The role of NLRP3 in the tumour microenvironment is elusive. In some cancers, the activation of NLRP3 causes a worse prognosis and in some cancers, NLRP3 increases chances of survivability. However, in many cases where NLRP3 has a protumorigenic role, inhibition of NLRP3 would be a crucial step in therapy. Consequently, activation of NLRP3 would be of essence when inflammation is required. Chitin and its derivatives are able to upregulate and downregulate the effect of the NLRP3 inflammasome based on its preparation, and these different reactions can be utilised to successfully target a broad range of cancers. Out of chitin, chitosan and chitooligosaccharide (COS), COS seems to be the best approach to actual products due its solubility being the highest, enabling it to be delivered more efficiently when compared to the other two. These specific preparations can be used on a case-by-case basis to help mitigate the negative effects of cancer and can potentially be used as a treatment or an adjuvant to cancer treatment.
- NLRP3
- chitin derivatives
- chitosan
- chito-oligosaccharides
1. Introduction
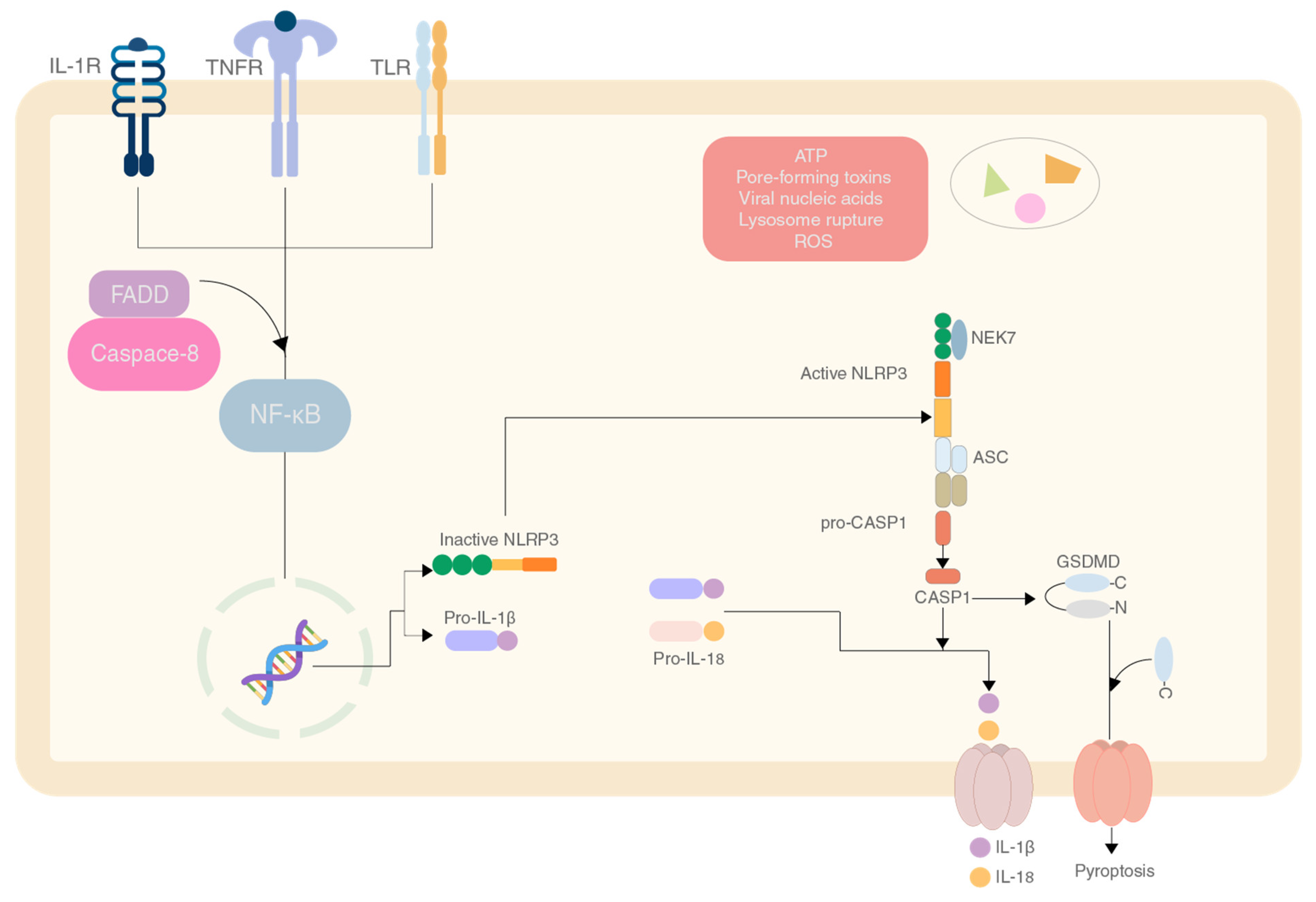
