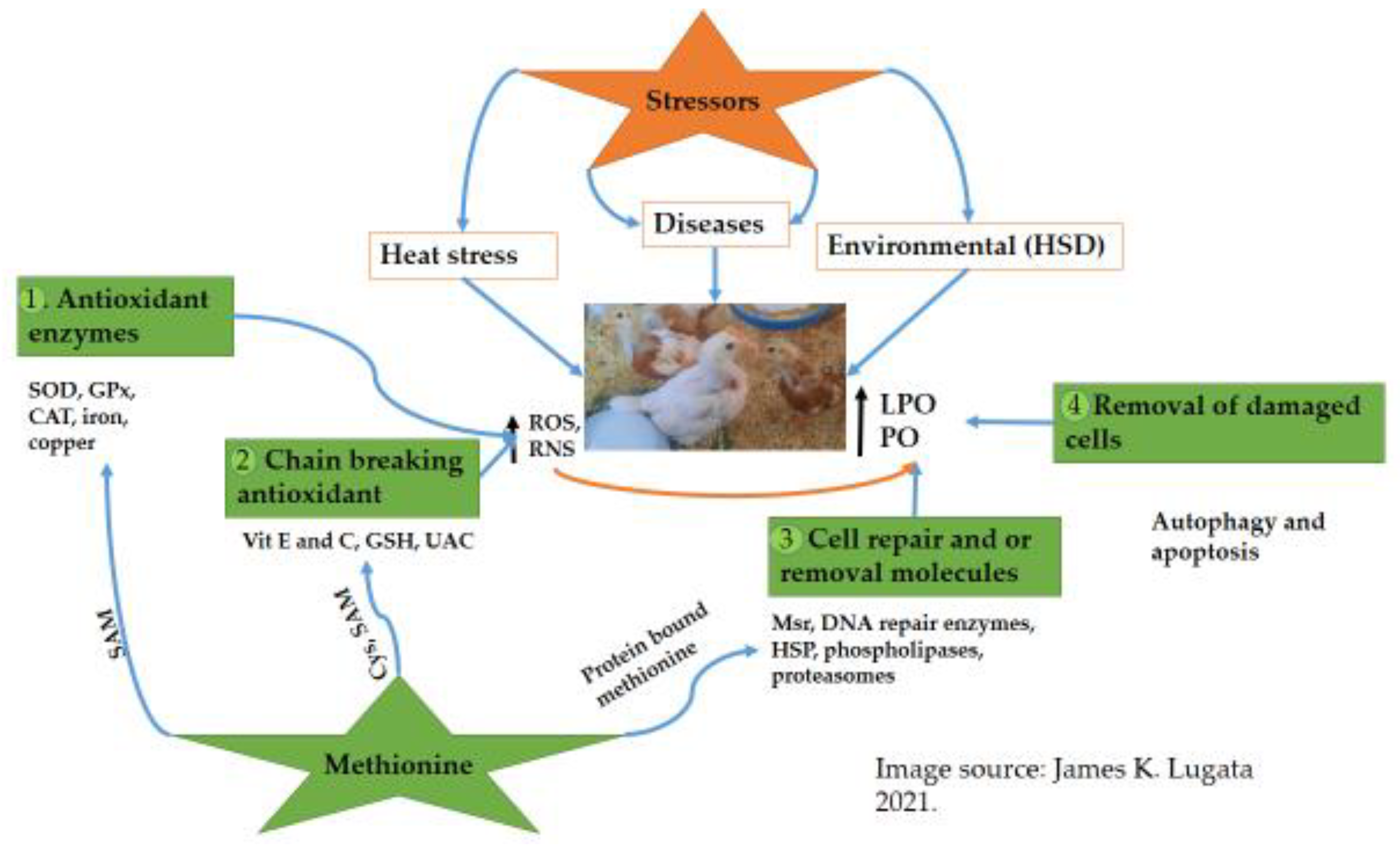
The physiological status of poultry can be disturbed by different stressors that may lead to oxidative stress conditions. Oxidative stress activates defense systems, which mitigates the adverse effects. Several lines of the poultry defense system exist, including enzyme systems such as catalase (CAT), superoxide dismutase (SOD), glutathione peroxidase (GPx), and non-enzymatic antioxidants such as Glutathione (GSH). Methionine—a vital amino acid in poultry nutrition—plays a significant role in protein synthesis, transsulfuration, and transmethylation and is also involved in several biochemical pathway activations that can affect the antioxidant system.
