1. The Biological Activity of the Cobalt Element
1.1. Cobalt Is the Core Element of VB12
Cobalt, a silvery-gray, lustrous, brittle but hard metal, is distributed widely in nature, including rocks, soil, plants, and animals. As a transition metal located in the fourth row of the periodic table, cobalt is a neighbor of iron and nickel with an atomic weight of 58.9 [
15]. Cobalt is an essential trace element for human health and can occur in organic and inorganic forms. The most widely known organic forms are the core element of cobalamin (vitamin B12 and its derivatives), which serve as cofactors of a wide range of enzymes and components of proteins [
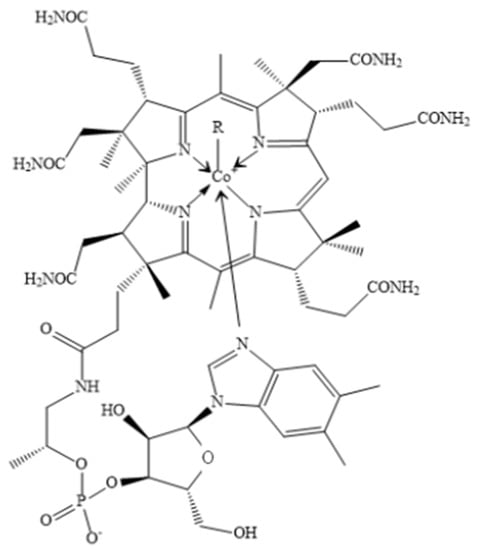
16]. As a water-soluble vitamin, vitamin B12 contains mineral cobalt, which is positioned centrally and coordinated with upper and lower ligands as a corrin ring [
17,
18] (
Figure 1). For this reason, the compounds with vitamin B12 activity are always called as “cobalamins”.
Figure 1. The chemical formula of VB12. R = 5′-deoxyadenosyl, CH3, OH, CN.
1.2. Physiological Function of Cobalt Based on Their Critical Roles in VB12
As an essential vitamin, vitamin B12 is obtained entirely from the diet. It is naturally found in animal food products, including meat, poultry, shell, fish, eggs, milk, and other dairy products [
19]. It is also synthesized naturally by some large intestine-resident bacteria in humans in the rumen from cobalt and has a major role in metabolism, especially in the peri-parturient period [
20,
21]. Vitamin B12 is always bound with proteins in food, which must be released by gastric acid breakdown in the stomach, where the salivary R-binder can bind with VB12 to prevent VB12 destruction [
22]. The absorption of vitamin B12 into the blood stream is dependent on their binding with the protein intrinsic factor (IF) to form the VB12-IF complex in the small intestine [
23].
Vitamin B12 is stored primarily in the liver and always acts as a cofactor for methionine synthesis from homocysteine and succinyl-CoA synthesis from methylmalonyl-CoA in mammalian systems [
24]. As an essential nutrient for folate metabolism and DNA synthesis, VB12 is critical for normal fetal and childhood growth and development [
25]. Maternal VB12 deficiency during pregnancy may increase the risk of neural tube defects and brain development retardation, as well as preterm birth and low birth weight [
26]. VB12 is also necessary for basic body functions, such as the nervous system, cardiovascular system, and immune system [
27], as well as the maintenance of skeletal muscle and neurobehavioral parameters, and modulation of gut microbiota [
28]. VB12 deficiency has also been associated with several metabolic disorders such as macrocytic anemia, cardiovascular, cerebrovascular, and neurological disorders [
29]. Clinical disease caused by VB12 deficiency usually results from the failure of the gastric or ileal phase of physiological B12 absorption, best exemplified by the autoimmune disease, pernicious anemia [
30].
1.3. The Physiological Function of the Cobalt in Hematopoiesis
As the central cofactor of vitamin B12 and the critical roles for proper nucleotide synthesis, cobalt can also stimulate the hematopoietic system of human bone marrow, which promotes the synthesis of hemoglobin and increase the number of red blood cells [
31]. The detailed mechanisms about cobalt stimulated hematopoiesis are summarized as following:
Firstly, as mentioned above, as the active center of the VB12, cobalt participates in the metabolism of ribonucleic acid and hematopoietic substances through VB12, which act on the hematopoietic process [
32]. Deficiency of cobalamin (vitamin B12) can result in megaloblastic anemia due to the inhibition of DNA synthesis caused by decreased availability of purines and pyrimidines (Thymidine monophosphate), which results in enlarged red blood cells and accumulation of significant, immature precursors (megaloblasts) of RBCs in the blood and bone marrow [
33].
