Your browser does not fully support modern features. Please upgrade for a smoother experience.
Please note this is an old version of this entry, which may differ significantly from the current revision.
Sjögren syndrome (SS) belongs to the family of rheumatic autoimmune diseases characterized by systemic compromise with exocrine glands as target organs that are affected by chronic inflammation and immune-mediated destruction of the tissue, leading to severe dryness of the mouth and eyes. Extra-glandular symptoms are frequent and include fatigue, polyarthralgias, myositis, polyneuropathy, and gammaglobulinopathies, among others.
- Sjögren syndrome
- nuclear medicine
- radiopharmaceuticals
1. Introduction
Since there is evidence of the presence of common pathophysiologic mechanisms shared by SS and thyroiditis, it is also common to find thyroid involvement in patients with SS as part of the peri-epithelial extra salivary manifestations of the disease [1]. Systemic complications regarding extra-glandular tissues are associated with vascular, respiratory, gastrointestinal, renal, and neurological systems, possibly affecting one-third of these patients [2].
SS may be present as a primary condition or accompanying other autoimmune diseases as secondary SS. In addition, SS has a predominant clinical presentation in females, with a female:male ratio of 9:1, higher than for all other autoimmune diseases [2]. The clinical phenotype of SS varies from benign conditions such as mild exocrinopathies to severe systemic manifestations such as B-cell non-Hodgkin’s lymphoma (NHL) [3][4]. These lymphomas arise predominantly from memory B cells in the marginal zone of lymphoid tissue, with mucosa-associated lymphoid tissue (MALT) lymphomas being the most frequent type [5] and the parotid and minor salivary glands being the most frequent sites of involvement [6]. Patients at high risk of developing NHL are male and have clinical manifestations of severe systemic disease, such as vasculitis, splenomegaly, lymphadenopathy, glomerulonephritis, haematologic manifestations, high expression of autoantibodies, cryoglobulins and hypergammaglobulinemia, high biopsy focus scores, and presence of ectopic germinal centers, among others [7].
2. Role of New Molecular Image Strategies in Sjogren’s Syndrome
The goal of SS is to be able to stratify pSS through the integration of clinical, laboratory, histopathology, and imaging data; thus, it could be possible to identify which patients have clinical manifestations related to inflammatory activity or sequelae; the possibility to identify which patients are on the risk of lymphoma development it is also important. Because new treatment strategies are emerging, there is also evident the need for new probes for a more reliable treatment response evaluation [8].
The potential for diagnosis and classification of pSS of the salivary glands ultrasound (SGUS) has been pointed out previously by different authors [9]; nowadays this technique is emerging as a complementary tool for biopsy purposes. In a recent study the authors reported that by adding SGUS as a minor item to ACR/EULAR criteria, the sensitivity could rise to 95.6% [10]. Other authors have reported that a combined positivity of SGUS and anti-SSA antibodies provides a high predictive value for the diagnosis of pSS [11] The main limitation of SGUS is the lack of a standardized scoring system, however, as an initiative to overcome this issue, the Outcome Measures in Rheumatology Clinical Trials (OMERACT) proposed a four-grade semiquantitative score with good intra and interobserver agreement results [12]. Last generation ultra-high resolution ultrasound (UHFUS) transducers, which produce frequencies up to 70 MHz with resolution up to 30 um, offer new possibilities to visualize labial salivary glands and to guide diagnostic biopsy procedures [13]. Recently, Baldini et al. demonstrated that the mean labial glandular surface area obtained by the high-resolution ultrasound-guided procedure was significantly higher than the area obtained by the traditional biopsy procedure. This procedure could facilitate the assessment of the focus score [14].
Promising innovations are ultrasound elastography (with algorithms to evaluate quantitatively the tissue stiffness of the salivary gland tissue [15], the application of artificial intelligence to automatically score for biopsy purposes [16], and the ultrasound-guided core needle biopsy of major salivary glands which could represent an alternative to surgical biopsy [17].
MRI does not belong to the common standard tools used for the diagnostic approach of pSS. Because of its high spatial resolution the major impact in such patients is for local staging of pSS associated with salivary glands lymphomas [18].
Salivary gland scintigraphy is a nuclear medicine technique that through the administration intravenously of 99mTc-04 allows for to evaluate of the function of major salivary glands (perfusion, concentration, and elimination characteristics) [19]. Sialoscintigraphy is no longer part of the recent classification criteria for pSS [20]; the reported diagnostic approach shows a sensitivity of up to 90% with specificity of around 50%, making this tool not able to distinguish the functional compromise of SS and other salivary glands pathologies. It is possible that this technique could have some potential indication in the future as part of the tools available for the follow-up of patients [19].
Molecular nuclear medicine imaging has emerged with several biomarkers with potential clinical impact for its contribution to diagnosing, assessing the inflammatory status, and assessing disease progression. These images allow in vivo the characterization of cells and the phenomena involved in inflammatory diseases. For this purpose, by using radiolabeled molecules (administered in nanomolar amounts), that participate in the biochemical and pathophysiological process of chronic inflammation, it is possible to make a qualitative and quantitative assessment of the inflammatory burden in vivo. Recent advances in the development of target-specific imaging agents, allow researchers to perform a non-invasive evaluation of various molecular events such as angiogenesis, apoptosis, and cell trafficking in living organisms. Cellular and molecular changes occur a long time before structural changes, therefore, non-invasive visualization and quantification of molecular processes facilitate the early detection of the disease, establish a prognosis, and could potentially estimate the potential impact of biologic therapies.
- a.
