Photoacoustics, being a noninvasive and functional modality, has the potential for small-animal imaging. However, the conventional photoacoustic tomographic systems use pulsed lasers, making it expensive, bulky, and require long acquisition time. We have developed a photoacoustic and ultrasound tomographic imaging system with LEDs as the light source and acoustic detection using a linear transducer array. We have demonstrated full-view tomographic imaging of a mouse.
- Photoacoustics
- Ultrasound
- Tomography
- Small animal imaging
- Light Emitting Diode (LED)
- Liver
- Fibrosis
1. Introduction
Studying different diseases and developing new drugs in a controlled environment is vital in biomedical research. Small animals are widely used for this purpose, as they allow for controlled disease staging and evaluating the performance of drugs through histopathological validation [1]. Longitudinal monitoring of disease progression and treatment response to drugs can improve the outcome of preclinical studies and can reduce the number of laboratory animal deaths. Imaging modalities can be used for longitudinal monitoring of small animal models. However, there are limitations in using conventional imaging modalities such as MRI, CT, and ultrasound for small animal imaging [2]. Micro MRI is costly and has a slow data acquisition. Micro CT and PET, on the other hand, use ionizing radiation, which hinders longitudinal observations [2]. Ultrasound (US) is a noninvasive and real-time imaging modality but, being a structural imaging modality, in most cases it cannot quantify disease state. Photoacoustic (PA) imaging is a new modality that has functional and molecular capability while being noninvasive and real-time. Thus, PA is considered to be ideal for small animal imaging [3].
PA imaging utilizes pulsed light excitation to induce a temperature rise in optical absorbing structures inside the tissue resulting in thermoelastic expansion and acoustic wave generation. These acoustic waves are detected for imaging [4]. The advantage of PA imaging is that with optical excitation and acoustic detection it combines optical contrast at ultrasound resolution. Additionally, the use of ultrasound transducers enables us to combine PA imaging with conventional US imaging providing co-registered structural and functional imaging of the tissue. The use of a pulsed laser source in these systems not only makes them expensive but demands laser safe small animal labs, eye safety goggles, and additional manpower to operate the system. Therefore, for the wide use of PA imaging in small animal labs, there is a requirement for low cost, compact, safe to use tomographic systems which can be operated by a non-expert. We have recently developed a tomographic imaging configuration using a linear transducer array and four LED arrays, to overcome the limited view and to improve the imaging depth [5,6,7].
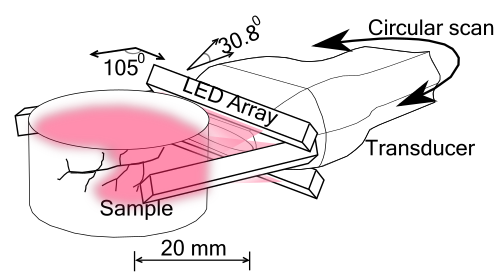
We have developed a system configuration as shown below.
Figure 1. (a) Tomographic system configuration with a linear transducer array and LED arrays.
We used a commercially PA and US handheld imaging system AcousticX (CYBERDYNE Inc., Japan) for tomographic imaging. A linear transducer array with 128 elements was used for the experiments. Four LED array units having 576 elements (36 × 4 in each array) at 850 nm wavelength were used for illumination. The LED arrays were arranged as shown in Fig. 1. Two other LED bars were placed in the imaging plane, with an angle of 105o and −105o relative to the transducer array, with the left LED array unit looking downwards with 5o and the right LED array looking upwards with 5o.
To demonstrate the tomographic capability of the system a leaf phantom was imaged as shown below. B-mode images from multiple angles were spatially compounded to from the tomographic image.

(a)
(b)
Figure 2. (a) Leaf skeleton stained with India ink. (b) Spatial compounding of B-mode images from multiple angles to form the tomographic image.