- poultry
- antioxidant defense system
- methionine sources
- oxidative stress
1. Introduction

2. Methionine Supplementation of Poultry
3. The Antioxidant System of Poultry
4. How Antioxidants Network Work under Oxidative Stress
This entry is adapted from the peer-reviewed paper 10.3390/agriculture12101701
References
- Havenstein, G.B.; Ferket, P.R.; Qureshi, M.A. Growth, Livability, and Feed Conversion of 1957 versus 2001 Broilers When Fed Representative 1957 and 2001 Broiler Diets. Poult. Sci. 2003, 82, 1500–1508.
- Djumadil, N.; Syafie, Y. Analyses of Factors Affecting the Production of Broiler Chickens in Ternate. In Proceedings of the 5th International Conference on Food, Agriculture and Natural Resources (FANRes 2019), Ternate, Indonesia, 17–18 September 2019; Atlantis Press: Dordrecht, The Netherlands, 2020.
- Baracho, M.S.; Nääs, I.A.; Lima, N.D.S.; Cordeiro, A.F.S.; Moura, D.J. Factors Affecting Broiler Production: A Meta-Analysis. Braz. J. Poult. Sci. 2019, 21, 001–010.
- Zhao, P.Y.; Kim, I.H. Effect of Diets with Different Energy and Lysophospholipids Levels on Performance, Nutrient Metabolism, and Body Composition in Broilers. Poult. Sci. 2017, 96, 1341–1347.
- Das, T.K.; Mondal, M.K.; Biswas, P.; Bairagi, B.; Samanta, C.C. Influence of Level of Dietary Inorganic and Organic Copper and Energy Level on the Performance and Nutrient Utilization of Broiler Chickens. Asian-Australas. J. Anim. Sci. 2009, 23, 82–89.
- Maharjan, P.; Mullenix, G.; Hilton, K.; Caldas, J.; Beitia, A.; Weil, J.; Suesuttajit, N.; Kalinowski, A.; Yacoubi, N.; Naranjo, V.; et al. Effect of Digestible Amino Acids to Energy Ratios on Performance and Yield of Two Broiler Lines Housed in Different Grow-out Environmental Temperatures. Poult. Sci. 2020, 99, 6884–6898.
- Liu, G.; Magnuson, A.D.; Sun, T.; Tolba, S.A.; Starkey, C.; Whelan, R.; Lei, X.G. Supplemental Methionine Exerted Chemical Form-Dependent Effects on Antioxidant Status, Inflammation-Related Gene Expression, and Fatty Acid Profiles of Broiler Chicks Raised at High Ambient Temperature. J. Anim. Sci. 2019, 97, 4883–4894.
- Magnuson, A.D.; Liu, G.; Sun, T.; Tolba, S.A.; Xi, L.; Whelan, R.; Lei, X.G. Supplemental Methionine and Stocking Density Affect Antioxidant Status, Fatty Acid Profiles, and Growth Performance of Broiler Chickens. J. Anim. Sci. 2020, 98, skaa092.
- Lara, L.J.; Rostagno, M.H. Impact of Heat Stress on Poultry Production. Animals 2013, 3, 356–369.
- Selye, H. Forty Years of Stress Research: Principal Remaining Problems and Misconceptions. Can. Med. Assoc. J. 1976, 115, 53–56.
