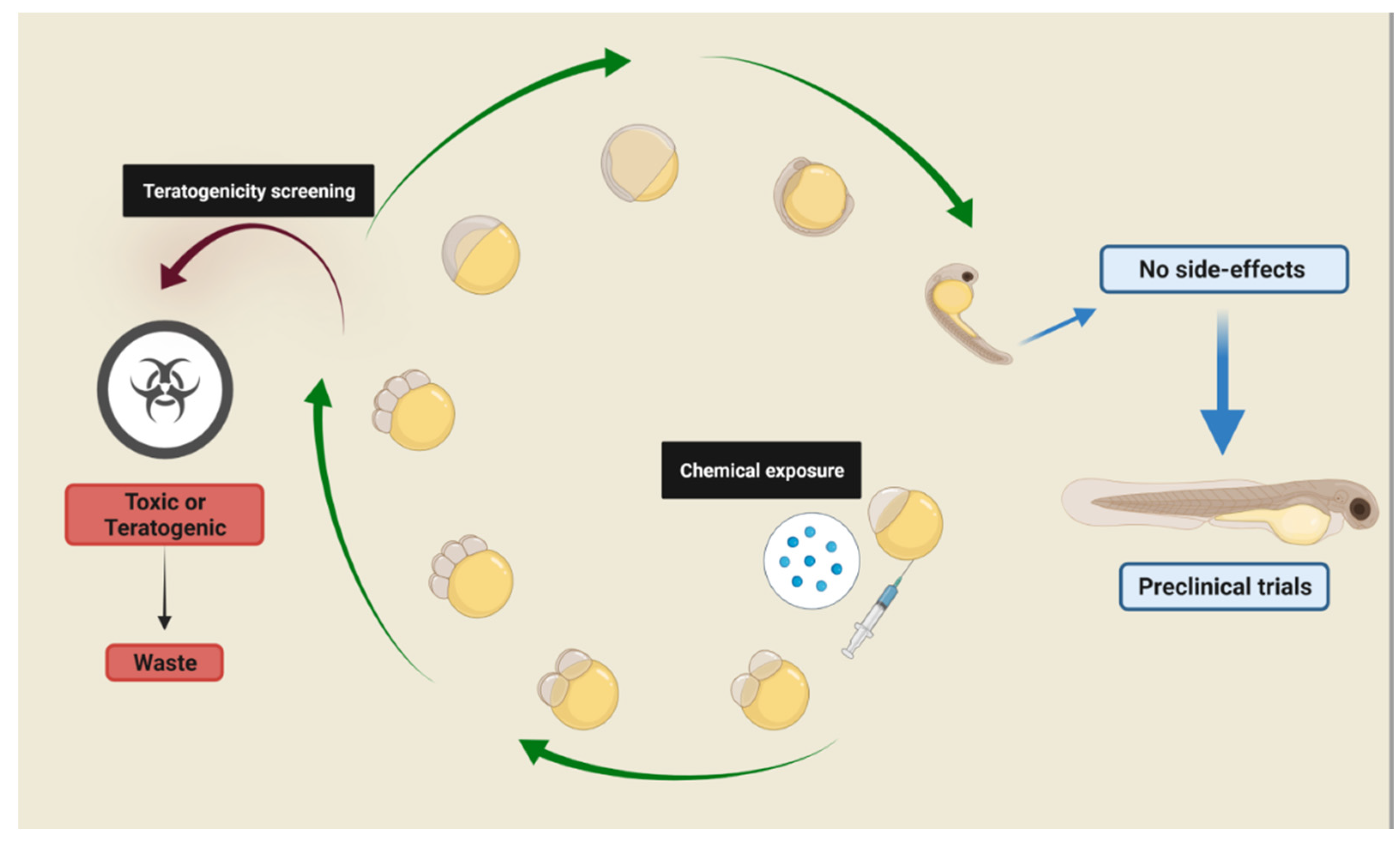
Phenotype-based screenings can transpose the organism’s compensatory pathways by adopting multi-target strategies for treating complex diseases, and zebrafish emerged as an important model for biomedical research and drug screenings. Zebrafish’s clear correlation between neuro-anatomical and physiological features and behavior is very similar to that verified in mammals, enabling the construction of reliable and relevant experimental models for neurological disorders research. Zebrafish presents highly conserved physiological pathways that are found in higher vertebrates, including mammals, along with a robust behavioral repertoire. Moreover, it is very sensitive to pharmacological/environmental manipulations, and these behavioral phenotypes are detected in both larvae and adults. These advantages align with the 3Rs concept and qualify the zebrafish as a powerful tool for drug screenings and pre-clinical trials.
