Brucellosis caused by Brucella spp. is transmitted by direct or indirect contact with infected animals or their secretions and through the consumption of infected animal meat and unpasteurized milk/milk products. Brucellosis is classified as one of the top neglected zoonosis by the World Health Organization (WHO), and despite this, it does not attract the appropriate attention it requires from both the Federal and State ministries of health in Nigeria. 15.8% (7178/45,363) seroprevalence of brucellosis was recorded in northern Nigeria as against 8.7% (1902/21,740) in the southern part. Brucella abortus, B. melitensis, B. suis, and B. canis were reported in 27 of the 36 states.
- human and animal brucellosis
- Brucella
- Nigeria
- prevalence and seroprevalence
- systemic review and meta-analysis
1. Introduction
2. Human and Animal Brucellosis in Nigeria

Limitations: Researchers' inability to determine the prevalence of the disease based on certain variables such as age, sex, cattle herds, breeds of animals, and mortality rate both in humans and animals are part of the limitations of this entry. The use of serological methods, as seen in this entry, is of national importance because of their usefulness in detecting antibodies against Brucella species. However, the results are only reliable when the diagnostic methods and procedures are properly conducted and the outcomes are well interpreted. Interestingly, serological methods have several shortcomings, including the antisera’s ability to cross-react with other bacterial pathogens such as Escherichia coli 0157, Salmonella Urbana group N, Francisella tularensis, and Yersinia enterocolitica 0:9 [130].
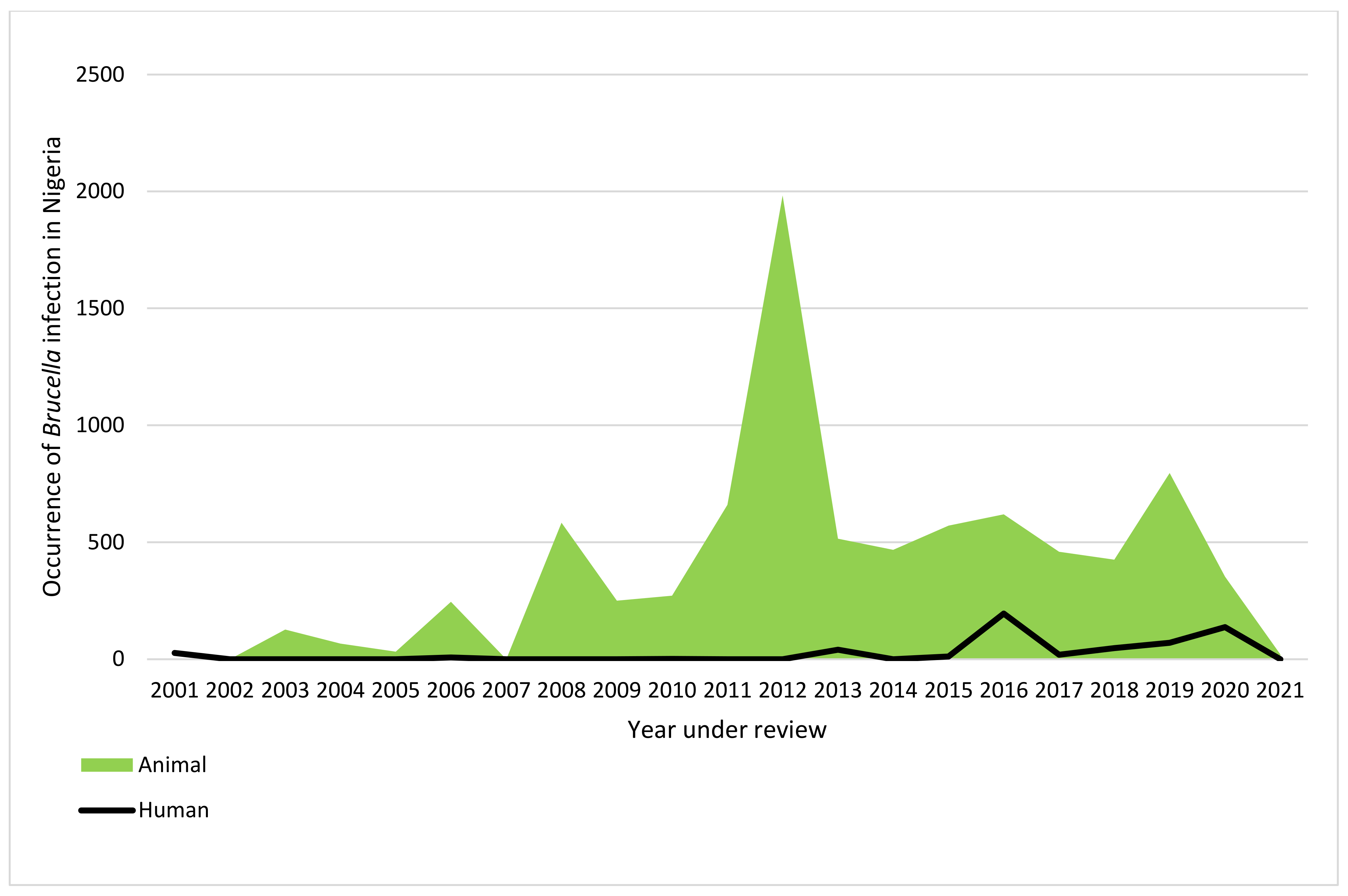
Conclusions: This entry revealed a national prevalence of 19.2% of human Brucella infection and 13.1% prevalence of Brucella infection in common domestic animals, respectively, from 2001 to 2021. This entry also indicated that only 27 states, including the FCT, reported brucellosis in Nigeria across the six geopolitical zones. Researchers' study also revealed the highest occurrence of brucellosis is in Northern Nigeria. Four Brucella species, B. abortus, B. melitensis, B. suis, and B. canis, have been reported in Nigeria. Culture and