- Yunianto, V.D.; Hayashi, K.; Kaneda, S.; Ohtsuka, A.; Tomita, Y. Effect of Environmental Temperature on Muscle Protein Turnover and Heat Production in Tube-Fed Broiler Chickens. Br. J. Nutr. 1997, 77, 897–909.
- Halliwell, B.; Gutteridge, J.M.C. Free Radicals in Biology and Medicine, 5th ed.; Oxford University Press: New York, NY, USA, 2015.
- Fry, J.L.; Stadelman, W.J. The Effect of Dietary Methionine on the Methionine and Cystine Content of Poultry Meat. Poult. Sci. 1960, 39, 614–617.
- Graber, G.; Scott, H.M.; Baker, D.H. Sulfur Amino Acid Nutrition of the Growing Chick: Effect of Age on the Dietary Methionine Requirement. Poult. Sci. 1971, 50, 854–858.
- Bao, Y. Amino Acid Nutrition and Chicken Gut Health. Worlds Poult. Sci. J. 2020, 76, 563–576.
- Stipanuk, M.H. Metabolism of Sulfur-Containing Amino Acids: How the Body Copes with Excess Methionine, Cysteine, and Sulfide. J. Nutr. 2020, 150, 2494S–2505S.
- Zhang, S.; Saremi, B.; Gilbert, E.R.; Wong, E.A. Physiological and Biochemical Aspects of Methionine Isomers and a Methionine Analogue in Broilers. Poult. Sci. 2017, 96, 425–439.
- Ren, Z.; Bütz, D.E.; Whelan, R.; Naranjo, V.; Arendt, M.K.; Ramuta, M.D.; Yang, X.; Crenshaw, T.D.; Cook, M.E. Effects of Dietary Methionine plus Cysteine Levels on Growth Performance and Intestinal Antibody Production in Broilers during Eimeria Challenge. Poult. Sci. 2020, 99, 374–384.
- Sigolo, S.; Deldar, E.; Seidavi, A.; Bouyeh, M.; Gallo, A.; Prandini, A. Effects of Dietary Surpluses of Methionine and Lysine on Growth Performance, Blood Serum Parameters, Immune Responses, and Carcass Traits of Broilers. J. Appl. Anim. Res. 2019, 47, 146–153.
- Zhang, S.; Gilbert, E.R.; Saremi, B.; Wong, E.A. Supplemental Methionine Sources Have a Neutral Impact on Oxidative Status in Broiler Chickens. J. Anim. Physiol. Anim. Nutr. 2018, 102, 1274–1283.
- Akter, N.; Islam, M.; Zaman, S.; Jahan, I.; Hossain, M. The Impact of Different Levels of L-Methionine (L-Met) on Carcass Yield Traits, Serum Metabolites, Tibial Characters, and Profitability of Broilers Fed Conventional Diet. J. Adv. Vet. Anim. Res. 2020, 7, 253.
- Esteve-Garcia, E.; Khan, D.R. Relative Bioavailability of DL and L-Methionine in Broilers. Open J. Anim. Sci. 2018, 8, 151–162.
- Jankowski, J.; Ognik, K.; Kubińska, M.; Czech, A.; Juśkiewicz, J.; Zduńczyk, Z. The Effect of DL-, L-Isomers and DL-Hydroxy Analog Administered at 2 Levels as Dietary Sources of Methionine on the Metabolic and Antioxidant Parameters and Growth Performance of Turkeys. Poult. Sci. 2017, 96, 3229–3238.
- Murawska, D.; Kubińska, M.; Gesek, M.; Zduńczyk, Z.; Brzostowska, U.; Jankowski, J. The Effect of Different Dietary Levels and Sources of Methionine on the Growth Performance of Turkeys, Carcass and Meat Quality. Ann. Anim. Sci. 2018, 18, 525–540.