- embryo-larval stage
- alternative model
- 3Rs
- behavioral repertoire
- drug discovery
- neurological drugs
- neurotransmitters
- anxiety-like behavior
1. Zebrafish as a Model for Phenotype-Based Screening
2. Zebrafish Neurotransmitter Systems
3. Neurological Functions and Behavior Models towards Pre-Clinical Assays
|
BEHAVIORAL TEST |
ENDPOINTS |
REFERENCE |
|---|---|---|
|
LIGHT-DARK TEST |
Total distance traveled |
[59] |
|
VISUAL MOTOR RESPONSE |
Velocity, total distance moved, and mobility time |
[60] |
|
LOCOMOTOR ACTIVITY |
Velocity, total distance moved, and mobility time |
[61] |
|
LOCOMOTOR ACTIVITY |
Total distance traveled |
[62] |
|
ACOUSTIC STARTLE RESPONSE |
Head angle |
[63] |
|
VISUAL MOTOR RESPONSE |
Total distance traveled |
[64] |
|
VISUAL MOTOR RESPONSE |
Average distance traveled |
[65] |
|
VISUAL MOTOR RESPONSE |
Burst swim |
[66] |
|
VISUAL MOTOR RESPONSE |
Total distance traveled |
[67] |
|
VIBRATIONAL STARTLE RESPONSE |
Total distance traveled |
[67] |
|
LOCOMOTOR ACTIVITY |
Total distance traveled, mean speed, turn angle |
[68] |
|
THIGMOTAXIS |
Entries in outer area |
[68] |
|
LIGHT-DARK TEST |
Total distance traveled |
[69] |
|
THIGMOTAXIS |
Distance traveled in outer area |
[69] |
|
THIGMOTAXIS |
Percentage of distance moved in outer zone |
[70] |
|
VISUAL MOTOR RESPONSE |
Total distance traveled |
[71] |
|
THIGMOTAXIS |
Distance traveled/time spent in each zone |
[71] |
|
THIGMOTAXIS |
Percentage of distance moved in outer zone |
[72] |
|
LOCOMOTOR ACTIVITY |
Average distance traveled |
[72] |
|
PHOTOMOTOR RESPONSE |
Movements/5 min |
[73] |
|
LOCOMOTOR ACTIVITY |
Total distance traveled |
[73] |
|
VISUAL MOTOR RESPONSE |
Total distance traveled |
[74] |
This entry is adapted from the peer-reviewed paper 10.3390/ijms23126647
References
- Swinney, D.C.; Anthony, J. How Were New Medicines Discovered? Nat. Rev. Drug Discov. 2011, 10, 507–519.
- MacRae, C.A.; Peterson, R.T. Zebrafish as Tools for Drug Discovery. Nat. Rev. Drug Discov. 2015, 14, 721–731.
- Moffat, J.G.; Vincent, F.; Lee, J.A.; Eder, J.; Prunotto, M. Opportunities and Challenges in Phenotypic Drug Discovery: An Industry Perspective. Nat. Rev. Drug Discov. 2017, 16, 531–543.
- Howe, K.; Clark, M.D.; Torroja, C.F.; Torrance, J.; Berthelot, C.; Muffato, M.; Collins, J.E.; Humphray, S.; McLaren, K.; Matthews, L.; et al. The Zebrafish Reference Genome Sequence and Its Relationship to the Human Genome. Nature 2013, 496, 498–503.
- Lowery, L.A.; De Rienzo, G.; Gutzman, J.H.; Sive, H. Characterization and Classification of Zebrafish Brain Morphology Mutants. Anat. Rec. Adv. Integr. Anat. Evol. Biol. 2009, 292, 94–106.