molecular methods of detection of brucellosis and reports of antimicrobial susceptibility testing remain a conjecture. This entry will help researchers redirect their research focus and serve as a guide for policymakers on measures for managing brucellosis in Nigeria and other sub-Saharan countries.The need for improved sanitary conditions of the abattoirs, the use of personal protective equipment by animal handlers, vaccination of animals against bovine brucellosis, and ranching of animals to curb the spread of the disease should be paramount to all the stakeholders.
| Region | State | No of Samples Tested | No of Positive Sample | Seroprevalence % |
Detection Methods | Brucella Type | Reference | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| RBPT | SAT | cELISA | iELISA | IgM | IgG | IgG/IgM ELISA |
Brucella spp. | Brucella abortus | Brucella melitensis |
||||||
| NC | Kwara | 189 | 42 | 22.2% | 42 | 0 | 0 | 0 | 0 | 0 | 0 | 42 | 0 | 0 | [42] |
| Abuja | 224 | 40 | 17.9% | 40 | 0 | 0 | 0 | 22 | 18 | 0 | 40 | 0 | 0 | [43] | |
| Nasarawa | 160 | 16 | 10% | 16 | 0 | 0 | 0 | 0 | 0 | 0 | 16 | 0 | 0 | [44] | |
| Total | 573 | 98 | 17.1% | 98 | 0 | 0 | 0 | 22 | 18 | 0 | 98 | 0 | 0 | ||
| NE | Bauchi | 285 | 95 | 33.3% | 95 | 0 | 0 | 0 | 6 | 18 | 0 | 95 | 0 | 0 | [45] |
| Not indicated | 500 | 26 | 5.2% | 26 | 0 | 0 | 0 | 0 | 0 | 0 | 26 | 0 | 0 | [46] | |
| Borno | 106 | 4 | 3.8% | 4 | 0 | 0 | 0 | 0 | 0 | 0 | 4 | 0 | 0 | [47] | |
| Borno | 100 | 11 | 11% | 11 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 11 | 0 | [48] | |
| Total | 990 | 136 | 13.7% | 136 | 0 | 0 | 0 | 6 | 18 | 0 | 125 | 11 | 0 | ||
| NW | Kaduna | 1 | 1 | 100% | 0 | 1 | 0 | 0 | 0 | 0 | 0 | 0 | 1 | 0 | [49] |
| Kaduna | 100 | 19 | 19% | 0 | 0 | 0 | 19 | 0 | 0 | 0 | 19 | 0 | 0 | [50] | |
| Sokoto | 137 | 1 | 0.7% | 1 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 1 | 0 | [51] | |
| Total | 238 | 21 | 8.8% | 1 | 1 | 0 | 19 | 0 | 0 | 0 | 19 | 2 | 0 | ||
| SE | Enugu | 682 | 195 | 28.6% | 195 | 0 | 0 | 0 | 0 | 0 | 0 | 195 | 0 | 0 | [41] |
| Total | 682 | 195 | 28.6% | 195 | 0 | 0 | 0 | 0 | 0 | 0 | 195 | 0 | 0 | ||
| SS | Akwa Ibom | 228 | 70 | 30.7% | 29 | 0 | 0 | 0 | 0 | 41 | 0 | 0 | 0 | 70 | [52] |
| Total | 228 | 70 | 30.7% | 29 | 0 | 0 | 0 | 0 | 41 | 0 | 0 | 0 | 70 | ||
