Y.; Sakakibara, H.; Sugiyama, T.; Hase, T. Differential interaction of maize root ferredoxin: NADP+ oxidoreductase with photosynthetic and non-photosynthetic ferredoxin isoproteins. Plant. Physiol. 2000, 123, 1037–1046.
- Tognetti, V.B.; Zurbriggen, M.D.; Morandi, E.N.; Fillat, M.F.; Valle, E.M. Enhanced plant tolerance to iron starvation by functional substitution of chloroplast ferredoxin with a bacterial flavodoxin. Proc. Natl. Acad. Sci. USA 2007, 104, 11495–11500.
- Porter, T.D.; Kasper, C.B. NADPH-cytochrome P-450 oxidoreductase: Flavin mononucleotide and flavin adenine dinucleotide domains evolved from different flavoproteins. Biochemistry 1986, 25, 1682–1687.
- Haniu, M.; Iyanagi, T.; Miller, P.; Lee, T.D.; Shively, J.E. Complete amino acid sequence of NADPH-cytochrome P-450 reductase from porcine hepatic microsomes. Biochemistry 1986, 25, 7906–7911.
- Bredt, D.S.; Hwang, P.M.; Glatt, C.E.; Lowenstein, C.; Reed, R.R.; Snyder, S.H. Cloned and expressed nitric oxide synthase structurally resembles cytochrome P-450 reductase. Nature 1991, 351, 714–718.
- Iyanagi, T.; Xia, C.; Kim, J.J. NADPH-cytochrome P450 oxidoreductase: Prototypic member of the diflavin reductase family. Arch. Biochem. Biophys. 2012, 528, 72–89.
- Ingelman, M.; Bianchi, V.; Eklund, H. The three-dimensional structure of flavodoxin reductase from Escherichia coli at 1.7 Å resolution. J. Mol. Biol. 1997, 268, 147–157.
- Iyanagi, T. Molecular mechanism of metabolic NAD(P)H-dependent systems: The role of redox cofactors. Biochim. Biophys. Acta Bioenerg. 2019, 1860, 233–258.
- Iyanagi, T.; Yamazaki, I. One-electron-transfer reactions in biochemical systems III. One-electron reduction of quinones by microsomal flavin enzymes. Biochim. Biophys. Acta Bioenerg. 1969, 172, 370–381.
- Iyanagi, T.; Yamazaki, I. One-electron-transfer reactions in biochemical systems V. Difference in the mechanism of quinone reduction by the NADH dehydrogenase and the NAD (P) H dehydrogenase (DT-diaphorase). Biochim. Biophys. Acta Bioenerg. 1970, 216, 282–294.
- Čenas, N.; Anusevičius, Ž.; Bironaite, D.; Bachmanova, G.I.; Archakov, A.I.; Öllinger, K. The electron transfer reactions of NADPH: Cytochrome P450 reductase with nonphysiological oxidants. Arch. Biochem. Biophys. 1994, 315, 400–406.
- Čenas, N.; Anusevičius, Ž.; Nivinskas, H.; Misevičienė, L.; Šarlauskas, J. Structure-Activity Relationships in Two-Electron Reduction of Quinones. Methods Enzym. B. 2004, 382, 258–277.
- Anusevičius, Ž.; Misevičienė, L.; Medina, M.; Martinez-Julvez, M.; Gomez- Moreno, C.; Čėnas, N. FAD semiquinone stability regulates single-and two-electron reduction of quinones by Anabaena PCC7119 ferredoxin: NADP+ reductase and its Glu301Ala mutant. Arch. Biochem. Biophys. 2005, 437, 144–150.
- Bianchet, M.A.; Faig, M.; Amzel, L.M. Structure and mechanism of NAD H: Quinone acceptor oxidoreductases (NQO). Methods Enzymol. 2004, 382, 144–174.
- Iyanagi, T. Molecular mechanism of phase I and phase II drug-metabolizing enzymes: Implications for detoxification. Int. Rev. Cytol. 2007, 260, 35–112.