-
Somatostatin Receptor Imaging
Radiolabelled peptides are highly specific and are used to reveal the presence of target molecules in inflammatory disease through molecular imaging. The peptides used in nuclear medicine images are easily synthesized, stabilized, and modified with good pharmacokinetic parameters; they also show high receptor binding affinity and are internalized into cells [21]. One of the most commonly used radiolabelled peptides for the inflammatory disease clinical approach is somatostatin, which has been used for inflammatory diseases for more than two decades, particularly in rheumatoid arthritis, SS, and autoimmune thyroid disease [22][23]. This is supported by the well-known presence of active and over-expressed somatostatin receptors in inflammatory and immunological cells from different tissues [24]. The diagnostic accuracy is also high because of the strong binding affinity of somatostatin to its five receptors. Different radiopharmaceuticals for somatostatin receptor scintigraphy are available, such as 68Ga-DOTA-TATE, 68Ga-DOTA-TOC, 68Ga-DOTA-NOC, and 99mTc-EDDA/HYNIC-TOC [25], offering the possibility of detecting active inflammation in tissues affected by SS. Some of the evidence that supports the use of these radiopharmaceuticals in SS has been provided by Anzola et al., who reported the normal biodistribution of 68-Ga-DOTA-NOC and reference values for salivary glands, thyroid glands, and major joints, constituting the starting point for PET studies for further analysis in patients affected by rheumatic inflammatory disorders [26]. Using a cohort of 62 patients with confirmed SS by AECG criteria, the same group [27] reported the ability of 99mTc-HYNIC-TOC scintigraphy to identify active inflammatory processes not only in salivary glands but also in many joints. This work highlighted the capability of the molecule to evaluate inflammatory compromise in sites different from the salivary glands, to evaluate the inflammation status of the salivary glands, and to hypothesize the usefulness of the probe to assess response to treatment. In a pilot study in 18 patients with rheumatoid arthritis and secondary SS who received infliximab as treatment, the authors [28] showed uptake of 99mTc HYNIC-TOC in all affected joints and in the salivary glands in 12 of 18 patients. They also demonstrated a significant reduction in uptake by joints but not salivary glands after treatment with infliximab, enhancing the potential of the molecule to assess disease activity in rheumatoid arthritis and to detect secondary SS (Figure 1 and Figure 2). It is important to highlight that because the synovia of patients with SS and rheumatoid arthritis highly express somatostatin receptors [29], radiolabelled somatostatin can identify sites of active inflammation in joints accompanying salivary gland compromise in primary or secondary SS.
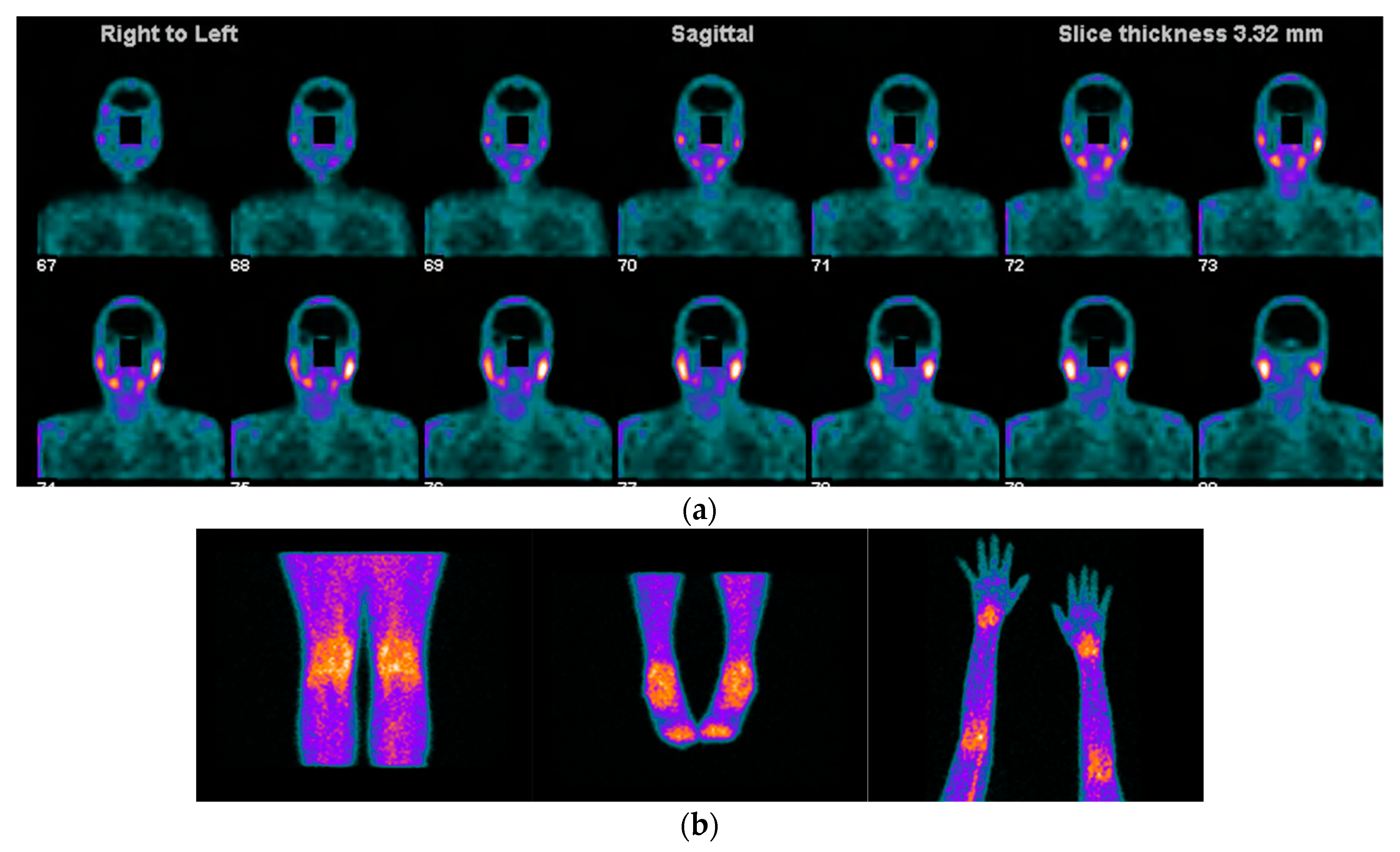
Figure 1. Images acquired after 99mTc-HYNIC-TOC administration i.v in a patient with SS, sicca symptoms, and painful joints. In (a) high abnormal uptake in parotids and submandibular salivary glands. In (b) shows high abnormal uptake in knees, ankles, elbows, and carpal joints.