| SW | Lagos | 422 | 27 | 6.4% | 27 | 0 | 0 | 3 | 0 | 0 | 0 | 27 | 0 | 0 | [53] |
| Oyo | 11 | 7 | 63.6% | 7 | 0 | 0 | 0 | 0 | 0 | 0 | 7 | 0 | 0 | [54] | |
| Total | 433 | 34 | 7.9% | 34 | 0 | 0 | 3 | 0 | 0 | 0 | 34 | 0 | 0 | ||
| Grand Total | 3144 | 554 | 17.6% | 493 | 1 | 0 | 22 | 28 | 77 | 0 | 471 | 13 | 70 | ||
This entry is adapted from the peer-reviewed paper 10.3390/vetsci9080384
References
- Corbel, M.J. Brucellosis in Humans and Animals; World Health Organization: Geneva, Switzerland, 2006.
- Ducrotoya, M.; Bertub, W.J.; Matopec, G.; Cadmusd, S.; Conde-Álvareze, R.; Gusib, A.M.; Welburna, S.; Ocholib, R.; Blascof, J.M.; Moriyón, I. Brucellosis in Sub-Saharan Africa: Current challenges for management, diagnosis and control. Acta Tropica. 2017, 165, 179–193.
- Bennett, N.J.; Bronze, M.S. Brucellosis. Available online: https://emedicine.medscape.com/article/213430-print (accessed on 20 April 2022).
- World Health Organization. Home/Newsroom/Fact sheets/Detail/Brucellosis; World Health Organization: Geneva, Switzerland, 2020.
- Rossetti, C.A.; Arenas-Gamboa, A.M.; Maurizio, E. Caprine brucellosis: A historically neglected disease with significant impact on public health. PLOS Negl. Trop. Dis. 2017, 11, e0005692.
- Bosilkovski, M.; Keramat, F.; Arapovic, J. The current therapeutic strategies in human brucellosis. Infection 2021, 49, 823–832.
- Cadmus, S.I.; Akporube, K.A.; Ola-Daniel, F.; Adelakun, O.D.; Akinseye, V.O. Seroprevalence and associated factors of brucellosis and Q-fever in cattle from Ibarapa area, Oyo state, south-western Nigeria. Pan Afr. Med. J. 2020, 36, 370.
- Wareth, G.; Dadar, M.; Ali, H.; Hamdy, M.E.R.; Al-Talhy, A.M.; Elkharsawi, A.R.; El Tawab, A.A.A.; Neubauer, H. The perspective of antibiotic therapeutic challenges of brucellosis in the Middle East and North African countries: Current situation and therapeutic management. Transbound. Emerg. Dis. 2022, 1, 1–16.
- El-Diasty, M.; El-Said, R.; Abdelkhalek, A. Seroprevalence and molecular diagnosis of sheep brucellosis in Dakahlia governorate, Egypt. Ger. J. Vet. Res. 2021, 1, 34–39.
- McDermott, J.J.; Arimi, S. Brucellosis in sub-Saharan Africa: Epidemiology, control and impact. Vet. Microbiol. 2002, 90, 111–134.
