Your browser does not fully support modern features. Please upgrade for a smoother experience.
Please note this is an old version of this entry, which may differ significantly from the current revision.
Subjects:
Others
Alteration in gut microbiota has been observed in COVID-19 patients. However, the underlying mechanisms remain poorly understood. Here, we outlined three potential interconnected pathways leading to gut dysbiosis as an adverse outcome following SARS-CoV-2 presence in the gastrointestinal tract.
- SARS-CoV-2 infection
- COVID-19
- gut dysbiosis
- microbiota
- gastrointestinal disorders
1. Introduction
Coronavirus disease 2019 (COVID-19) caused by severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) is still a global public health emergency. A better understanding of the mechanisms underlying the progression and severity of the disease is needed. Particularly, COVID-19 is markedly heterogeneous in terms of clinical outcomes, with a high variation at the individual level. Poor clinical outcomes in COVID-19 patients were notably associated with elderliness and certain pre-existing medical conditions, including but not limited to diabetes, cardiovascular diseases, obesity, and high LDH levels [1,2,3,4,5]. Older age and the comorbidities mentioned above are associated with alterations in the gut microbiota [6,7,8]. Besides, COVID-19 patients exhibit fecal microbiome alterations compared to controls [9,10,11,12]. These changes correlated to COVID-19 severity [12]. Gut dysbiosis, defined as a reduction in gut microbiota diversity or the depletion of beneficial bacteria with an enrichment of the pathogenic ones, may alter susceptibility to SARS-CoV-2 infection [13,14,15]. This is aligned with the evidence that many pathophysiological dimensions of diseases are underpinned by the gut microbiota, especially in chronic inflammatory diseases [16] such as inflammatory bowel diseases (IBD). Although the exact etiologies of IBD remain uncertain, many studies have provided important insights into the central role of gut dysbiosis and barrier dysfunction in inflammatory status [17,18]. The gut microbiota plays an essential role in the education and functions of both the local and systemic immune systems. Besides, emerging evidence has demonstrated important cross-talks between the gut microbiota and many other organs via communication axes such as the gut–lung [19], gut–liver [20,21], and gut–brain [22] axes. Notably, gut dysbiosis during respiratory viral infection has been shown to worsen pulmonary symptoms [23]. Similarly, gut dysbiosis and disrupted intestinal barrier can cause neurological inflammation [22] or hepatic inflammation through the translocation of endotoxins and bacteria via the portal vein [24]. Consistently, taking into account gut microbiome-mediated mechanisms may help depict a comprehensive overview of COVID-19 pathogenesis. Exploring how gut dysbiosis as a pre-existing condition in some COVID-19 patients mechanistically influences the disease progression and impacts the clinical outcomes might help identify high-risk patients, and has been discussed elsewhere [5]. Here, we aim to investigate how SARS-CoV-2 might directly alter the gut microbiota, thus considering gut dysbiosis as a direct consequence of the virus in the gastrointestinal (GI) tract. Recently, animal studies have provided evidence for a direct impact of SARS-CoV-2 infection on the gut microbiota. A study conducted in transgenic mice expressing human ACE2 showed that the gut microbiome is affected by SARS-CoV-2 in a dose-dependent manner after intranasal inoculation [25]. In Syrian hamsters, SARS-CoV-2 infection was associated with mild intestinal inflammation, relative alteration of the intestinal barrier property, and alteration of the gut microbiota [26]. SARS-CoV-2 infection in nonhuman primates was associated with changes in the gut microbiota composition and functional activity [27]. However, despite the dynamic research, the underlying pathways leading to gut dysbiosis in COVID-19 are still poorly understood.
To contribute to deciphering these mechanisms, the Joint Research Centre of the European Commission initiated an interdisciplinary project, the CIAO project, to model the pathogenesis of COVID-19 using the Adverse Outcome Pathway (AOP) framework [28,29,30,31]. The AOP approach is well established in regulatory toxicology [32] but is innovatively applied here to a viral disease of high societal relevance. The project relies on the assumption that an AOP-driven organization of the relevant knowledge will improve the integration of the tsunami of data on COVID-19 [28]. The AOP approach does not capture all the details in a biological pathway, but aims for a pragmatic identification of successively linked key events (KE) that represent essential steps in a pathway leading to an adverse outcome [33,34,35,36]. A key event describes a measurable and essential change in a biological system that can be quantified in experimental or clinical settings [32]. The AOP framework also provides a structured approach for the evaluation of the level of evidence currently available to ascertain the causal relationships between pairs of successive key events [37]. AOPs do not build on the correlation between two events but gather and weigh the evidence for their causal relationship. Because of this mechanistic and causal description of the pathways, AOPs help elucidate the pathophysiological mechanisms also by learning from other diseases, such as IBD or respiratory virus-related diseases presenting gut dysbiosis. Finally, an AOP integrates knowledge across the different biological levels (from molecular, cellular, tissue, organ, and up to organism level). While research tends to compartmentalize in silos, this pandemic calls for an interdisciplinary integration of data from the different experimental systems. Hence, the AOP approach allows the structured review and organization of rapidly growing relevant in vitro, in vivo, and clinical data. Assessing the evidence currently available using the AOP framework permits the identification of critical inconsistencies and knowledge gaps guiding future research needs. The AOPs are steered by the Organization for Economic Co-operation and Development (OECD), which maintains a centralized online platform called AOP wiki (https://aopwiki.org/ accessed on 29 June 2022), where information captured in AOPs is openly accessible. Numbers in the text refer to these AOP-wiki pages (Table 1).