- Geisler, R.; Köhler, A.; Dickmeis, T.; Strähle, U. Archiving of Zebrafish Lines Can Reduce Animal Experiments in Biomedical Research. EMBO Rep. 2017, 18, 1–2.
- Strähle, U.; Scholz, S.; Geisler, R.; Greiner, P.; Hollert, H.; Rastegar, S.; Schumacher, A.; Selderslaghs, I.; Weiss, C.; Witters, H.; et al. Zebrafish Embryos as an Alternative to Animal Experiments—A Commentary on the Definition of the Onset of Protected Life Stages in Animal Welfare Regulations. Reprod. Toxicol. 2012, 33, 128–132.
- Saint-Amant, L.; Drapeau, P. Time Course of the Development of Motor Behaviors in the Zebrafish Embryo. J. Neurobiol. 1998, 37, 622–632.
- Orger, M.B.; de Polavieja, G.G. Zebrafish Behavior: Opportunities and Challenges. Annu. Rev. Neurosci. 2017, 40, 125–147.
- Cassar, S.; Adatto, I.; Freeman, J.L.; Gamse, J.T.; Iturria, I.; Lawrence, C.; Muriana, A.; Peterson, R.T.; Van Cruchten, S.; Zon, L.I. Use of Zebrafish in Drug Discovery Toxicology. Chem. Res. Toxicol. 2020, 33, 95–118.
- Förster, D.; Arnold-Ammer, I.; Laurell, E.; Barker, A.J.; Fernandes, A.M.; Finger-Baier, K.; Filosa, A.; Helmbrecht, T.O.; Kölsch, Y.; Kühn, E.; et al. Genetic Targeting and Anatomical Registration of Neuronal Populations in the Zebrafish Brain with a New Set of BAC Transgenic Tools. Sci. Rep. 2017, 7, 5230.
- Li, Z.H.; Alex, D.; Siu, S.O.; Chu, I.K.; Renn, J.; Winkler, C.; Lou, S.; Tsui, S.K.-W.; Zhao, H.Y.; Yan, W.R.; et al. Combined in Vivo Imaging and Omics Approaches Reveal Metabolism of Icaritin and Its Glycosides in Zebrafish Larvae. Mol. Biosyst. 2011, 7, 2128.
- Jeong, J.-Y.; Kwon, H.-B.; Ahn, J.-C.; Kang, D.; Kwon, S.-H.; Park, J.A.; Kim, K.-W. Functional and Developmental Analysis of the Blood–Brain Barrier in Zebrafish. Brain Res. Bull. 2008, 75, 619–628.
- Goldstone, J.V.; McArthur, A.G.; Kubota, A.; Zanette, J.; Parente, T.; Jönsson, M.E.; Nelson, D.R.; Stegeman, J.J. Identification and Developmental Expression of the Full Complement of Cytochrome P450 Genes in Zebrafish. BMC Genom. 2010, 11, 643.
- Khan, K.M.; Collier, A.D.; Meshalkina, D.A.; Kysil, E.V.; Khatsko, S.L.; Kolesnikova, T.; Morzherin, Y.Y.; Warnick, J.E.; Kalueff, A.V.; Echevarria, D.J. Zebrafish Models in Neuropsychopharmacology and CNS Drug Discovery. Br. J. Pharmacol. 2017, 174, 1925–1944.
- Sakai, C.; Ijaz, S.; Hoffman, E.J. Zebrafish Models of Neurodevelopmental Disorders: Past, Present, and Future. Front. Mol. Neurosci. 2018, 11, 294.
- Haug, M.F.; Gesemann, M.; Mueller, T.; Neuhauss, S.C.F. Phylogeny and Expression Divergence of Metabotropic Glutamate Receptor Genes in the Brain of Zebrafish (Danio rerio). J. Comp. Neurol. 2013, 521, 1533–1560.
- Doldn, M.J.; Prego, B.; Holmqvist, B.I.; de Miguel, E. Distribution of GABA-Immunolabeling in the Early Zebrafish (Danio rerio) Brain. Eur. J. Morphol. 1999, 37, 126–129.