- United Nations, Department of Economic and Social Affairs, Population Division. World Population Prospects 2019: Highlights; ST/ESA/SER.A/423; United Nations: New York, NY, USA, 2019.
- Adamu, N.N.; Ajogi, I. Serological investigations of camels (Camelus dromedarius) slaughtered at Kano municipal abattoir for evidence of brucellosis. Trop. Vet. 1999, 18, 45–48.
- Ducrotoy, M.J.; Bertu, W.J.; Ocholi, R.A.; Gusi, A.M.; Bryssinckx, W.; Welburn, S.; Moriyón, I. Brucellosis as an Emerging Threat in Developing Economies: Lessons from Nigeria. PLoS Negl. Trop. Dis. 2014, 8, e3008.
- Dean, A.S.; Crump, L.; Greter, H.; Hattendorf, J.; Schelling, E.; Zinsstag, J. Clinical Manifestations of Human Brucellosis: A Systematic Review and Meta-Analysis. PLOS Negl. Trop. Dis. 2012, 6, e1929.
- Laine, C.G.; Wade, A.; Scott, H.M.; Krecek, R.C.; Arenas-Gamboa, A.M. Scoping review of brucellosis in Cameroon: Where do we stand, and where are we going? PLoS ONE 2020, 15, e0239854.
- Kahariri, S.M.; Kitala, P.M.; Muchemi, G.M.; Njenga, K.; Nanyingi, M. Sero-prevalence and risk factors for human brucellosis in Marsabit county, Kenya (2014). PAMJ One Health 2021, 4, 9.
- Mehari, S.; Zerfu, B.; Desta, K. Prevalence and risk factors of human brucellosis and malaria among patients with fever in malaria-endemic areas, attending health institutes in Awra and Gulina district, Afar Region, Ethiopia. BMC Infect. Dis. 2021, 21, 942.
- Tumwine, G.; Matovu, E.; Kabasa, J.D.; Owiny, D.O.; Majalija, S. Human brucellosis: Sero-prevalence and associated risk factors in agro-pastoral communities of Kiboga District, Central Uganda. BMC Public Health 2015, 15, 900.
- Diab, M.S.; Zidan, S.A.A.; Hassan, N.A.A.; Elaadli, H.; Bayoumi, A.M. Seroprevalence and Associated Risk Factors of Brucellosis in Livestock and Residents of New Valley Governorate, Egypt. World’s Vet. J. 2020, 10, 531–539.
- Sagamiko, F.D.; Mfune, R.L.; Hang’Ombe, B.M.; Karimuribo, E.D.; Mwanza, A.M.; Sindato, C.; Muma, J.B. Seroprevalence of human Brucellosis and associated risk factors among high-risk occupations in Mbeya Region of Tanzania. J. Epidemiol. Res. 2020, 6, 1.
- Njeru, J.; Wareth, G.; Melzer, F.; Henning, K.; Pletz, M.W.; Heller, R.; Neubauer, H. Systematic review of brucellosis in Kenya: Disease frequency in humans and animals and risk factors for human infection. BMC Public Health 2016, 16, 853.
- Bale, J.O.; Kumi-Diaka, J. Serological and bacteriological study of bovine Brucellae from livestock investigation and breeding centers in Nigeria. Br. Vet. J. 1981, 37, 256–261.
- Gameel, S.E.A.M.; Mohamed, S.O.; Mustafa, A.A.; Azwai, S.M. Prevalence of camel brucellosis in Libya. Trop. Anim. Health Prod. 1993, 25, 91–93.
- Bello-Onaghise, G.; Vaikosen, S.E.; Evivie, S.E. Abortion cases in pig farms in Benin city and some surrounding communities in Edo State, Nigeria. Niger. J. Agri. Food Env. 2012, 8, 37–42.
- Ogugua, A.; Akinseye, V.; Ayoola, M.; Oyesola, O.; Shima, F.; Tijjani, A.; Musa, A.N.A.; Adesokan, H.; Perrett, L.; Taylor, A.; et al. Seroprevalence and risk factors of brucellosis in goats in selected states in Nigeria and the public health implications. Afr. J. Med. Med Sci. 2014, 43 (Suppl. 1), 121–129.