Secondly, cobalt is involved in metabolism modulating transcriptional activator hypoxia-inducible factor-1 (HIF-1), which stimulates erythropoietin (EPO) production [
32]. HIF-1 is a transcription factor that controls hypoxia-induced autophagy by upregulating the expression of its downstream proteins [
34]. Cobalt can activate HIF-1 at normal oxygen levels, which is stabilized, translocated to the nucleus and then dimerized with the constitutively expressed HIF-1 to elicit the transcription of target genes necessary for increased oxygen demand [
10]. Study has also demonstrated that cobalt treatment may increase hypoxic tolerance of different tissues, improve muscle metabolism and exercise performance [
35].
Third, cobalt can promote the absorption of iron. Fe is an essential element important in a wide variety of metabolic processes, including oxygen transport, DNA synthesis, and electron transport. Fe is required for the production of red blood cells and forms part of hemoglobin, helping in the binding and transportation of oxygen in the body [
36]. Cobalt can promote the absorption of iron in the intestinal mucosa and accelerate the storage of iron into the bone marrow, which therefore very important for hematopoiesis.
1.4. The Anti-Infective Activity of Cobalt
In recent decades, an increasing number of studies have focused on investigating the structure and chemical behavior of some metal compounds to discover new drugs with antibacterial capabilities. Among them, cobalt has proven the ability to act as a potential candidate for antibiotic [
37]. Cobalt alloys have high corrosion resistance with a balance among biocompatibility and mechanical strength [
38], which makes it suitable for the artificial joint materials manufacturing. It was also recently reported that in addition to inducing a hypoxic response, Co(2+) incorporation could also improve the antibacterial ability of titanium-based bone implants, which suggested that Co(2+) had an additional effect as an antimicrobial agent.
Co
2+ can directly bind to the DNA of bacteria to induce bacterial cell death by different pathways [
39], for example, by inducing reactive oxygen species (ROS) production. ROS is the reduction products of oxygen, such as peroxides, which can destroy the cell membrane of bacteria and play a significant role in DNA and other cellular damage [
40]. Apart from that, cobalt can inhibit the function of RecBCD, which is crucial for initiating the SOS repair. The SOS response can promote the integrity of DNA, it also includes error-prone factors that allow for improved survival and continuous replication in the presence of extensive DNA damage [
41]. Moreover, cobalt can also be helpful to cure infectious diseases [
42], which is also partially associated with the immunological regulation effects. Cobalt can help in the creation and repair of the myelin sheath, which encircles the nerve cells and further protects them from external damage. A study has shown that cobalt is able to induce new blood vessel formation, as well as to improve wound closure and avoid bacterial infection [
43].
1.5. The Immunoregulatory Role of Cobalt
The immune system, which is integrated into all physiological systems and critical for human health, protects the host against infections [
44] and provides constant surveillance of native cells that may be harmful, such as cancerous cells [
45]. It is reported that metals are critically implicated in regulating both the innate immune sensing of and the host defense against invading pathogens [
46], which suggests that metals play an important role in regulating the immune system against infection. A recent study has shown that transition metal-based compounds could modulate autophagy, one of the most important host immunological responses, which therefore provides a new therapeutic strategy based on transition metal-based compounds for disease treatment [
47]. For example, CoCl
2 treatment activates autophagy through the target genes induced by HIF, and correlates with the expression of certain pro-apoptotic factors [
48]. Additionally, Co(2+) can induce an HIF-1α-dependent metabolic shift from oxphos towards glycolysis in macrophages, which plays an early and pivotal role in the inflammatory responses [
49]. Thus, the growing interests of researchers in transition metal-based compounds is not only due to their potent antibacterial, antifungal, antiviral, antitumor, and anti-inflammatory properties [
50], but also due to the promising immunological regulation effects.