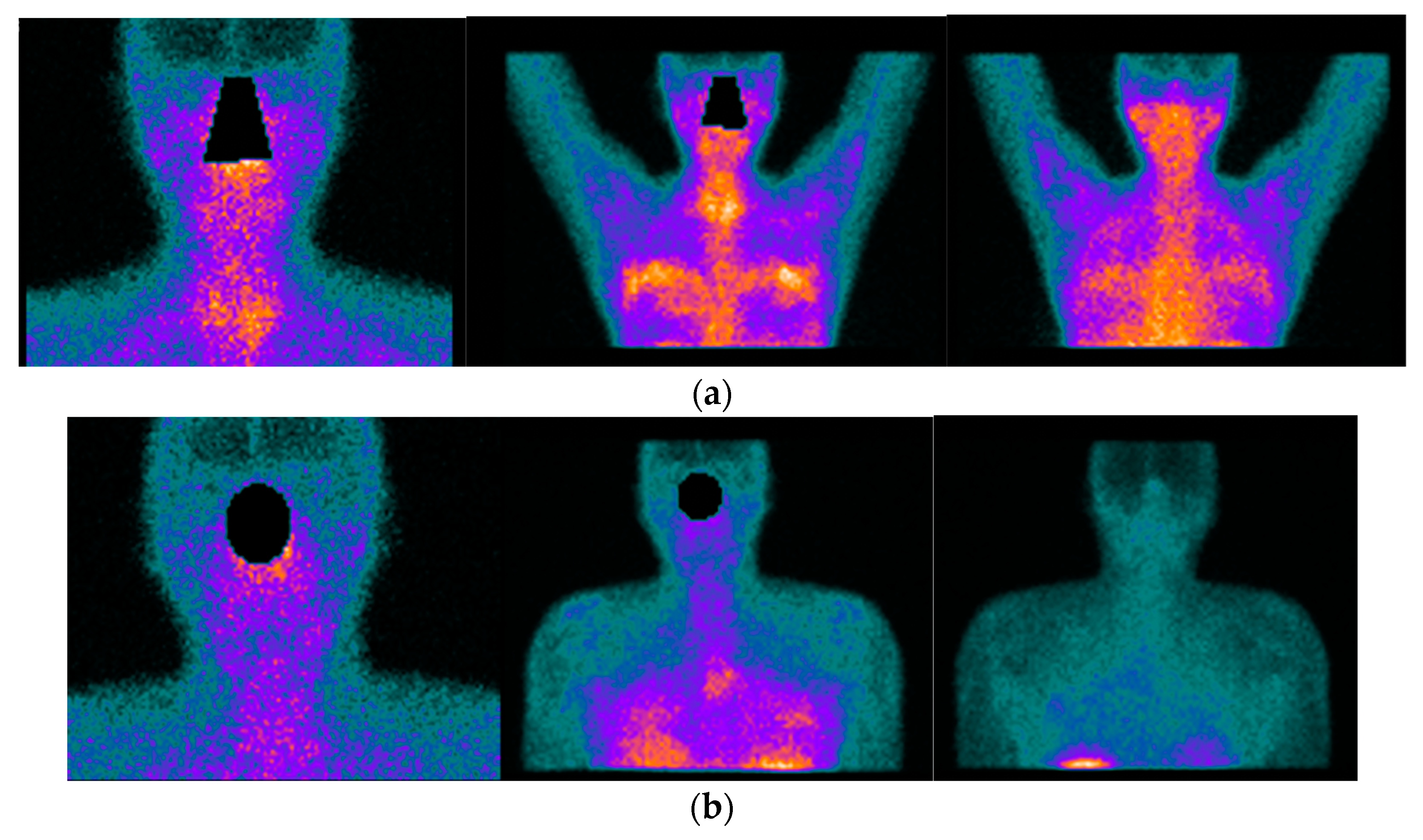
Figure 2. 42-year-old female patient with the previous history of bilateral mammoplasty; four years later she complained of dryness (VAS 5/10), fatigue (VAS 10/10), pain (VAS:8/10) ESSPRI: 23, and thyroiditis. She was diagnosed with SS under ACR-EULAR criteria and she started treatment with DMARDS for two years without relieving symptoms. She asked her surgeon to remove the implants and six months after the removal of the implants there was an improvement in her clinical condition, dryness (VAS: 2/10), fatigue (VAS:6/10), pain (VAS: 5/10) post-surgery ESSPRI: 13. In (a) 99mTc HYNIC-TOC images show high abnormal uptake of the radiotracer in the thyroid gland, submandibular glands, with a high abnormal bilateral mammary periprosthetic distribution of the radiotracer. SS was diagnosed under ACR-EULAR criteria. In (b) 99mTc HYNIC-TOC images 6 months post removal of prosthesis show a significant decrease in uptake of the radiotracer in submandibular glands and thyroid gland. There was a concern that silicone could be associated with SS.
One systematic review [22] reported promising results of radiolabelled somatostatin analogs as diagnostic tools for localizing and identifying sites of active inflammation in joints as well as extra-articular involvement, such as the salivary glands, in patients with chronic inflammatory diseases (Figure 1a,b and Figure 2a,b).