- Assad, N.; Luz, W.L.; Santos-Silva, M.; Carvalho, T.; Moraes, S.; Picanço-Diniz, D.L.W.; Bahia, C.P.; de Oliveira Batista, E.J.; da Conceição Passos, A.; Oliveira, K.R.H.M.; et al. Acute Restraint Stress Evokes Anxiety-Like Behavior Mediated by Telencephalic Inactivation and GabAergic Dysfunction in Zebrafish Brains. Sci. Rep. 2020, 10, 5551.
- Mueller, T.; Vernier, P.; Wullimann, M.F. A Phylotypic Stage in Vertebrate Brain Development: GABA Cell Patterns in Zebrafish Compared with Mouse. J. Comp. Neurol. 2006, 494, 620–634.
- Roberts, A.C.; Bill, B.R.; Glanzman, D.L. Learning and Memory in Zebrafish Larvae. Front. Neural Circuits 2013, 7, 126.
- McLean, D.L.; Fetcho, J.R. Ontogeny and Innervation Patterns of Dopaminergic, Noradrenergic, and Serotonergic Neurons in Larval Zebrafish. J. Comp. Neurol. 2004, 480, 38–56.
- Kaslin, J.; Panula, P. Comparative Anatomy of the Histaminergic and Other Aminergic Systems in Zebrafish (Danio rerio). J. Comp. Neurol. 2001, 440, 342–377.
- Ek, F.; Malo, M.; Åberg Andersson, M.; Wedding, C.; Kronborg, J.; Svensson, P.; Waters, S.; Petersson, P.; Olsson, R. Behavioral Analysis of Dopaminergic Activation in Zebrafish and Rats Reveals Similar Phenotypes. ACS Chem. Neurosci. 2016, 7, 633–646.
- Schweitzer, J.; Driever, W. Development of the Dopamine Systems in Zebrafish. In Development and Engineering of Dopamine Neurons; Springer: Berlin/Heidelberg, Germany, 2009; Volume 651, pp. 1–14.
- Tay, T.L.; Ronneberger, O.; Ryu, S.; Nitschke, R.; Driever, W. Comprehensive Catecholaminergic Projectome Analysis Reveals Single-Neuron Integration of Zebrafish Ascending and Descending Dopaminergic Systems. Nat. Commun. 2011, 2, 171.
- Alsop, D.; Vijayan, M. The Zebrafish Stress Axis: Molecular Fallout from the Teleost-Specific Genome Duplication Event. Gen. Comp. Endocrinol. 2009, 161, 62–66.
- Clemente, D.; Porteros, Á.; Weruaga, E.; Alonso, J.R.; Arenzana, F.J.; Aijón, J.; Arévalo, R. Cholinergic Elements in the Zebrafish Central Nervous System: Histochemical and Immunohistochemical Analysis. J. Comp. Neurol. 2004, 474, 75–107.
- Mueller, T.; Vernier, P.; Wullimann, M.F. The Adult Central Nervous Cholinergic System of a Neurogenetic Model Animal, the Zebrafish Danio rerio. Brain Res. 2004, 1011, 156–169.
- Papke, R.L.; Ono, F.; Stokes, C.; Urban, J.M.; Boyd, R.T. The Nicotinic Acetylcholine Receptors of Zebrafish and an Evaluation of Pharmacological Tools Used for Their Study. Biochem. Pharmacol. 2012, 84, 352–365.
- Bhattarai, P.; Thomas, A.K.; Cosacak, M.I.; Papadimitriou, C.; Mashkaryan, V.; Zhang, Y.; Kizil, C. Modeling Amyloid-Β42 Toxicity and Neurodegeneration in Adult Zebrafish Brain. J. Vis. Exp. 2017, 128, 56014.
- Koehler, D.; Shah, Z.A.; Williams, F.E. The GSK3β Inhibitor, TDZD-8, Rescues Cognition in a Zebrafish Model of Okadaic Acid-Induced Alzheimer’s Disease. Neurochem. Int. 2019, 122, 31–37.
- Vaz, R.L.; Outeiro, T.F.; Ferreira, J.J. Zebrafish as an Animal Model for Drug Discovery in Parkinson’s Disease and Other Movement Disorders: A Systematic Review. Front. Neurol. 2018, 9, 347.