- Park, I.; Pasquetti, T.; Malheiros, R.D.; Ferket, P.R.; Kim, S.W. Effects of Supplemental L-Methionine on Growth Performance and Redox Status of Turkey Poults Compared with the Use of DL-Methionine. Poult. Sci. 2018, 97, 102–109.
- Li, L.; Abouelezz, K.F.M.; Cheng, Z.; Gad-Elkareem, A.E.G.; Fan, Q.; Ding, F.; Gao, J.; Jiang, S.; Jiang, Z. Modelling Methionine Requirements of Fast- and Slow-Growing Chinese Yellow-Feathered Chickens during the Starter Phase. Animals 2020, 10, 443.
- National Research Council (U.S.). Nutrient Requirements of Poultry. In Nutrient Requirements of Domestic Animals, 9th ed.; National Academy Press: Washington, DC, USA, 1994; ISBN 978-0-309-04892-7.
- Rehman, A.U.; Arif, M.; Husnain, M.M.; Alagawany, M.; Abd El-Hack, M.E.; Taha, A.E.; Elnesr, S.S.; Abdel-Latif, M.A.; Othman, S.I.; Allam, A.A. Growth Performance of Broilers as Influenced by Different Levels and Sources of Methionine Plus Cysteine. Animals 2019, 9, 1056.
- Tian, Q.Y.; Zeng, Z.K.; Zhang, Y.X.; Long, S.F.; Piao, X.S. Effect of L- or DL-Methionine Supplementation on Nitrogen Retention, Serum Amino Acid Concentrations and Blood Metabolites Profile in Starter Pigs. Asian-Australas. J. Anim. Sci. 2016, 29, 689–694.
- Ullrich, C.; Langeheine, M.; Brehm, R.; Taube, V.; Rosillo Galera, M.; Rohn, K.; Popp, J.; Visscher, C. Influence of Different Methionine Sources on Performance and Slaughter Characteristics of Broilers. Animals 2019, 9, 984.
- Agostini, P.S.; Dalibard, P.; Mercier, Y.; Van der Aar, P.; Van der Klis, J.D. Comparison of Methionine Sources around Requirement Levels Using a Methionine Efficacy Method in 0 to 28 Day Old Broilers. Poult. Sci. 2016, 95, 560–569.
- Fouad, A.M.; Ruan, D.; Lin, Y.C.; Zheng, C.T.; Zhang, H.X.; Chen, W.; Wang, S.; Xia, W.G.; Li, Y. Effects of Dietary Methionine on Performance, Egg Quality and Glutathione Redox System in Egg-Laying Ducks. Br. Poult. Sci. 2016, 57, 818–823.
- Zduńczyk, Z.; Jankowski, J.; Kubińska, M.; Ognik, K.; Czech, A.; Juśkiewicz, J. The Effect of Different Dietary Levels of Dl-Methionine and Dl-Methionine Hydroxy Analogue on the Antioxidant and Immune Status of Young Turkeys. Arch. Anim. Nutr. 2017, 71, 347–361.
- Murillo, M.G.; Jensen, L.S.; Ruff, M.D.; Rahn, A.P. Effect of Dietary Methionine Status on Response of Chicks to Coccidial Infection. Poult. Sci. 1976, 55, 642–649.
- Meirelles, H.T.; Albuquerque, R.; Borgatti, L.M.O.; Souza, L.W.O.; Meister, N.C.; Lima, F.R. Performance of Broilers Fed with Different Levels of Methionine Hydroxy Analogue and DL-Methionine. Braz. J. Poult. Sci. 2003, 5, 69–74.
- Swennen, Q.; Geraert, P.-A.; Mercier, Y.; Everaert, N.; Stinckens, A.; Willemsen, H.; Li, Y.; Decuypere, E.; Buyse, J. Effects of Dietary Protein Content and 2-Hydroxy-4-Methylthiobutanoic Acid or DL-Methionine Supplementation on Performance and Oxidative Status of Broiler Chickens. Br. J. Nutr. 2011, 106, 1845–1854.