- b.
-
B-lymphocyte Imaging in SS
Currently, the importance of the role of B lymphocytes in the pathogenesis of rheumatic inflammatory diseases through the production of auto-antibodies, T-cell activation, and pro-inflammatory cytokines is well recognized [30]. Researchers previously reviewed how these cells are found in pathological infiltrates of affected tissues and are implicated in disease progression, with B-cell hyperactivity being fundamental to the disease. The maturation of B cells from stem cells suggests several steps, with changes in cell surface markers. Indeed, these surface markers have gained attention for use in B-cell-depleting therapies through the use of different monoclonal antibodies against them, acting directly (CD19, CD20, CD22) or indirectly via blockade of cytokine pathways (TNF-alpha, interleukin-6, B lymphocyte stimulator (BLyS) and proliferation-inducing ligand APRIL) [31][32]. Although some molecules are commonly used, scarce evidence is available, and the results of trials are inconclusive. No biologic drug has yet been approved for the treatment of pSS, though three biologics, i.e., rituximab, belimumab, and abatacept, have shown effectiveness in open studies for extra-glandular symptoms but not for dryness [33]. Anti-tumor necrosis factor-alpha (TNF-alpha) drug trials in pSS have failed to show promising results; although a potential protective role for TNF-alfa against lymphoproliferation has been hypothesized, it has been demonstrated that depletion of TNF-alpha may increase BAFF (B-cell activating factor) levels in humans, discouraging its use for pSS in clinical practice [34]. Rituximab is a chimeric monoclonal antibody directed against CD20, which is expressed on membrane B cells. Expert opinion [35][36][37] supports its use as a therapeutic option in pSS with systemic compromise and as second-line therapy for hematological manifestations. In addition, a radiolabelled anti-CD20 mAb probe for in vivo imaging of CD20-positive lymphocyte infiltration in inflammatory diseases has been described, offering the possibility of a better approach to disease staging [38].
In vivo imaging of lymphocyte B through the use of anti-CD20 radiotracer has potential applicability for SS patients. Malviya et al. [39] used radiolabelled anti-CD20 (rituximab) and showed B Lymphocyte infiltration in affected tissues of patients with different chronic inflammatory autoimmune diseases and who were candidates for rituximab treatment. The authors reported the in vivo localization pattern of the B-lymphocytes mediating the inflammatory process, with variable uptake by salivary glands and lacrimal glands in two SS patients. Despite the small sample size, this work provides the basis for further studies assessing whether an antibody accumulates in inflamed tissue before using the same antibody for therapeutic purposes, thereby improving the selection of patients who might benefit from the therapy, which is called immune scintigraphy for therapeutic decision-making.
- c.
-
T-lymphocyte Imaging in SS
Because activated T lymphocytes are involved in chronic inflammatory diseases and IL2 is mainly secreted by these cells to play a regulatory role during inflammation, some authors have developed a means of detecting these cells in vivo through the synthesis of different radiopharmaceuticals for SPECT and PET systems [40]. Reports to date have demonstrated its usefulness in different clinical inflammatory scenarios, such as in insulitis in type 1 diabetes [41], Crohn’s disease [42], and Hashimoto thyroiditis, among others, for diagnostic and prognostic purposes and for therapy follow-up.
In a recently published study [43], in a cohort of 48pSS, the authors reported the utility of 99mTc-IL2 for evaluating in vivo the extent and severity of lympho-mononuclear cell infiltration in the salivary glands. When they compared the results with a control group they found a statistically significant difference (p < 0.0001) in the radiotracer uptake in salivary glands between the two groups; they also observed that the uptake in patients with a longer history of the disease was lower compared with the recently diagnosed patients. They highlighted the capacity of the molecule to detect active inflammation mediated by IL2 and the possibility to treat those patients with immune-modulatory drugs and using the probe for evaluating the efficacy of the treatment (Figure 3).