- Bertu, W.; Ocholi, R.; Gusi, A.; Ngulukun, S.; Ducrotoy, M.; Moriyon, I. Isolation of brucella strains in cattle from sedentary and nomadic communities and its public health implication. Int. J. Infect. Dis. 2020, 101, 533.
- Bertu, W.J.; Ocholi, R.A.; Gusi, A.M.; Abdullahi, S.; Zwandor, N.J.; Durbi, I.A.A.; Opara, J.; Okewole, P.A. Brucella abortus infection in a multispecies livestock farm in Nigeria. Int. J. Biotec. Food Sci. 2015, 3, 36–40.
- Ocholi, R.; Kwaga, J.; Ajogi, I.; Bale, J. Phenotypic characterization of Brucella strains isolated from livestock in Nigeria. Vet. Microbiol. 2004, 103, 47–53.
- Ocholi, R.A.; Kwaga, J.K.; Ajogi, I.; Bale, J.O. Abortion due to Brucella abortus in sheep in Nigeria. Rev. Sci. Tech. 2005, 24, 973–979.
- Maurice, N.A. Bacteriological and serological studies of bovine brucellosis in Obudu cattle ranch, Cross River State. Nigeria. Eur. J. Exp. Biol. 2013, 3, 484–488.
- Yusuf, Y.; Abdulrasheed, A. Survey on Bovine Brucellosis in Sokoto Metropolitan Abattoir, Nigeria. J. Anim. Vet. Adv. 2019, 18, 143–146.
- Lucero, N.E.; Ayala, S.M.; Escobar, G.I.; Jacob, N.R. Brucella isolated in humans and animals in Latin America from 1968 to 2006. Epidemiol. Infect. 2008, 136, 496–503.
- Minharro, S.; Mol, J.P.S.; Dorneles, E.M.S.; Pauletti, R.B.; Neubauer, H.; Melzer, F.; Poester, F.P.; Dasso, M.G.; Pinheiro, E.S.; Filho, P.M.S.; et al. Biotyping and Genotyping (MLVA16) of Brucella abortus Isolated from Cattle in Brazil, 1977 to 2008. PLoS ONE 2013, 8, e81152.
- Sanogo, M.; Thys, E.; Achi, Y.L.; Fretin, D.; Michel, P.; Abatih, E.; Berkvens, D.; Saegerman, C. Bayesian estimation of the true prevalence, sensitivity and specificity of the Rose Bengal and indirect ELISA tests in the diagnosis of bovine brucellosis. Vet. J. 2013, 195, 114–120.
- Liu, F.; Li, J.-M.; Zeng, F.-L.; Zong, Y.; Leng, X.; Shi, K.; Diao, N.-C.; Li, D.; Li, B.-Y.; Zhao, Q.; et al. Prevalence and Risk Factors of Brucellosis, Chlamydiosis, and Bluetongue Among Sika Deer in Jilin Province in China. Vector Borne Zoonotic Dis. 2018, 18, 226–230.
- Alton, G.G.; Jones, L.M.; Angus, R.D.; Verger, J.M. Techniques for the Brucellosis Laboratory; INRA: Paris, France, 1988.
- Bricker, B.J.; Ewalt, D.R.; Halling, S.M. Brucella ‘HOOF-Prints’: Strain typing by multi-locus analysis of variable number tandem repeats (VNTRs). BMC Microbiol. 2003, 3, 15.
- Le Flèche, P.; Jacques, I.; Grayon, M.; Al Dahouk, S.; Bouchon, P.; Denoeud, F.; Nöckler, K.; Neubauer, H.; Guilloteau, L.A.; Vergnaud, G. Evaluation and selection of tandem repeat loci for a Brucella MLVA typing assay. BMC Microbiol. 2006, 6, 9–14.
- Robinson, R. Emergency Prevention System for Transboundary Animal and Plant Pests and Diseases. In Guidelines for Coordinated Human and Animal Brucellosis Surveillance; Food and Agriculture Organization of the United Nations: Rome, Italy, 2003.