- Yodseranee, R.; Bunchasak, C. Effects of Dietary Methionine Source on Productive Performance, Blood Chemical, and Hematological Profiles in Broiler Chickens under Tropical Conditions. Trop. Anim. Health Prod. 2012, 44, 1957–1963.
- Xiao, X.; Wang, Y.; Liu, W.; Ju, T.; Zhan, X. Effects of Different Methionine Sources on Production and Reproduction Performance, Egg Quality and Serum Biochemical Indices of Broiler Breeders. Asian-Australas. J. Anim. Sci. 2017, 30, 828–833.
- Albrecht, A.; Herbert, U.; Miskel, D.; Heinemann, C.; Braun, C.; Dohlen, S.; Zeitz, J.O.; Eder, K.; Saremi, B.; Kreyenschmidt, J. Effect of Methionine Supplementation in Chicken Feed on the Quality and Shelf Life of Fresh Poultry Meat. Poult. Sci. 2017, 96, 2853–2861.
- Jankowski, J.; Tykałowski, B.; Ognik, K.; Koncicki, A.; Kubińska, M.; Zduńczyk, Z. The Effect of Different Dietary Levels of DL-Methionine and DL-Hydroxy Analogue on the Antioxidant Status of Young Turkeys Infected with the Haemorrhagic Enteritis Virus. BMC Vet. Res. 2018, 14, 404.
- Wan, J.; Ding, X.; Wang, J.; Bai, S.; Peng, H.; Luo, Y.; Su, Z.; Xuan, Y.; Zhang, K. Dietary Methionine Source and Level Affect Hepatic Sulfur Amino Acid Metabolism of Broiler Breeder Hens. Anim. Sci. J. 2017, 88, 2016–2024.
- Sangali, C.P.; Bruno, L.D.G.; Nunes, R.V.; de Oliveira Neto, A.R.; Pozza, P.C.; de Oliveira, T.M.M.; Frank, R.; Schöne, R.A. Bioavailability of Different Methionine Sources for Growing Broilers. R. Bras. Zootec. 2014, 43, 140–145.
- Millecam, J.; Khan, D.R.; Dedeurwaerder, A.; Saremi, B. Optimal Methionine plus Cystine Requirements in Diets Supplemented with L-Methionine in Starter, Grower, and Finisher Broilers. Poult. Sci. 2021, 100, 910–917.
- Shen, Y.B.; Ferket, P.; Park, I.; Malheiros, R.D.; Kim, S.W. Effects of Feed Grade L-Methionine on Intestinal Redox Status, Intestinal Development, and Growth Performance of Young Chickens Compared with Conventional DL-Methionine. J. Anim. Sci. 2015, 93, 2977–2986.
- Martínez, Y.; Li, X.; Liu, G.; Bin, P.; Yan, W.; Más, D.; Valdivié, M.; Hu, C.-A.A.; Ren, W.; Yin, Y. The Role of Methionine on Metabolism, Oxidative Stress, and Diseases. Amino Acids 2017, 49, 2091–2098.
- Surai, P.F.; Kochish, I.I.; Fisinin, V.I.; Kidd, M.T. Antioxidant Defence Systems and Oxidative Stress in Poultry Biology: An Update. Antioxidants 2019, 8, 235.
- Horváth, M.; Babinszky, L. Impact of Selected Antioxidant Vitamins (Vitamin A, E and C) and Micro Minerals (Zn, Se) on the Antioxidant Status and Performance under High Environmental Temperature in Poultry. A Review. Acta Agric. Scand. Sect. A—Anim. Sci. 2018, 68, 152–160.
- Mishra, B.; Jha, R. Oxidative Stress in the Poultry Gut: Potential Challenges and Interventions. Front. Vet. Sci. 2019, 6, 60.
- Luo, S.; Levine, R.L. Methionine in Proteins Defends against Oxidative Stress. FASEB J. 2009, 23, 464–472.