- Martínez-Júlvez, M.; Medina, M.; Gómez-Moreno, C. Ferredoxin-NADP+ reductase uses the same site for the interaction with ferredoxin and flavodoxin. J. Biol. Chem. 1999, 4, 568–578.
- Medina, M. Structural and mechanistic aspects of flavoproteins: Photosynthetic electron transfer from photosystem I to NADP+. FEBS J. 2009, 276, 3942–3958.
- Kurisu, G.; Kusunoki, M.; Katoh, E.; Yamazaki, T.; Teshima, K.; Onda, Y.; Kimata-Ariga, Y.; Hase, T. Structure of the electron transfer complex between ferredoxin and ferredoxin-NADP+ reductase. Nat. Struct. Biol. 2002, 8, 117–121.
- Medina, M.; Abagyan, R.; Gómez-Moreno, C.; Fernandez-Recio, J. Docking analysis of transient complexes: Interaction of ferredoxin-NADP+ reductase with ferredoxin and flavodoxin. Proteins 2008, 72, 848–862.
- Chikuma, Y.; Miyata, M.; Lee, Y.H.; Hase, T.; Kimata-Ariga, Y. Molecular mechanism of negative cooperativity of ferredoxin-NADP+ reductase by ferredoxin and NADP (H): Involvement of a salt bridge between Asp60 of ferredoxin and Lys33 of FNR. Biosci. Biotechnol. Biochem. 2021, 85, 860–865.
- Buchert, F.; Hamon, M.; Gäbelein, P.; Scholz, M.; Hippler, M.; Wollman, F.A. The labile interactions of cyclic electron flow effector proteins. J. Biol. Chem. 2018, 293, 17559–17573.
- Kean, K.M.; Carpenter, R.A.; Pandini, V.; Zanetti, G.; Hall, A.R.; Faber, R.; Aliverti, A.; Karplus, P.A. High resolution studies of hydride transfer in the ferredoxin: NADP+ reductase superfamily. FEBS J. 2017, 284, 3302–3319.
- Nogués, I.; Tejero, J.; Hurley, J.K.; Paladini, D.; Frago, S.; Tollin, G.; Mayhew, S.G.; Gomez-Moreno, G.; Eduardo, A.; Ceccarelli, E.A.; et al. Role of the C-terminal tyrosine of ferredoxin-nicotinamide adenine dinucleotide phosphate reductase in the electron transfer processes with its protein partners ferredoxin and flavodoxin. Biochemistry 2004, 43, 6127–6137.
- Dumit, V.I.; Essigke, T.; Cortez, N.; Ullmann, G.M. Mechanistic insights into ferredoxin–NADP(H) reductase catalysis involving the conserved glutamate in the active site. J. Mol. Biol. 2010, 397, 814–825.
- Faro, M.; Gómez-Moreno, C.; Stankovich, M.; Medina, M. Role of critical charged residues in reduction potential modulation of ferredoxin-NADP+ reductase: Differential stabilization of FAD redox forms. Eur. J. Biochem. 2002, 269, 2656–2661.
- Batie, C.J.; Kamin, H. Association of ferredoxin-NADP+ reductase with NADP(H) specificity and oxidation-reduction properties. J. Biol. Chem. 1986, 261, 11214–11223.
- Aliverti, A.; Faber, R.; Finnerty, C.M.; Ferioli, C.; Pandini, V.; Negri, A.; Karplus, P.A.; Zanetti, G. Biochemical and crystallographic characterization of Ferredoxin− NADP+ Reductase from nonphotosynthetic tissues. Biochemistry 2001, 40, 14501–14508.
- Corrado, M.E.; Aliverti, A.; Zanetti, G.; Mayhew, S.G. Analysis of the oxidation-reduction potentials of recombinant ferredoxin-NADP+ reductase from spinach chloroplasts. Eur. J. Biochem. 1996, 239, 662–667.