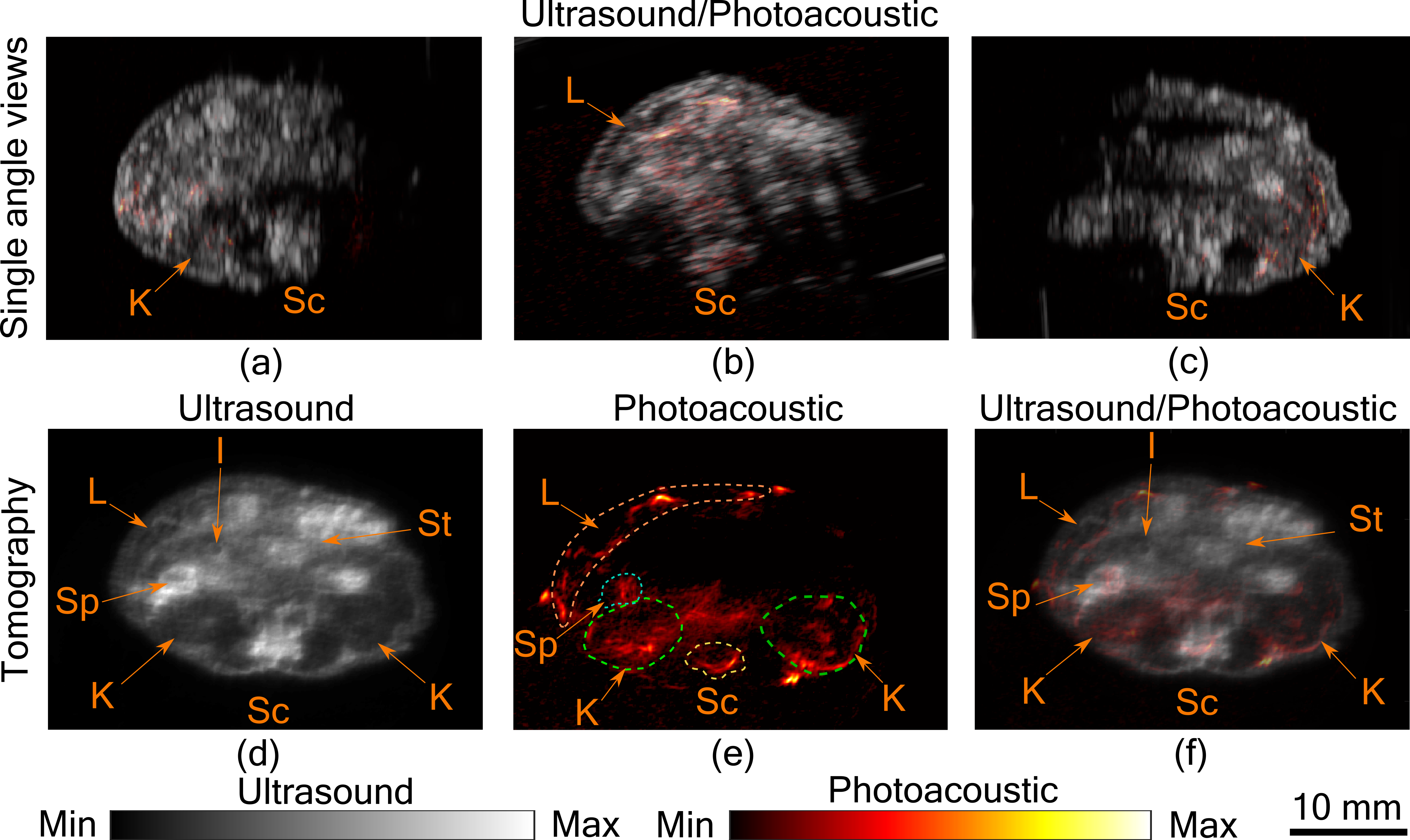
In Figure 3(a)–(c), co-registered ultrasound and photoacoustic B-scan images of a mouse abdomen, obtained from three different angles, have been presented. The images demonstrate the limited view problem in small animal imaging using a linear transducer array. The limited view arises from the directional sensitivity of the transducer resulting in structures oriented away from the transducer being undetected. The tomographic images obtained from spatial compounding of the B-scan images are shown in Figure 3(d)–(f). The ultrasound image shows the capability of the tomographic system to image major abdominal organs such as the liver, kidney, spleen, stomach, and intestines. In PA images major blood vessels are visible beneath the skin and around the spinal cord. In addition to that, high PA signals were also observed from the liver, kidney, and spleen.
Figure 3. Tomographic photoacoustic and ultrasound imaging. (a–c) Co-registered ultrasound and photoacoustic B-Scan images of mouse abdomen, acquired from three different angles. Tomographic (d) ultrasound, (e) photoacoustic, and (f) co-registered ultrasound and photoacoustic image. Several organs in the abdominal region indicated by Sc-spinal cord, K-kidney, L-liver, Sp -spleen, St-stomach, and I-intestine. Dashed lines are used to mark the organs in the photoacoustic image.
2. Conclusion
In this entry, we have demonstrated a tomographic ultrasound and LED-based photoacoustic system for small animal imaging. Results show that the tomographic ultrasound imaging can be used to distinguish major organs in the abdominal region of a small animal. The tomographic photoacoustic imaging is capable of imaging an approximate depth of 10 mm from the surface of the skin, allowing imaging of blood vessels and major organs. Interested readers can check more about our system configuration [8], its application in finger joint imaging [5] and in liver fibrosis research [7].
This entry is adapted from the peer-reviewed paper 10.3390/s20102793