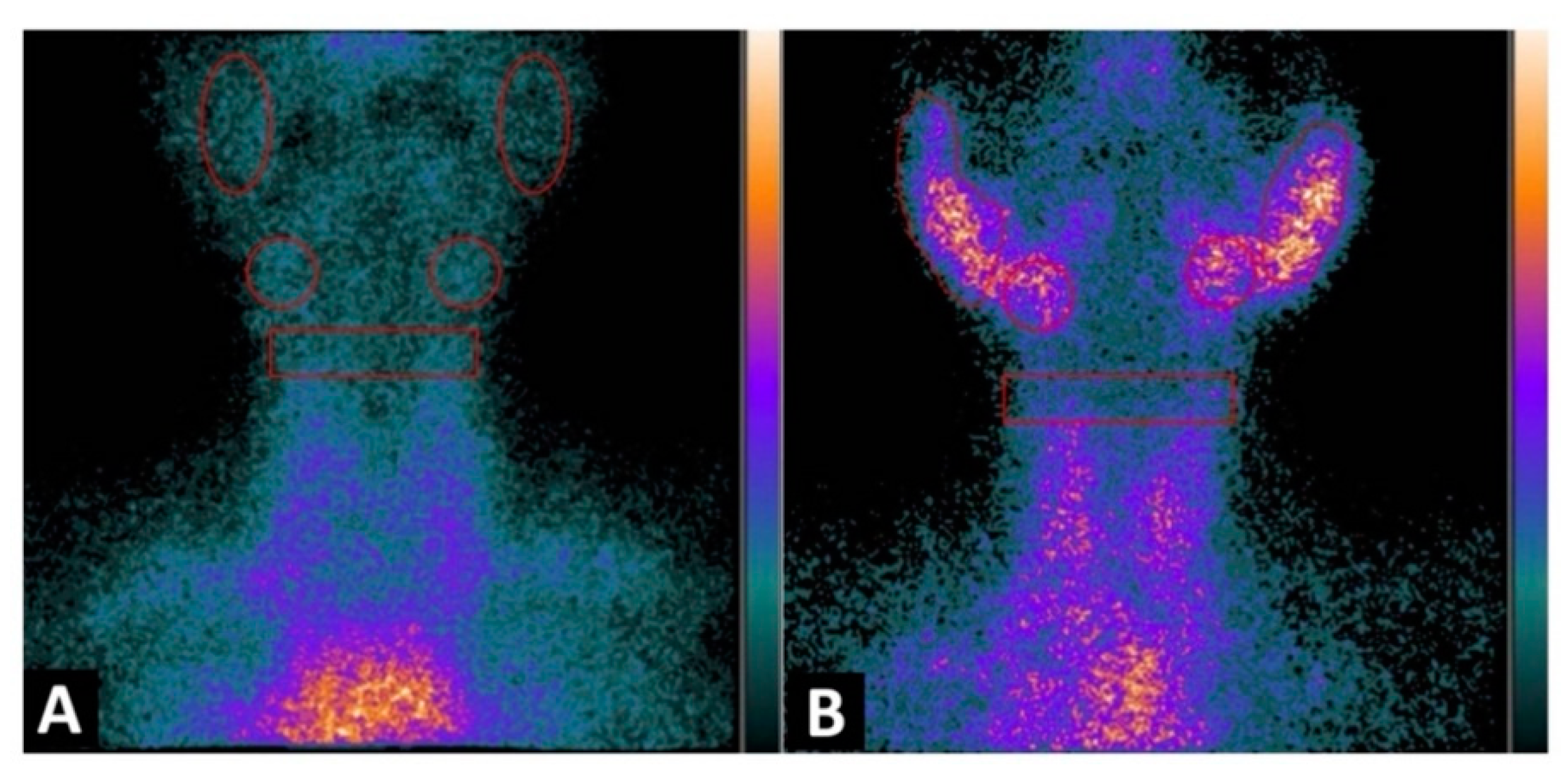
Figure 3. Planar image of the neck was obtained 1h after 99mTc-IL2 injection in a control subject (A) and in a patient with Sjögren syndrome at the time of diagnosis (B). In (A) the scan shows no 99mTc-IL2 uptake by the salivary glands. In (B) an evident accumulation of 99mTc-IL2 can be observed in both parotids and submandibular glands, indicating the presence of activated lymphocytes. The calculated parotid to background (P/B) ratios are 1.35 and 1.30 in right and left glands, respectively, and the submandibular gland to background (S/B) ratios are 1.57 and 1.64 in right and left glands, respectively. Reprinted with permission from Imaging Activated-T-Lymphocytes in the Salivary Glands of Patients with Sjögren’s Syndrome by 99mTc-Interleukin-2: Diagnostic and Therapeutic Implications Campagna, G.; Anzola, L. K.; Varani, M.; Lauri, C.; Gentiloni Silveri, G.; Chiurchioni, L.; et al. J. Clin. Med. 2022, 11 (15), 4368. [43].
New radiotracers, such as 18F-fluorbenzoyl interleukin-2, have also been developed for PET systems, allowing detection of activated T lymphocytes for the same purposes. In a preclinical study, Di Gialleonardo et al. [44] identified activated T lymphocytes in inflamed tissues, highlighting the potential of the probe for detecting activated T lymphocytes in autoimmune diseases such as SS. Because T lymphocytes are also mainly implicated in the pathogenesis of SS, molecular images for detecting T lymphocyte activity would play an important role in diagnostic approaches [9].
- d.
-
Other PET Radiopharmaceuticals Used in SS
Recently, in an age- and sex-matched study with SS patients and healthy volunteers using MRI, 11C-MET-PET, and 18F-FDG-PET, Jimenez-Royo et al. [45] reported how molecular imaging findings correlate with disease characteristics, providing information about the inflammatory and functional status of the salivary glands. 11C-MET PET (a protein synthesis marker incorporated into cellular proteins) information [46] was used to evaluate residual salivary gland function, and 18F-FDG PET information was used to evaluate the inflammatory condition through glucose utilization by the inflamed cell [47]. Their main findings were significantly lower 11C-MET uptake in the parotid and submandibular glands in SS patients than in volunteers and higher uptake of 18F-FDG in the salivary glands of SS patients, indicating the presence of inflammation. Furthermore, a negative correlation between 11C-MET and 18F-FDG uptake (loss of function with highly inflamed tissues) was reported. Regarding histological analysis, the most relevant finding was related to a moderate positive correlation between 18F-FDG uptake in the parotid gland and CD20+ B-cell infiltration in the minor salivary gland of patients, suggesting that 18F-FDG uptake shows B-cell tissue infiltration By using 18F-FDG PET in SS patients, Cohen et al. [48] found evidence of the added value of the tool, demonstrating systemic compromise in SS. These authors observed that although the most frequent site of uptake was the salivary glands, the lymph nodes and lungs were equally affected. They also described a PETC/CT inflammation score that correlated with ESSDAI and gammaglobulin levels, suggesting that 18F-FDG PET may help in assessing disease activity and represent an inflammation biomarker for SS.
Although interesting characteristics of 18F-FDG PET have been described as inflammation markers in SS, it is important to take into consideration that new probes would make possible more specific detection of inflammation by targeting specific molecules and cells expressed in compromised tissues, providing good characteristics not only for diagnostic purposes but also for therapy decision-making and follow-up [49].