One study shows that Co(2+) has a significant influence on osteoblastic activity, differentiation, and inflammatory processes [
51]. The formation of Co NPs in the wear process of MoM hip implants may lead to inflammatory fluid collections or osteolysis [
4]. In vitro studies have shown that Co(2+) can activate the production of bone-resorbing cytokines through the activation of redox-dependent mechanisms and activate the biosynthesis of inducible NO synthase and pro-inflammatory interleukins in macrophages [
52], thus inducing inflammatory responses in macrophages [
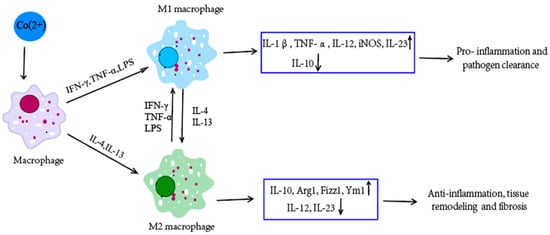
53] (
Figure 2). Macrophages are the predominant immune cells in periprosthetic tissues, which induce a type IV hypersensitivity reaction. Both T-lymphocytes and B-cells (to a lesser extent) are involved in the production of inflammatory mediators. Pro- and anti-inflammatory cytokines, the interleukins IL-1, IL-6, IL-4, and tumor necrosis factor-α (TNF-α), play a central role in the inflammatory reaction [
54]. A study identified a new downstream effect of cobalt-induced ROS production, which reduced RhoA expression in modulating macrophage migration and cytoskeleton organization, leading to an enhancement in macrophage spreading, adhesion, and inhibition of migration. These effects could induce a prolonged immune cell retention, which thereby propagates the chronic inflammation [
55]. Co(2+) can also promote pro-inflammatory gene expression by binding to the human TLR4 receptor [
56], which could activate dendritic cells migration to draining lymph nodes and present allergen-induced epitopes to trigger antigen-specific T cell proliferation, differentiation, and memory formation [
57]. Co(2+) has also been shown to increase the binding of T lymphocytes to endothelial cells and the transendothelial migration of these lymphocytes [
58], which independently induce circulating cytokine or chemokine production to promote the accumulation of T lymphocytes [
55]. Moreover, the fibrotic response could be further enhanced by the presence of macrophages. In the light of these results, Co(2+) and macrophages act synergistically to influence the functional properties of fibroblasts and extracellular matrix (ECM) homeostasis [
59]. Cobalt can also modulate immune cell functions in the lung and induce airway hyperreactivity with a mixed neutrophilic and eosinophilic inflammatory responses, which are accompanied by dendritic cells and innate lymphoid cells [
60]. However, epidemiological studies have demonstrated a higher risk of asthma in workers exposed to cobalt, and several case series of cobalt-induced asthma have also been reported [
60], which also highlight the toxicity issue of cobalt to human health.
Figure 2. Co(2+) can induce inflammatory responses in macrophages [
53]. Different environmental factors can induce the differentiation of macrophages into different subsets: M1 and M2 macrophages, which have different phenotypes, secrete different cytokines, and have different biological activities. M1 macrophages cells are activated by the classical way (activators include IFN-γ, TNF-α and LPS, etc.), which mainly play a role in killing microorganisms and promoting inflammation. M2 macrophage cells are activated by alternative ways (activators include IL-4 and IL-13), which are mainly involved in immune regulation, inhibition of inflammation, and tissue repair, and are related to the chronic progression of infectious diseases. M1 and M2 macrophages can transform into each other in different pathological processes and microenvironments. ↑ means upregulate and ↓ means downregulate.
In summary, as an essential trace element of the body, cobalt has an important physiological role. It is a component of vitamin B12 and some other enzymes, participates in the metabolism of the human body, and has the effect of stimulating hematopoiesis in various ways. The release of cobalt into the human body can also trigger the body’s immune system, which provide new therapeutic strategies for infectious diseases.
2. The Synthesis of Cobalt Nanomaterials
Along with the time, various chemical and physical synthesis methods of cobalt nanomaterials have emerged [
61], such as thermal decomposition, hydrothermal synthesis, chemical wet processing, thermal reduction, micro-emulsion, precipitation, sol–gel, microwave-assisted, reverse micelles, evaporation–condensation, and laser ablation [
62]. Cobalt nanomaterials are always synthesized by top-down or bottom-up methods [
63]. In the top-down method, cobalt compounds based on bulk materials are transformed to NPs through sputtering techniques, grinding, and milling. While in bottom-up methods, the self-assembly of miniature compounds into NPs is performed [
64]. A typical feature of physical methods for cobalt nanomaterial preparation is the production of particles by the so-called “top-down” approach, such as laser ablation [
65], which is opposite to the chemical methods characterized by a “bottom-up” approach. These physical and chemical methods used for cobalt nanomaterial preparation showed a narrow range of size and controlled morphology, which are very suitable for the industry production of cobalt nanomaterials [
66]. However, the use of physical methods always requires high temperature or pressure, and most chemical methods need some chemicals that are toxic and hazardous to the environment and biological systems [
67]. Therefore, there is still a demand for the development of safer, greener, eco-friendly, and cost-effective synthetic methods that can eliminate the arduousness and complications of current physicochemical methods for cobalt nanomaterials [
9].