2. The Role of Chitin and Its Derivatives
2.1. Effect of Chitin
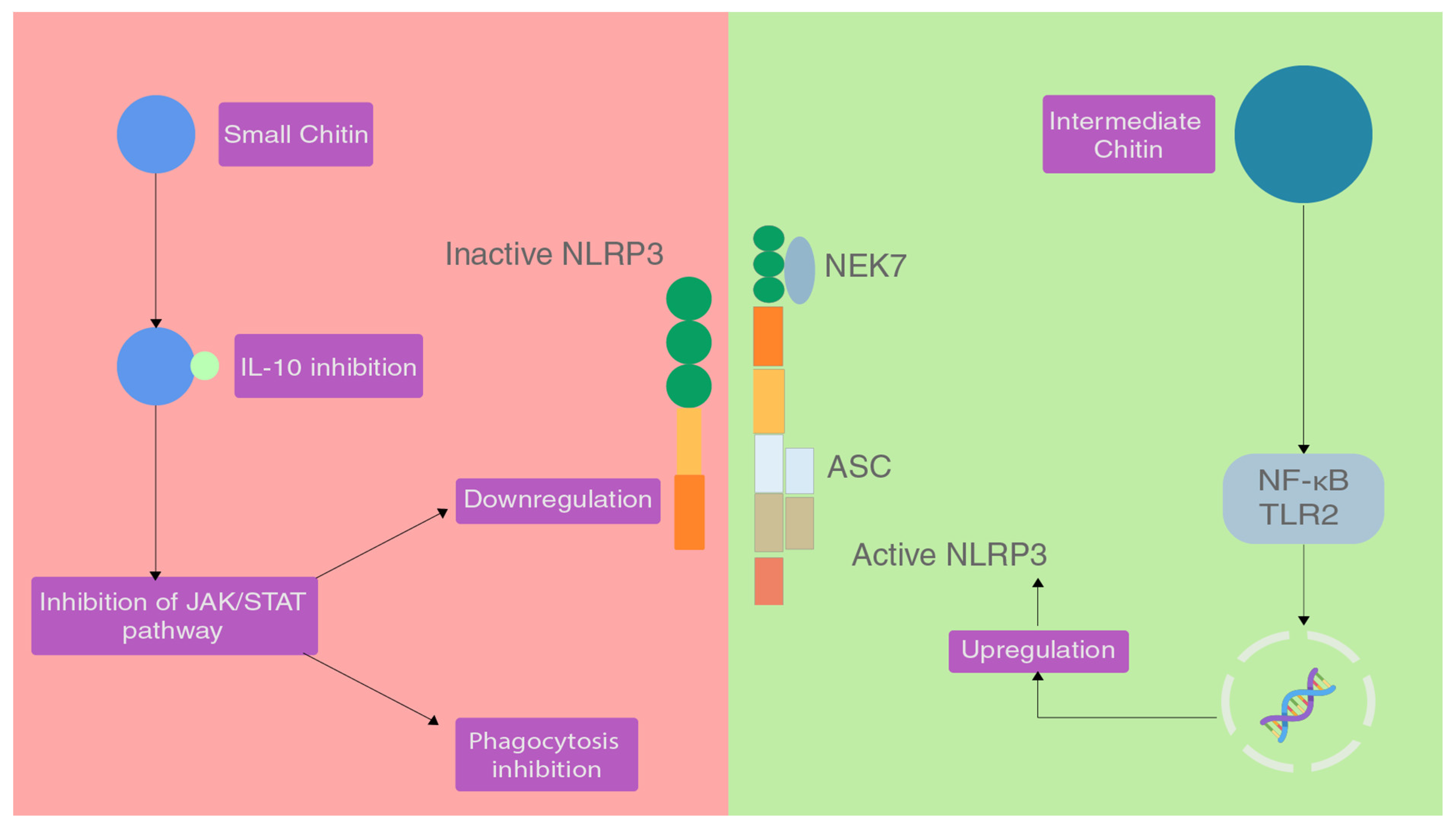
2.2. Effect of Chitosan

2.3. Effect of Chitooligosaccharides
This entry is adapted from the peer-reviewed paper 10.3390/md20070452
References
- Missiroli, S.; Missiroli, S.; Perrone, M.; Boncompagni, C.; Borghi, C.; Campagnaro, A.; Marchetti, F.; Anania, G.; Greco, P.; Fiorica, F.; et al. Targeting the nlrp3 inflammasome as a new therapeutic option for overcoming cancer. Cancers 2021, 13, 2297.
- He, Y.; Hara, H.; Núñez, G. Mechanism and regulation of nlrpinflammasome activation. Trends Biochem. Sci. 2016, 41, 1012–1021.
- Ashrafizadeh, M.; Zarrabi, A.; Hashemi, F.; Moghadam, E.R.; Hashemi, F.; Entezari, M.; Hushmandi, K.; Mohammadinejad, R.; Najafi, M. Curcumin in cancer therapy: A novel adjunct for combination chemotherapy with paclitaxel and alleviation of its adverse effects. Life Sci. 2020, 256, 117984.
- Azuma, K.; Osaki, T.; Minami, S.; Okamoto, Y. Anticancer and anti-Inflammatory properties of chitin and chitosan oligosaccharides. J. Funct. Biomater. 2015, 6, 33–49.