This entry is adapted from the peer-reviewed paper 10.3390/jcm11175227
References
- Anaya, J.-M.; Restrepo-Jiménez, P.; Rodríguez, Y.; Rodríguez-Jiménez, M.; Acosta-Ampudia, Y.; Monsalve, D.M.; Pacheco, Y.; Ramírez-Santana, C.; Molano-González, N.; Mantilla, R.D. Sjögren’s Syndrome and Autoimmune Thyroid Disease: Two Sides of the Same Coin. Clin. Rev. Allergy Immunol. 2019, 56, 362–374.
- Mariette, X.; Criswell, L.A. Primary Sjögren’s Syndrome. N. Engl. J. Med. 2018, 378, 931–939.
- Brito-Zerón, P.; Kostov, B.; Solans, R.; Fraile, G.; Suárez-Cuervo, C.; Casanovas, A.; Rascón, F.J.; Qanneta, R.; Pérez-Alvarez, R.; Ripoll, M.; et al. Systemic Activity and Mortality in Primary Sjögren Syndrome: Predicting Survival Using the EULAR-SS Disease Activity Index (ESSDAI) in 1045 Patients. Ann. Rheum. Dis. 2016, 75, 348–355.
- Kassan, S.S. Increased Risk of Lymphoma in Sicca Syndrome. Ann. Intern. Med. 1978, 89, 888.
- Ioannidis, J.P.A.; Vassiliou, V.A.; Moutsopoulos, H.M. Long-Term Risk of Mortality and Lymphoproliferative Disease and Predictive Classification of Primary Sjögren’s Syndrome: Classification of Sjögren’s Syndrome. Arthritis Rheum. 2002, 46, 741–747.
- Kapsogeorgou, E.K.; Voulgarelis, M.; Tzioufas, A.G. Predictive Markers of Lymphomagenesis in Sjögren’s Syndrome: From Clinical Data to Molecular Stratification. J. Autoimmun. 2019, 104, 102316.
- Goules, A.V.; Tzioufas, A.G. Lymphomagenesis in Sjögren’s Syndrome: Predictive Biomarkers towards Precision Medicine. Autoimmun. Rev. 2019, 18, 137–143.
- Zandonella Callegher, S.; Giovannini, I.; Zenz, S.; Manfrè, V.; Stradner, M.H.; Hocevar, A.; Gutierrez, M.; Quartuccio, L.; De Vita, S.; Zabotti, A. Sjögren Syndrome: Looking Forward to the Future. Ther. Adv. Musculoskelet. Dis. 2022, 14, 1759720X221100295.
- Van Ginkel, M.S.; Glaudemans, A.W.J.M.; van der Vegt, B.; Mossel, E.; Kroese, F.G.M.; Bootsma, H.; Vissink, A. Imaging in Primary Sjögren’s Syndrome. J. Clin. Med. 2020, 9, 2492.
- Jousse-Joulin, S.; Gatineau, F.; Baldini, C.; Baer, A.; Barone, F.; Bootsma, H.; Bowman, S.; Brito-Zerón, P.; Cornec, D.; Dorner, T.; et al. Weight of Salivary Gland Ultrasonography Compared to Other Items of the 2016 ACR/EULAR Classification Criteria for Primary Sjögren’s Syndrome. J. Intern. Med. 2020, 287, 180–188.
- Kroese, F.G.M.; Haacke, E.A.; Bombardieri, M. The Role of Salivary Gland Histopathology in Primary Sjögren’s Syndrome: Promises and Pitfalls. Clin. Exp. Rheumatol. 2018, 36 (Suppl. 112), 222–233.
- Zabotti, A.; Callegher, S.Z.; Tullio, A.; Vukicevic, A.; Hocevar, A.; Milic, V.; Cafaro, G.; Carotti, M.; Delli, K.; De Lucia, O.; et al. Salivary Gland Ultrasonography in Sjögren’s Syndrome: A European Multicenter Reliability Exercise for the HarmonicSS Project. Front. Med. 2020, 7, 581248.
- Manfrè, V.; Cafaro, G.; Riccucci, I.; Zabotti, A.; Perricone, C.; Bootsma, H.; De Vita, S.; Bartoloni, E. One Year in Review 2020: Comorbidities, Diagnosis and Treatment of Primary Sjögren’s Syndrome. Clin. Exp. Rheumatol. 2020, 38 (Suppl. 126), 10–22.
- Baldini, C.; Ferro, F.; Izzetti, R.; Vitali, S.; Fonzetti, S.; Governato, G.; Aringhieri, G.; Elefante, E.; Mosca, M.; Donati, V.; et al. Fri0153 Ultra High-Resolution Ultrasound (Uhfus) of Labial Salivary Glands: Potential Applications in Primary Sjögren’s Syndrome. Ann. Rheum. Dis. 2020, 79 (Suppl. 1), 660–661.
- Cindil, E.; Oktar, S.O.; Akkan, K.; Sendur, H.N.; Mercan, R.; Tufan, A.; Ozturk, M.A. Ultrasound Elastography in Assessment of Salivary Glands Involvement in Primary Sjögren’s Syndrome. Clin. Imaging 2018, 50, 229–234.
- Radovic, M.; Vukicevic, A.; Zabotti, A.; Milic, V.; De Vita, S.; Filipovic. Deep Learning Based Approach for Assessment of Primary Sjögren’s Syndrome from Salivary Gland Ultrasonography Images. Available online: https://www.springerprofessional.de/en/deep-learning-based-approach-for-assessment-of-primary-sjoegren-/17790232 (accessed on 23 August 2022).
- Lorenzon, M.; Di Franco, F.T.; Zabotti, A.; Pegolo, E.; Giovannini, I.; Manfrè, V.; Mansutti, E.; De Vita, S.; Zuiani, C.; Girometti, R. Sonographic Features of Lymphoma of the Major Salivary Glands Diagnosed with Ultrasound-Guided Core Needle Biopsy in Sjögren’s Syndrome. Clin. Exp. Rheumatol. 2021, 39 (Suppl. 133), 175–183.