- Woodard, A.; Barbery, B.; Wilkinson, R.; Strozyk, J.; Milner, M.; Doucette, P.; Doran, J.; Appleby, K.; Atwill, H.; Bell, W.E.; et al. The Role of Neuronal Nitric Oxide and Its Pathways in the Protection and Recovery from Neurotoxin-Induced de Novo Hypokinetic Motor Behaviors in the Embryonic Zebrafish (Danio rerio). AIMS Neurosci. 2019, 6, 25–42.
- Lundegaard, P.R.; Anastasaki, C.; Grant, N.J.; Sillito, R.R.; Zich, J.; Zeng, Z.; Paranthaman, K.; Larsen, A.P.; Armstrong, J.D.; Porteous, D.J.; et al. MEK Inhibitors Reverse CAMP-Mediated Anxiety in Zebrafish. Chem. Biol. 2015, 22, 1335–1346.
- Wong, K.; Stewart, A.; Gilder, T.; Wu, N.; Frank, K.; Gaikwad, S.; Suciu, C.; DiLeo, J.; Utterback, E.; Chang, K.; et al. Modeling Seizure-Related Behavioral and Endocrine Phenotypes in Adult Zebrafish. Brain Res. 2010, 1348, 209–215.
- Rahn, J.J.; Bestman, J.E.; Josey, B.J.; Inks, E.S.; Stackley, K.D.; Rogers, C.E.; Chou, C.J.; Chan, S.S.L. Novel Vitamin K Analogs Suppress Seizures in Zebrafish and Mouse Models of Epilepsy. Neuroscience 2014, 259, 142–154.
- Yang, X.; Lin, J.; Peng, X.; Zhang, Q.; Zhang, Y.; Guo, N.; Zhou, S.; Li, Q. Effects of Picrotoxin on Zebrafish Larvae Behaviors: A Comparison Study with PTZ. Epilepsy Behav. 2017, 70, 224–231.
- Patten, S.A.; Parker, J.A.; Wen, X.-Y.; Drapeau, P. Simple Animal Models for Amyotrophic Lateral Sclerosis Drug Discovery. Expert Opin. Drug Discov. 2016, 11, 797–804.
- Ferguson, R.; Holloway, D.E.; Chandrasekhar, A.; Acharya, K.R.; Subramanian, V. The Catalytic Activity and Secretion of Zebrafish RNases Are Essential for Their in Vivo Function in Motor Neurons and Vasculature. Sci. Rep. 2019, 9, 1107.
- Martineau, P.R.; Mourrain, P. Tracking Zebrafish Larvae in Group—Status and Perspectives. Methods 2013, 62, 292–303.
- Mirat, O.; Sternberg, J.R.; Severi, K.E.; Wyart, C. ZebraZoom: An Automated Program for High-Throughput Behavioral Analysis and Categorization. Front. Neural Circuits 2013, 7, 107.
- Kirsten, K.; Soares, S.M.; Koakoski, G.; Carlos Kreutz, L.; Barcellos, L.J.G. Characterization of Sickness Behavior in Zebrafish. Brain. Behav. Immun. 2018, 73, 596–602.
- Agetsuma, M.; Aizawa, H.; Aoki, T.; Nakayama, R.; Takahoko, M.; Goto, M.; Sassa, T.; Amo, R.; Shiraki, T.; Kawakami, K.; et al. The Habenula Is Crucial for Experience-Dependent Modification of Fear Responses in Zebrafish. Nat. Neurosci. 2010, 13, 1354–1356.
- Perathoner, S.; Cordero-Maldonado, M.L.; Crawford, A.D. Potential of Zebrafish as a Model for Exploring the Role of the Amygdala in Emotional Memory and Motivational Behavior. J. Neurosci. Res. 2016, 94, 445–462.
- Kenney, J.W.; Scott, I.C.; Josselyn, S.A.; Frankland, P.W. Contextual Fear Conditioning in Zebrafish. Learn. Mem. 2017, 24, 516–523.