- Ahmad, S.I.; Ahmad, R.; Khan, M.S.; Kant, R.; Shahid, S.; Gautam, L.; Hasan, G.M.; Hassan, M.I. Chitin and its derivatives: Structural properties and biomedical applications. Int. J. Biol. Macromol. 2020, 164, 526–539.
- Bussink, A.P.; Speijer, D.; Aerts, J.M.F.G.; Boot, R.G. Evolution of mammalian chitinase(-like) members of family 18 glycosyl hydrolases. Genetics 2007, 177, 959–970.
- Bouhenna, M.; Salah, R.; Bakour, R.; Drouiche, N.; Abdi, N.; Grib, H.; Lounici, H.; Mameri, N. Effects of chitin and its derivatives on human cancer cells lines. Environ. Sci. Pollut. Res. 2015, 22, 15579–15586.
- Lee, C.G.; Da Silva, C.A.; Cruz, C.S.D.; Ahangari, F.; Ma, B.; Kang, M.J.; He, C.H.; Takyar, S.; Elias, J.A. Role of chitin and chitinase/chitinase-like proteins in inflammation, tissue remodeling, and injury. Annu. Rev. Physiol. 2011, 73, 479–501.
- da Silva, C.A.; Chalouni, C.; Williams, A.; Hartl, D.; Lee, C.G.; Elias, J.A. Chitin is a size-dependent regulator of macrophage TNF and IL-10 production. J. Immunol. 2009, 182, 3573–3582.
- Iyer, S.S.; Cheng, G. Role of interleukin 10 transcriptional regulation in inflammation and autoimmune disease. Crit. Rev. Immunol. 2012, 32, 23–63.