- Druhan, L.J.; Swenson, R.P. Role of Methionine 56 in the Control of the Oxidation−Reduction Potentials of the Clostridium beijerinckii Flavodoxin: Effects of Substitutions by Aliphatic Amino Acids and Evidence for a Role of Sulfur−Flavin Interactions. Biochemistry 1998, 37, 9668–9678.
- Ishikita, H. Influence of the protein environment on the redox potentials of flavodoxins from Clostridium beijerinckii. J. Biol. Chem. 2007, 282, 25240–25246.
- Chang, C.W.; He, T.F.; Guo, L.; Stevens, J.A.; Li, T.; Wang, L.; Zhong, D. Mapping solvation dynamics at the function site of flavodoxin in three redox states. J. Am. Chem. Soc. 2010, 132, 12741–12747.
- Rwere, F.; Im, S.; Waskell, L. The FMN “140s Loop” of Cytochrome P450 Reductase Controls Electron Transfer to Cytochrome P450. Int. J. Mol. Sci. 2021, 22, 10625.
- Iyanagi, T.; Makino, N.; Mason, H.S. Redox properties of the reduced nicotinamide adenine dinucleotide phosphate-cytochrome P-450 and reduced nicotinamide adenine dinucleotide-cytochrome b5 reductases. Biochemistry 1974, 13, 1701–1710.
- Munro, A.W.; Noble, M.A.; Robledo, L.; Daff, S.N.; Chapman, S.K. Determination of the redox properties of human NADPH-cytochrome P450 reductase. Biochemistry 2001, 40, 1956–1963.
- Haque, M.M.; Tejero, J.; Bayachou, M.; Kenney, C.T.; Stuehr, D.J. A cross-domain charge interaction governs the activity of NO synthase. J. Biol. Chem. 2018, 293, 4545–4554.
- Gao, Y.T.; Smith, S.M.; Weinberg, J.B.; Montgomery, H.J.; Newman, E.; Guillemette, J.G.; Ghosh, D.K.; Roman, L.J.; Martasek, P.; Salerno, J.C. Thermodynamics of oxidation-reduction reactions in mammalian nitric-oxide synthase isoforms. J. Biol. Chem. 2004, 279, 18759–18766.
- Pueyo, J.J.; Gomez-Moreno, C.; Mayhew, S.G. Oxidation-reduction potentials of ferredoxin-NADP+ reductase and flavodoxin. from Anabaena PCC 7119 and their electrostatic and covalent complexes. Eur. J. Biochem. 1991, 202, 1065–1071.
- Diaz-Quintana, A.; Leibl, W.; Bottin, H.; Sétif, P. Electron transfer in photosystem I reaction centers follows a linear pathway in which iron-sulfur cluster FB is the immediate electron donor to soluble ferredoxin. Biochemistry 1998, 37, 3429–3439.
- Batie, C.J.; Kamin, H. Electron transfer by ferredoxin: NADP+ reductase. Rapid -reaction evidence for participation of a ternary complex. J. Biol. Chem. 1984, 259, 11976–11985.
- Walker, M.C.; Pueyo, J.; Navarro, J.; Gómez-Moreno, C.; Tollin, G. Laser flash photolysis studies of the kinetics of reduction of ferredoxins and ferredoxin-NADP+ reductases from Anabaena PCC 7119 and spinach: Electrostatic effects on intracomplex electron transfer. Arch. Biochem. Biophys. 1991, 287, 351–358.
- Carrillo, N.; Ceccarelli, E.A. Open questions in ferredoxin-NADP+ reductase catalytic mechanism. Eur. J. Biochem. 2003, 270, 1900–1915.
- Tejero, J.; Peregrina, J.R.; Martínez-Júlvez, M.; Gutiérrez, A.; Gomez-Moreno, C.; Scrutton, N.S.; Medina, M. Catalytic mechanism of hydride transfer between NADP+/H and ferredoxin-NADP+ reductase from Anabaena PCC 7119. Arch. Biochem. Biophys. 2007, 459, 79–90.