- Grevers, G.; Ihrler, S.; Vogl, T.J.; Weiss, M. A Comparison of Clinical, Pathological and Radiological Findings with Magnetic Resonance Imaging Studies of Lymphomas in Patients with Sjögren’s Syndrome. Eur. Arch. Otorhinolaryngol. 1994, 251, 214–217.
- Baldini, C.; Zabotti, A.; Filipovic, N.; Vukicevic, A.; Luciano, N.; Ferro, F.; Lorenzon, M.; De Vita, S. Imaging in Primary Sjögren’s Syndrome: The “Obsolete and the New”. Clin. Exp. Rheumatol. 2018, 36 (Suppl. 112), 215–221.
- Shiboski, C.H.; Shiboski, S.C.; Seror, R.; Criswell, L.A.; Labetoulle, M.; Lietman, T.M.; Rasmussen, A.; Scofield, H.; Vitali, C.; Bowman, S.J.; et al. 2016 ACR-EULAR Classification Criteria for Primary Sjögren’s Syndrome: A Consensus and Data-Driven Methodology Involving Three International Patient Cohorts. Arthritis Rheumatol 2017, 69, 35–45.
- Sosabowsky, J.; Melendez-Alafort, L.; Mather, S. Radiolabelling of Peptides for Diagnosis and Therapy of Non-Oncological Diseases. Q. J. Nucl. Med. 2003, 47, 223–237.
- Anzola, L.K.; Glaudemans, A.W.J.M.; Dierckx, R.A.J.O.; Martinez, F.A.; Moreno, S.; Signore, A. Somatostatin Receptor Imaging by SPECT and PET in Patients with Chronic Inflammatory Disorders: A Systematic Review. Eur. J. Nucl. Med. Mol. Imaging 2019, 46, 2496–2513.
- Signore, A.; Anzola, K.L.; Auletta, S.; Varani, M.; Petitti, A.; Pacilio, M.; Galli, F.; Lauri, C. Current Status of Molecular Imaging in Inflammatory and Autoimmune Disorders. Curr. Pharm. Des. 2018, 24, 743–753.
- Duet, M.; Lioté, F. Somatostatin and Somatostatin Analog Scintigraphy: Any Benefits for Rheumatology Patients? Joint Bone Spine 2004, 71, 530–535.
- Cascini, G.L.; Cuccurullo, V.; Tamburrini, O.; Rotondo, A.; Mansi, L. Peptide Imaging with Somatostatin Analogues: More than Cancer Probes. Curr. Radiopharm. 2013, 6, 36–40.
- Anzola, L.; Lauri, C.; Granados, C.; Lagana, B.; Signore, A. Uptake Pattern of Ga-DOTA-NOC in Tissues: Implications for Inflammatory Diseases. Q. J. Nucl. Med. Mol. Imaging 2019, 66, 156–161.
- Anzola, L.K.; Rivera, J.N.; Dierckx, R.A.; Lauri, C.; Valabrega, S.; Galli, F.; Lopez, S.M.; Glaudemans, A.W.J.M.; Signore, A. Value of Somatostatin Receptor Scintigraphy with 99mTc-HYNIC-TOC in Patients with Primary Sjögren Syndrome. J. Clin. Med. 2019, 8, 763.
- Anzola-Fuentes, L.K.; Chianelli, M.; Galli, F.; Glaudemans, A.W.J.M.; Martin, L.M.; Todino, V.; Migliore, A.; Signore, A. Somatostatin Receptor Scintigraphy in Patients with Rheumatoid Arthritis and Secondary Sjögren’s Syndrome Treated with Infliximab: A Pilot Study. EJNMMI Res. 2016, 6, 49.
- Reubi, J.C.; Waser, B.; Markusse, H.M.; Krenning, E.P.; VanHagen, M.; Laissue, J.A. Vascular Somatostatin Receptors in Synovium from Patients with Rheumatoid Arthritis. Eur. J. Pharmacol. 1994, 271, 371–378.
- Tedder, T.F.; Boyd, A.W.; Freedman, A.S.; Nadler, L.M.; Schlossman, S.F. The B Cell Surface Molecule B1 Is Functionally Linked with B Cell Activation and Differentiation. J. Immunol. Baltim. Md 1985, 135, 973–979.
- Malviya, G.; Galli, F.; Sonni, I.; Pacilio, M.; Signore, A. Targeting T and B Lymphocytes with Radiolabelled Antibodies for Diagnostic and Therapeutic Applications. Q. J. Nucl. Med. Mol. Imaging 2010, 54, 654–676.
- Dörner, T.; Kinnman, N.; Tak, P.P. Targeting B Cells in Immune-Mediated Inflammatory Disease: A Comprehensive Review of Mechanisms of Action and Identification of Biomarkers. Pharmacol. Ther. 2010, 125, 464–475.
- Gandolfo, S.; De Vita, S. Emerging Drugs for Primary Sjögren’s Syndrome. Expert Opin. Emerg. Drugs 2019, 24, 121–132.
- Mavragani, C.P.; Niewold, T.B.; Moutsopoulos, N.M.; Pillemer, S.R.; Wahl, S.M.; Crow, M.K. Augmented Interferon-Alpha Pathway Activation in Patients with Sjögren’s Syndrome Treated with Etanercept. Arthritis Rheum. 2007, 56, 3995–4004.
- Giacomelli, R.; Afeltra, A.; Alunno, A.; Baldini, C.; Bartoloni-Bocci, E.; Berardicurti, O.; Carubbi, F.; Cauli, A.; Cervera, R.; Ciccia, F.; et al. International Consensus: What Else Can We Do to Improve Diagnosis and Therapeutic Strategies in Patients Affected by Autoimmune Rheumatic Diseases (Rheumatoid Arthritis, Spondyloarthritides, Systemic Sclerosis, Systemic Lupus Erythematosus, Antiphospholipid Syndrome and Sjogren’s Syndrome)?: The Unmet Needs and the Clinical Grey Zone in Autoimmune Disease Management. Autoimmun. Rev. 2017, 16, 911–924.