Table 1. AOP-wiki pages.
| KER1739 | https://aopwiki.org/events/1739 | accessed on 29 June 2022 |
| KER1738 | https://aopwiki.org/events/1738 | accessed on 29 June 2022 |
| KER1847 | https://aopwiki.org/events/1847 | accessed on 29 June 2022 |
| KER1901 | https://aopwiki.org/events/1901 | accessed on 29 June 2022 |
| KER1493 | https://aopwiki.org/events/1493 | accessed on 29 June 2022 |
| KER1497 | https://aopwiki.org/events/1497 | accessed on 29 June 2022 |
| KER1954 | https://aopwiki.org/events/1954 | accessed on 29 June 2022 |
| KER2311 | https://aopwiki.org/events/2311 | accessed on 29 June 2022 |
2. Central Role of Gut Microbiota in COVID-19 and Potential Modulation
The three above proposed pathways leading to an alteration of gut microbiota following SARS-CoV-2 presence in the gut lumen are non-mutually exclusive but rather interconnected (Figure 4).
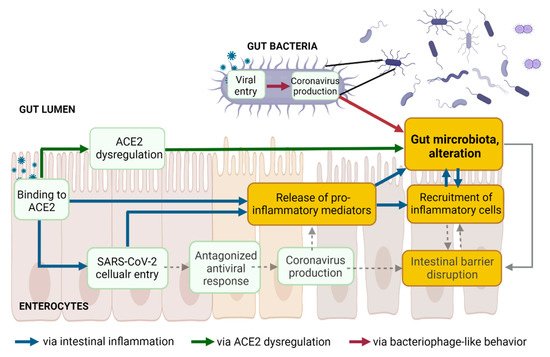
Figure 4. The three proposed pathways leading to gut dysbiosis following SARS-CoV-2 presence in the gut lumen are not mutually exclusive but might be interconnected. Created with Biorender.com.
2.1. Gut Microbiota and Intestinal Barrier Integrity in COVID-19
Together with the mucosal barrier and the cellular immune system, the intestinal epithelial cell monolayer and the tight junction proteins act simultaneously as a physical barrier against harmful external substances, as well as a selective barrier. Increased intestinal permeability, a sign of an impaired barrier function, enhances the translocation of gut bacteria and bacterial toxins from the intestinal lumen into the systemic circulation. The gut microbiota ensures intestinal barrier integrity through diverse mechanisms [48] (Figure 4, dashed grey lines). Beneficial butyrate-producing bacteria are proposed to maintain intestinal integrity, as butyrate, a short-chain fatty acid (SCFA), facilitates the regeneration of healthy colonocytes [49]. A reduced relative proportion of bacteria producing SCFA was observed in Syrian hamsters infected with SARS-CoV-2, compared to non-infected controls, with a transient decrease in systemic SCFA amounts [26]. Decreases in the abundance of butyrate-producing bacteria and a decline in SCFA were observed in severe COVID-19 [10,12,133]. Besides the reduction of beneficial bacteria, the overgrowth of pathobionts, such as Escherichia coli or Salmonella enterica, disrupts intestinal barrier function [134,135,136]. Outgrowth of pathogenic Prevotella has been associated with reduced mucus secretion, one crucial protective layer of the intestinal barrier [137]. Blooms of pathogenic bacteria have been observed in hospitalized COVID-19 patients, along with the translocation of gut bacteria into the blood [25]. Lowered levels of butyrate-producers and higher levels of opportunistic pathogens (including E. coli and S. enterica) were observed in COVID-19 patients compared with H1N1 patients and healthy controls [9]. In addition, gut microbiota composition correlated with plasma levels of tissue damage markers, altered tight junctions, and microbial translocation in COVID-19 patients [10]. Finally, the colonic mucus barrier is shaped by the composition of the gut microbiota [138]. Alteration of the gut microbiota might contribute to disrupting the mucus barrier.
Human intestinal organoid co-cultures with microbes could represent useful systems to investigate the protective function of bacteria on gut permeability upon SARS-CoV-2 infection [139]. In addition, similar to the treatment of other diseases, treating SARS-CoV-2 infected mice or Syrian hamsters with SCFA supplementation [26,51], prebiotics, or probiotics (such as Lactobacillus reuteri in rodents), [140] and evaluating the intestinal permeability (dextran and bacterial translocation) in parallel with microbiota omics could strengthen our understanding of the relationship between gut microbiota and the intestinal barrier in COVID-19 pathophysiology.