- Mulo, P.; Medina, M. Interaction and electron transfer between ferredoxin–NADP+ oxidoreductase and its partners: Structural, functional, and physiological implications. Photosynth. Res. 2017, 134, 265–280.
- Saen-Oon, S.; de Vaca, I.C.; Masone, D.; Medina, M.; Guallar, V. A theoretical multiscale treatment of protein–protein electron transfer: The ferredoxin/ ferredoxin-NADP+ reductase and flavodoxin/ferredoxin-NADP+ reductase systems. Biochim. Biophys. Acta Bioenerg. 2015, 1847, 1530–1538.
- Nogués, I.; Martínez-Júlvez, M.; Navarro, J.A.; Hervás, M.; Armenteros, L.; de la Rosa, M.A.; Brodie, T.B.; Hurley, J.K.; Tollin, G.; Gomez-Moreno, C.; et al. Role of hydrophobic interactions in the flavodoxin mediated electron transfer from photosystem I to ferredoxin-NADP+ reductase in Anabaena PCC 7119. Biochemistry 2003, 42, 2036–2045.
- Utschig, L.M.; Brahmachari, U.; Mulfort, K.L.; Niklas, J.; Poluektov, O.G. Biohybrid photosynthetic charge accumulation detected by flavin semiquinone formation in ferredoxin-NADP+ reductase. Chem. Sci. 2022, 12, 6502–6511.
- Iyanagi, T.; Mason, H.S. Properties of hepatic reduced nicotinamide adenine dinucleotide phosphate-cytochrome c reductase. Biochemistry 1973, 12, 2297–2308.
- Leclerc, D.; Wilson, A.; Dumas, R.; Gafuik, C.; Song, D.; Watkins, D.; Heng, H.H.; Rommens, J.M.; Scherer, S.W.; Rosenblatt, D.S.; et al. Cloning and mapping of a cDNA for methionine synthase reductase, a flavoprotein defective in patients with homocystinuria. Proc. Natl. Acad. Sci. USA 1998, 95, 3059–3064.
- Wolthers, K.R.; Scrutton, N.S. Cobalamin uptake and reactivation occurs through specific protein interactions in the methionine synthase–methionine synthase reductase complex. FEBS J. 2009, 276, 1942–1951.
- Paine, M.J.; Garner, A.P.; Powell, D.; Sibbald, J.; Sales, M.; Pratt, N.; Smith, T.; Tew, D.G.; Wolf, C.R. Cloning and characterization of a novel human dual flavin reductase. J. Biol. Chem. 2000, 275, 1471–1478.
- Ostrowski, J.; Barber, M.J.; Rueger, D.C.; Miller, B.E.; Siegel, L.M.; Kredich, N.M. Characterization of the flavoprotein moieties of NADPH-sulfite reductase from Salmonella typhimurium and Escherichia coli. Physicochemical and catalytic properties, amino acid sequence deduced from DNA sequence of cysJ, and comparison with NADPH-cytochrome P-450 reductase. J. Biol. Chem. 1989, 264, 15796–15808.
- Narhi, L.O.; Fulco, A.J. Characterization of a catalytically self-sufficient 119,000-dalton cytochrome P-450 monooxygenase induced by barbiturates in Bacillus megaterium. J. Biol. Chem. 1986, 261, 7160–7169.
- Wang, M.; Roberts, D.L.; Paschke, R.; Shea, T.M.; Masters, B.S.S.; Kim, J.J.P. Three-dimensional structure of NADPH–cytochrome P450 reductase: Prototype for FMN-and FAD-containing enzymes. Proc. Natl. Acad. Sci. USA 1997, 94, 8411–8416.
- Xia, C.; Misra, I.; Iyanagi, T.; Kim, J.J.P. Regulation of interdomain interactions by calmodulin in inducible nitric-oxide synthase. J. Biol. Chem. 2009, 284, 30708–30717.