- Braida, D.; Ponzoni, L.; Martucci, R.; Sala, M. A New Model to Study Visual Attention in Zebrafish. Prog. Neuro-Psychopharmacol. Biol. Psychiatry 2014, 55, 80–86.
- Gaspary, K.V.; Reolon, G.K.; Gusso, D.; Bonan, C.D. Novel Object Recognition and Object Location Tasks in Zebrafish: Influence of Habituation and NMDA Receptor Antagonism. Neurobiol. Learn. Mem. 2018, 155, 249–260.
- Santacà, M.; Dadda, M.; Miletto Petrazzini, M.E.; Bisazza, A. Stimulus Characteristics, Learning Bias and Visual Discrimination in Zebrafish (Danio rerio). Behav. Processes 2021, 192, 104499.
- Al-Imari, L.; Gerlai, R. Sight of Conspecifics as Reward in Associative Learning in Zebrafish (Danio rerio). Behav. Brain Res. 2008, 189, 216–219.
- Maximino, C.; Meinerz, D.L.; Fontana, B.D.; Mezzomo, N.J.; Stefanello, F.V.; de Prestes, A.S.; Batista, C.B.; Rubin, M.A.; Barbosa, N.V.; Rocha, J.B.T.; et al. Extending the Analysis of Zebrafish Behavioral Endophenotypes for Modeling Psychiatric Disorders: Fear Conditioning to Conspecific Alarm Response. Behav. Processes 2018, 149, 35–42.
- Menezes, F.P.; Amorim, R.R.; Silva, P.F.; Luchiari, A.C. Alcohol Exposure and Environmental Enrichment Effects on Contextual Fear Conditioning in Zebrafish. Behav. Processes 2022, 197, 104608.
- Panula, P.; Sallinen, V.; Sundvik, M.; Kolehmainen, J.; Torkko, V.; Tiittula, A.; Moshnyakov, M.; Podlasz, P. Modulatory Neurotransmitter Systems and Behavior: Towards Zebrafish Models of Neurodegenerative Diseases. Zebrafish 2006, 3, 235–247.
- Alsop, D.; Vijayan, M.M. Development of the Corticosteroid Stress Axis and Receptor Expression in Zebrafish. Am. J. Physiol. Integr. Comp. Physiol. 2008, 294, R711–R719.
- Barcellos, L.J.G.; Ritter, F.; Kreutz, L.C.; Quevedo, R.M.; da Silva, L.B.; Bedin, A.C.; Finco, J.; Cericato, L. Whole-Body Cortisol Increases after Direct and Visual Contact with a Predator in Zebrafish, Danio rerio. Aquaculture 2007, 272, 774–778.
- Cachat, J.; Stewart, A.; Utterback, E.; Hart, P.; Gaikwad, S.; Wong, K.; Kyzar, E.; Wu, N.; Kalueff, A.V. Three-Dimensional Neurophenotyping of Adult Zebrafish Behavior. PLoS ONE 2011, 6, e17597.
- Bencan, Z.; Sledge, D.; Levin, E.D. Buspirone, Chlordiazepoxide and Diazepam Effects in a Zebrafish Model of Anxiety. Pharmacol. Biochem. Behav. 2009, 94, 75–80.
- Blaser, R.E.; Chadwick, L.; McGinnis, G.C. Behavioral Measures of Anxiety in Zebrafish (Danio rerio). Behav. Brain Res. 2010, 208, 56–62.
- Basnet, R.; Zizioli, D.; Taweedet, S.; Finazzi, D.; Memo, M. Zebrafish Larvae as a Behavioral Model in Neuropharmacology. Biomedicines 2019, 7, 23.
- Suryanto, M.E.; Audira, G.; Uapipatanakul, B.; Hussain, A.; Saputra, F.; Siregar, P.; Chen, K.H.-C.; Hsiao, C.-D. Antidepressant Screening Demonstrated Non-Monotonic Responses to Amitriptyline, Amoxapine and Sertraline in Locomotor Activity Assay in Larval Zebrafish. Cells 2021, 10, 738.
- Colwill, R.M.; Creton, R. Locomotor Behaviors in Zebrafish (Danio rerio) Larvae. Behav. Processes 2011, 86, 222–229.