2.2. Central Role of the Gut (Microbiota) in COVID-19 and Long COVID
Dysbiosis, intestinal inflammation, and leaky gut are intimately interconnected (Figure 4) and intestinal homeostasis is increasingly recognized as an underpinning clinical driver in several noncommunicable diseases as well as in COVID-19. Accumulating evidence supports that altered gut microbiota and associated leaky gut may contribute to the GI symptoms and the cytokine storm and multiorgan complications in COVID-19 [141,142]. In critically ill patients with sepsis and respiratory distress, bacterial translocation is widely documented [143,144]. Higher plasma levels of gut permeability markers were found in COVID-19 patients, along with abnormal presence of gut bacteria in the blood [145,146]. These markers correlated with higher levels of C-reactive peptide (a marker of hyperinflammation) and with a higher mortality rate [146]. Serum levels of lipopolysaccharide-binding protein were higher in patients with severe COVID-19 and were associated with circulating inflammation biomarkers [147]. Altered intestinal homeostasis induces diarrhea [148], which is the digestive symptom most commonly reported in COVID-19 patients [149,150,151,152,153].
Despite the well-documented prevalence of GI symptoms and the high rate of SARS-CoV-2 fecal RNA shedding, the isolation of replication-competent virus from fecal samples has not been reproducibly and systematically demonstrated [38]. The biological, clinical, and epidemiological relevance of SARS-CoV-2 shedding remains unclear [154]. SARS-CoV-2 shedding in stools has been reported from one week to seven months after diagnosis [154,155]. The prolonged presence of viral RNA in feces [154], but not in respiratory samples, and its association with GI symptoms suggests that SARS-CoV-2 infects the GI tract, and that this infection can be prolonged in a subset of individuals with COVID-19. SARS-CoV-2 infection leading to perturbation of the gut microbiome may contribute to the underlying etiology of GI symptoms observed in COVID-19 and long COVID [45,156]. Alteration in the gut microbiome persists long after a patient recovers, suggesting that the gut microbiome may play an important role in long COVID [157]. Long COVID or post-acute COVID-19 syndrome (PACS) is rapidly emerging across the globe and many studies following patients who have recovered from the respiratory effects of COVID-19 identified persistent GI sequelae, including dysbiosis [154,155,158]. While the pathogenesis of long COVID is still under intense investigation, on the four current leading hypotheses [45], it is interesting to note that gut dysbiosis is considered as one of them [157,159]. A comprehensive understanding of the dynamics of fecal clearance of SARS-CoV-2 RNA and its link with gut dysbiosis is currently lacking. Further studies are needed as the gut microbiota could serve as a potential prognosis indicator and could be therapeutically valuable.
2.3. Potential Modulation of Gut Microbiota to Mitigate COVID-19
In light of the current insight into the central role of the gut in COVID-19 and long COVID, modulating the gut microbiota to improve disease prevention and management may be relevant. First, fecal microbiota transplantation (FMT) enables stool infusion from a healthy individual to a severely ill patient to restore intestinal microbial balance [160]. So far, FMT has been remarkably successful in the treatment of Clostridium difficile infection, but much less in treating other conditions, such as IBD or metabolic disorders. COVID-19 being an infectious disease and not an inflammatory disorder, FMT could be more successful [141]. However, COVID-19 could potentially be transmitted via FMT, particularly from asymptomatic donors who tested negative for the presence of the virus in their respiratory tract but positive in their stools [161]. No cases of COVID-19 transmission through FMT have been reported so far, but only FMT products generated from stools donated before December/November 2019 were used according to the FDA and Hong Kong recommendations, respectively. Secondly, gut microbiota modulation with probiotics, prebiotics, or diet and therapies preventing gut barrier defects may represent easy-to-implement strategies to mitigate COVID-19 [162]. Clinical trials of probiotics with expected anti-inflammatory effects for preventing or treating SARS-CoV-2 infection are currently ongoing [163]. Next-generation probiotics focusing on butyrate-producing bacteria, or simply increasing the daily intake of dietary fiber are proposed as potential beneficial approaches for COVID-19 patients [141]. A few reports cite indirect evidence for the association between probiotics and COVID-19, primarily based on previous coronaviruses and other viral infections [164,165]. The health benefits of prebiotics to the GI tract, including the inhibition of pathogens and stimulation of the immune system, are due to their ability to modulate the composition and activity of human microbiota [166,167,168]. However, to date, there is no information directly linking prebiotics to COVID-19 infections, although an indirect effect may be hypothesized [169]. Thus, using conventional probiotics is not currently warranted, but is considered promising, and a better understanding of SARS-CoV-2 pathogenesis and its mutual effect on gut microbiota is needed. More generally, diet is obviously a factor impacting gut microbiota [170,171,172,173]. Dietary adaptation may be the easiest method to be implemented in the preventive arsenal against COVID-19 and for general health improvement [141].
This entry is adapted from the peer-reviewed paper 10.3390/jcm11185400
This entry is offline, you can click here to edit this entry!