- Wills-Karp, M.; Nathan, A.; Page, K.; Karp, C.L. New insights into innate immune mechanisms underlying allergenicity. Mucosal Immunol. 2010, 3, 104–110.
- He, X.; Howard, B.A.; Liu, Y.; Neumann, A.K.; Li, L.; Menon, N.; Roach, T.; Kale, S.D.; Samuels, D.C.; Li, H.; et al. LYSMD3: A mammalian pattern recognition receptor for chitin. Cell Rep. 2021, 36, 109392.
- Schlosser, A.; Thomsen, T.; Moeller, J.B.; Nielsen, O.; Tornøe, I.; Mollenhauer, J.; Moestrup, S.K.; Holmskov, U. Characterization of FIBCD1 as an acetyl group-binding receptor that binds chitin. J. Immunol. 2009, 183, 3800–3809.
- Rozbeský, D.; Ivanova, L.; Hernychová, L.; Grobárová, V.; Novák, P.; Černý, J. Nkrp1 family, from lectins to protein interacting molecules. Molecules 2015, 20, 3463–3478.
- Cash, H.L.; Whitham, C.V.; Behrendt, C.L.; Hooper, L.V. Symbiotic bacteria direct expression of an intestinal bactericidal lectin. Science 2006, 313, 1126–1130.
- Seetharaman, J.; Kfanigsberg, A.; Slaaby, R.; Leffler, H.; Barondes, S.H.; Rini, J.M. X-ray crystal structure of the human galectin-3 carbohydrate recognition domain at 2.1-Å resolution. J. Biol. Chem. 1998, 273, 13047–13052.
- Ioelovich, M. Crystallinity and hydrophility of chitin and chitosan. J. Chem. 2014, 3, 7–14.
- Elieh-Ali-Komi, D.; Hamblin, M.R.; Daniel, E.-A.-K. Chitin and chitosan: Production and application of versatile biomedical nanomaterials. Int. J. Adv. Res. 2016, 4, 411.
- Younes, I.; Rinaudo, M. Chitin and chitosan preparation from marine sources. Structure, properties and applications. Mar. Drugs 2015, 13, 1133–1174.
- Kou, S.; Peters, L.M.; Mucalo, M.R. Chitosan: A review of sources and preparation methods. Int. J. Biol. Macromol. 2021, 169, 85–94.
- Ravindranathan, S.; Koppolu, B.P.; Smith, S.G.; Zaharoff, D.A. Effect of chitosan properties on immunoreactivity. Mar. Drugs 2016, 14, 91.
- Chang, S.H.; Lin, Y.Y.; Wu, G.J.; Huang, C.H.; Tsai, G.J. Effect of chitosan molecular weight on anti-inflammatory activity in the RAW 264.7 macrophage model. Int. J. Biol. Macromol. 2019, 131, 167–175.
- Kapadnis, G.; Dey, A.; Dandekar, P.; Jain, R. Effect of degree of deacetylation on solubility of low-molecular-weight chitosan produced via enzymatic breakdown of chitosan. Polym. Int. 2019, 68, 1054–1063.
- Zou, P.; Yuan, S.; Yang, X.; Guo, Y.; Li, L.; Xu, C.; Zhai, X.; Wang, J. Structural characterization and antitumor effects of chitosan oligosaccharides against orthotopic liver tumor via NF-κB signaling pathway. J. Funct. Foods 2019, 57, 157–165.
- Shen, K.T.; Chen, M.H.; Chan, H.Y.; Jeng, J.H.; Wang, Y.J. Inhibitory effects of chitooligosaccharides on tumor growth and metastasis. Food Chem. Toxicol. 2009, 47, 1864–1871.
- Nam, K.-S.; Kim, M.-K.; Shon, Y.-H. Chemopreventive Effect of Chitosan Oligosaccharide against Colon Carcinogenesis. J. Microbiol. Biotechnol. 2007, 17, 1546–1549. Available online: http://europepmc.org/abstract/MED/18062235 (accessed on 4 March 2022).