- Afrikanova, T.; Serruys, A.-S.K.; Buenafe, O.E.M.; Clinckers, R.; Smolders, I.; de Witte, P.A.M.; Crawford, A.D.; Esguerra, C.V. Validation of the Zebrafish Pentylenetetrazol Seizure Model: Locomotor versus Electrographic Responses to Antiepileptic Drugs. PLoS ONE 2013, 8, e54166.
- Burgess, H.A.; Granato, M. Sensorimotor Gating in Larval Zebrafish. J. Neurosci. 2007, 27, 4984–4994.
- Fernandes, A.M.; Fero, K.; Arrenberg, A.B.; Bergeron, S.A.; Driever, W.; Burgess, H.A. Deep Brain Photoreceptors Control Light-Seeking Behavior in Zebrafish Larvae. Curr. Biol. 2012, 22, 2042–2047.
- Emran, F.; Rihel, J.; Dowling, J.E. A Behavioral Assay to Measure Responsiveness of Zebrafish to Changes in Light Intensities. J. Vis. Exp. 2008, 20, e923.
- Gao, Y.; Zhang, G.; Jelfs, B.; Carmer, R.; Venkatraman, P.; Ghadami, M.; Brown, S.A.; Pang, C.P.; Leung, Y.F.; Chan, R.H.M.; et al. Computational Classification of Different Wild-Type Zebrafish Strains Based on Their Variation in Light-Induced Locomotor Response. Comput. Biol. Med. 2016, 69, 1–9.
- Faria, M.; Prats, E.; Bellot, M.; Gomez-Canela, C.; Raldúa, D. Pharmacological Modulation of Serotonin Levels in Zebrafish Larvae: Lessons for Identifying Environmental Neurotoxicants Targeting the Serotonergic System. Toxics 2021, 9, 118.
- Zimmermann, F.F.; Gaspary, K.V.; Leite, C.E.; De Paula Cognato, G.; Bonan, C.D. Embryological Exposure to Valproic Acid Induces Social Interaction Deficits in Zebrafish (Danio rerio): A Developmental Behavior Analysis. Neurotoxicol. Teratol. 2015, 52, 36–41.
- Thompson, W.A.; Arnold, V.I.; Vijayan, M.M. Venlafaxine in Embryos Stimulates Neurogenesis and Disrupts Larval Behavior in Zebrafish. Environ. Sci. Technol. 2017, 51, 12889–12897.
- Schnörr, S.J.; Steenbergen, P.J.; Richardson, M.K.; Champagne, D.L. Measuring Thigmotaxis in Larval Zebrafish. Behav. Brain Res. 2012, 228, 367–374.
- Han, S.; Zhang, D.; Dong, Q.; Wang, X.; Wang, L. Overexpression of Neuroserpin in Larval and Adult Zebrafish Shows Different Behavioral Phenotypes. Neurosci. Lett. 2021, 762, 136175.
- Maphanga, V.B.; Skalicka-Wozniak, K.; Budzynska, B.; Skiba, A.; Chen, W.; Agoni, C.; Enslin, G.M.; Viljoen, A.M. Mesembryanthemum tortuosum L. Alkaloids Modify Anxiety-like Behaviour in a Zebrafish Model. J. Ethnopharmacol. 2022, 290, 115068.
- Copmans, D.; Kildgaard, S.; Rasmussen, S.A.; Ślęzak, M.; Dirkx, N.; Partoens, M.; Esguerra, C.V.; Crawford, A.D.; Larsen, T.O.; de Witte, P.A.M. Zebrafish-Based Discovery of Antiseizure Compounds from the North Sea: Isoquinoline Alkaloids TMC-120A and TMC-120B. Mar. Drugs 2019, 17, 607.
- Ganzen, L.; Ko, M.J.; Zhang, M.; Xie, R.; Chen, Y.; Zhang, L.; James, R.; Mumm, J.; van Rijn, R.M.; Zhong, W.; et al. Drug Screening with Zebrafish Visual Behavior Identifies Carvedilol as a Potential Treatment for an Autosomal Dominant Form of Retinitis Pigmentosa. Sci. Rep. 2021, 11, 11432.