In the current scenario, green chemistry is known as an intellectual approach for nanomaterial preparation. The surging popularity of green methods has triggered the synthesis of Co NPs using different sources, such as bacteria, fungi, algae, and plants, resulting in large-scale production with less contamination [
68]. The bacterial synthesis of nanoparticles has been adopted due to the relative ease of manipulating the bacteria [
69]. The fungi-mediated approach exhibits unique advantages, as the growth process of fungi is easily handled and isolated, with the large amount of biomass and high yield of proteins [
64]. Compared with bacteria and fungi-based cobalt nanomaterial preparation methods, plant extracts have been extensively used to synthesize Co NPs as it is an inexpensive, biocompatible, and easy scale-up method that can fully avoid the requirement of additional stabilizing agents during the nanoparticle synthesis [
70]. For example, IsmatBibi et al. fabricated cobalt–oxide nanoparticles using Punica granatum peel extract from cobalt nitrate hexahydrate at low temperature [
71]. Furthermore, the nanoparticles obtained from plant extracts exhibit greater reduction and stabilization effects, which therefore allows the cobalt nanoparticles to show multiple properties, including catalyst/photocatalyst, magnetic, antibacterial, anticancer, and gas sensing [
72].
3. The Characteristics of Cobalt Nanomaterials
Nanomaterials, due to their excellent physical and chemical characteristics, have become one of the most rapidly growing research areas in the biomedical field in recent years [
73]. In broad terms, nanomaterials are inorganic, organic, or polymeric materials that possess physicochemical features with a size range of 1–1000 nm [
74]. The small size of nanomaterials allows them to easily distribute throughout the body, traverse biological barriers, and enter the systemic circulation [
75]. Nanoparticles always show large surface areas, which therefore can help them interact with biological systems more precisely [
76]. In addition, nanomaterials can be engineered to show different properties such as size, shape, charge, and surface chemistry [
77], which could lead to different applications in the biomedical fields [
78]. For example, nanomaterials allow molecular scale detection for the diagnostic application of pathogenic microbes [
79]. Additionally, small-sized nanomaterials always exhibit enhanced permeability and retention (EPR) effects in tumors, with relative increases in local tumor concentrations of contrasting agents [
80]. Such promising properties therefore make nanomaterials potential candidates for novel diagnostic and therapeutic method development.
There are various kinds of nanomaterials, including metal nanomaterials, ceramic nanomaterials, carbon-based nanomaterials, polymeric nanomaterials, and lipid-based nanomaterials [
75]. Among them, metal nanomaterials are widely used due to their low toxicity, biocompatibility, and excellent conductivity [
81]. For example, MNPs have been largely implemented to facilitate the conjugation with biomolecules for the improvement of biosensors [
82]. The NiCo@f-MWCNT nanocomposite has been proved to be a kind of novel sensor with high stability and excellent electrochemical properties [
83]. Recently, cobalt nanomaterials (Co NMs) have attracted considerable attention due to their low costs in preparation [
84], great electrical property, magnetic property, and catalytic property [
85]. For example, Mn
0.75Co
0.25Fe
2O
4 NPs can be utilized in industrial and environmental objects such as water treatment from the pollutant dyes due to their effective photocatalytic performance to protect the environment from toxic pigments [
86].
Co NMs are renowned catalysts, particularly in Fischer–Tropsch reactions [
87], with high Curie temperature, which makes it applicable over a wide temperature range [
88]. The high saturation magnetization and large anisotropy field of pure cobalt nanoparticles give them an intrinsic advantage within the strict requirements of hyperthermia [
89]. Co NMs have a variety of size-dependent structural, electrical, magnetic, and catalytic capabilities [
90]. Therefore, efforts have been conducted to attain various structures and sizes such as spheres, sheets, snowflakes or cauliflower like particles, and flakes [
91]. The instability of Co NMs is their main drawback, which can be diminished by using different stabilizers such as surfactants and dendrimers [
92]. Based on these features, Co NMs have demonstrated various biological and medical applications, such as antimicrobial, anticancer, antioxidant, anti-fungal, and enzyme inhibition properties [
9].
This entry is adapted from the peer-reviewed paper 10.3390/pharmaceutics14112351