- Gottenberg, J.-E.; Cinquetti, G.; Larroche, C.; Combe, B.; Hachulla, E.; Meyer, O.; Pertuiset, E.; Kaplanski, G.; Chiche, L.; Berthelot, J.-M.; et al. Efficacy of Rituximab in Systemic Manifestations of Primary Sjogren’s Syndrome: Results in 78 Patients of the AutoImmune and Rituximab Registry. Ann. Rheum. Dis. 2013, 72, 1026–1031.
- Carubbi, F.; Cipriani, P.; Marrelli, A.; Benedetto, P.D.; Ruscitti, P.; Berardicurti, O.; Pantano, I.; Liakouli, V.; Alvaro, S.; Alunno, A.; et al. Efficacy and Safety of Rituximab Treatment in Early Primary Sjögren’s Syndrome: A Prospective, Multi-Center, Follow-up Study. Arthritis Res. Ther. 2013, 15, R172.
- Malviya, G.; Conti, F.; Chianelli, M.; Scopinaro, F.; Dierckx, R.A.; Signore, A. Molecular Imaging of Rheumatoid Arthritis by Radiolabelled Monoclonal Antibodies: New Imaging Strategies to Guide Molecular Therapies. Eur. J. Nucl. Med. Mol. Imaging 2010, 37, 386–398.
- Malviya, G.; Anzola, K.L.; Podestà, E.; Laganà, B.; Del Mastro, C.; Dierckx, R.A. (99m)Tc-Labeled Rituximab for Imaging B Lymphocyte Infiltration in Inflammatory Autoimmune Disease Patients. Mol. Imaging Biol. 2012, 14, 637–646.
- Signore, A.; Picarelli, A.; Annovazzi, A.; Britton, K.E.; Grossman, A.B.; Bonanno, E.; Maras, B.; Barra, D.; Pozzilli, P. 123I-Interleukin-2: Biochemical Characterization and in Vivo Use for Imaging Autoimmune Diseases. Nucl. Med. Commun. 2003, 24, 305–316.
- Signore, A.; Parman, A.; Pozzilli, P.; Andreani, D.; Beverley, P.C. Detection of Activated Lymphocytes in Endocrine Pancreas of BB/W Rats by Injection of 123I-Interleukin-2: An Early Sign of Type 1 Diabetes. Lancet Lond. Engl. 1987, 2, 537–540.
- Signore, A.; Chianelli, M.; Alessio, A.; Bonanno, E.; Spagnoli, L.G.; Pozzilli, P.; Pallone, F.; Biancone, L. 123I-Interleukin-2 Scintigraphy for in Vivo Assessment of Intestinal Mononuclear Cell Infiltration in Crohn’s Disease. J. Nucl. Med. Off. Publ. Soc. Nucl. Med. 2000, 41, 242–249.
- Campagna, G.; Anzola, L.K.; Varani, M.; Lauri, C.; Silveri, G.G.; Chiurchioni, L.; Spinelli, F.R.; Priori, R.; Conti, F.; Signore, A. Imaging Activated-T-Lymphocytes in the Salivary Glands of Patients with Sjögren’s Syndrome by 99mTc-Interleukin-2: Diagnostic and Therapeutic Implications. J. Clin. Med. 2022, 11, 4368.
- Di Gialleonardo, V.; Signore, A.; Glaudemans, A.W.J.M.; Dierckx, R.A.J.O.; De Vries, E.F.J. N -(4- 18 F-Fluorobenzoyl)Interleukin-2 for PET of Human-Activated T Lymphocytes. J. Nucl. Med. 2012, 53, 679–686.
- Jimenez-Royo, P.; Bombardieri, M.; Ciurtin, C.; Kostapanos, M.; Tappuni, A.R.; Jordan, N.; Saleem, A.; Fuller, T.; Port, K.; Pontarini, E.; et al. Advanced Imaging for Quantification of Abnormalities in the Salivary Glands of Patients with Primary Sjögren’s Syndrome. Rheumatology 2021, 60, 2396–2408.
- Glaudemans, A.W.J.M.; Enting, R.H.; Heesters, M.A.A.M.; Dierckx, R.A.J.O.; Van Rheenen, R.W.J.; Walenkamp, A.M.E.; Slart, R.H.J.A. Value of 11C-Methionine PET in Imaging Brain Tumours and Metastases. Eur. J. Nucl. Med. Mol. Imaging 2013, 40, 615–635.
- Ziakas, P.D.; Poulou, L.S.; Thanos, L. Towards Integrating Positron Emission Tomography for Work-up of Patients with Sjögren’s Syndrome and Associated Lymphomas. Autoimmun. Rev. 2014, 13, 327–329.
- Cohen, C.; Mekinian, A.; Uzunhan, Y.; Fauchais, A.-L.; Dhote, R.; Pop, G.; Eder, V.; Nunes, H.; Brillet, P.-Y.; Valeyre, D.; et al. 18F-Fluorodeoxyglucose Positron Emission Tomography/Computer Tomography as an Objective Tool for Assessing Disease Activity in Sjögren’s Syndrome. Autoimmun. Rev. 2013, 12, 1109–1114.
- Signore, A.; Lauri, C.; Auletta, S.; Anzola, K.; Galli, F.; Casali, M.; Versari, A.; Glaudemans, A.W. mmuno-Imaging to Predict Treatment Response in Infection, Inflammation and Oncology. J. Clin. Med. 2019, 8, 681.
This entry is offline, you can click here to edit this entry!