- World Organization for Animal Health (OIE). Bovine Brucellosis. In Manual of Diagnostic Test and Vaccines for Terrestrial Animals; OIE: Paris, France, 2009; Chapter 2.4.3.; pp. 1–35.
- Ogbodo, S.O.; Isiofia, O.P.; Uzodinma, B.A. Co-existence and seroprevalence of brucellosis in a malaria-endemic metropolis of south-eastern Nigeria. J. Exp. Res. 2016, 4, 2.
- Bamidele, F.; Gidado, S.; Edukugho, A.; Cadmus, S. Seroprevalence of Brucellosis in abattoir workers and slaughtered cattle in Ilorin metropolis Kwara State Nigeria. Int. J. Infect. Dis. 2020, 101, 532–533.
- Aworh, M.; Okolocha, E.; Kwaga, J.; Fasina, F.; Lazarus, D.; Suleman, I.; Poggensee, G.; Nguku, P.; Nsubuga, P. Human brucellosis: Seroprevalence and associated exposure factors among abattoir workers in Abuja, Nigeria—2011. Pan Afr. Med. J. 2013, 16, 103.
- Agada, C.; Mohammed, J.; Okoh, A.E.J.; Ogugua, J.A. Prevalence and risk factors associated with brucellosis among high-risk individuals in Lafia, Nasarawa state, Nigeria. Int. J. One Health 2018, 4, 45–51.
- Igawe, P.B.; Okolocha, E.; Kia, G.S.; Irmiya, I.B.; Balogun, M.S.; Nguku, P.; Bugun, I.I. Seroprevalence of brucellosis and associated exposure factors among in Bauchi state, Nigeria. Pan Afr. Med. J. 2020, 35, 33.
- Baba, M.M.; Sarkindared, S.E.; Brisibe, F. Serological evidence of brucellosis among predisposed patients with pyrexia of unknown origin in the north eastern Nigeria. Central Eur. J. Public Health 2001, 9, 158–161.
- Atsanda, N.; Liba, J.; Francis, M.; Malgwi, H. Serological survey of brucellosis among internally displaced persons in Maiduguri, North eastern Nigeria. Sokoto J. Vet. Sci. 2018, 16, 61–65.
- Adamu, N.B.; Adeniyi, S.O.; Adamu, S.G.; Bale, J.O.O.; Okoh, A.E.J.; Umaru, G.A.; Umar, Y.A. Seroprevalence of brucellosis among livestock workers at Maiduguri cattle market, Borno State, North-Eastern, Nigeria. J. Public Health Epidemiol. 2015, 7, 253–257.
- Obiako, O.R.; Ogoina, D.; Danbauchi, S.S.; Kwaifa, S.I.; Chom, N.D.; Nwokorie, E. Neurobrucellosis—a case report and review of literature. Niger. J. Clin. Pract. 2010, 13, 347–350.
- Adesiyun, A.G.; Folagbade, O.B.; Olayinka, A.T.; Randawa, A.; Bawa, U. Seroprevalence of brucellosis among women with miscarriage at Ahmadu Bello University Teaching Hospital, Zaria. Trop. J. Obstet. Gynaecol. 2017, 34, 145–151.
- Cadmus, S.; Salam, S.P.; Adesokan, H.K.; Akporube, K.; Ola-Daniel, F.; Awosanya, E.J. Seroprevalence of brucellosis and Q fever infections amongst pastoralists and their cattle herds in Sokoto State, Nigeria. PLoS ONE 2021, 16, e0254530.
- Owowo, E.E.; Antia, U.E.; Christopher, M.A.; Okon, I.E. Sero-Prevalence of Brucellosis among Nomadic Herdsmen, Abattoir and Livestock Workers in Niger-Delta Region, Nigeria. J. Biosci. Med. 2019, 7, 32–41.
- Adeyemi, A.K. Survey of Brucellosis among People at Risk in Lagos, Nigeria. Master’s Dissertation, University of South Africa, Pretoria, South Africa, 2018.
- Cadmus, S.; Ijagbone, I.; Oputa, H.; Adesokan, H.; Stack, J. Serological survey of Brucellosis in livestock animals and workers in Ibadan, Nigeria. Afr. J. Biomed. Res. 2009, 9, 3.
