Searching for new and efficient pharmaceuticals is a constant struggle for medicinal chemists. New substances are needed in order to treat different pathologies affecting the health of humans and animals, and these new compounds should be safe, effective and have the fewest side effects possible. Some functional groups are known for having biological activity; in this matter, the nitro group (NO2) is an efficient scaffold when synthesizing new bioactive molecules. Nitro compounds display a wide spectrum of activities that include antineoplastic, antibiotic, antihypertensive, antiparasitic, tranquilizers and even herbicides, among others.
- antimicrobial
- Antibacterial
- Antitubercular
- Antifungal
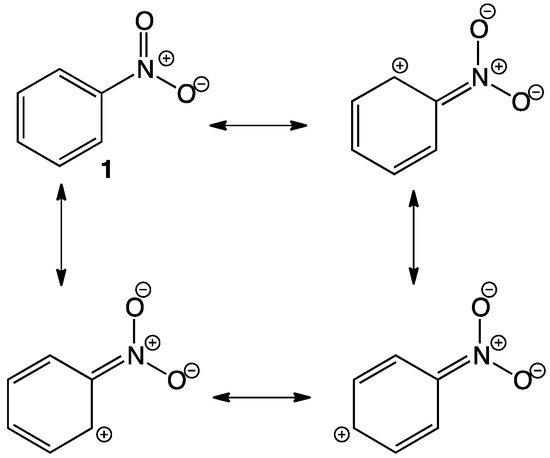
- Nitroaromatic compounds
1. Introduction
2. Antimicrobial Activity
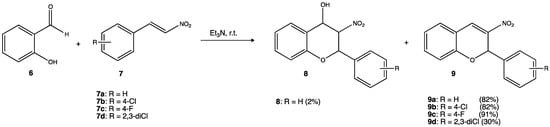
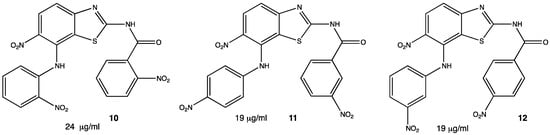
2.1. Antibacterial
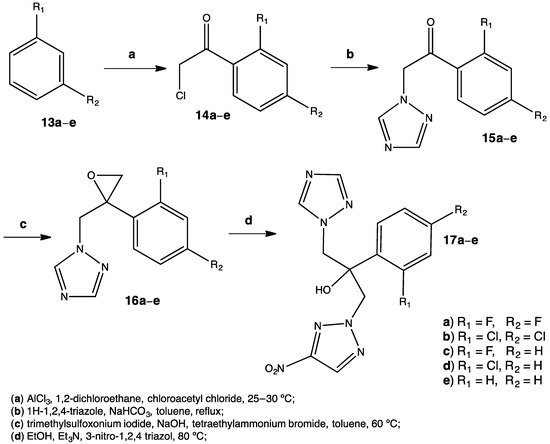
2.2. Antifungal
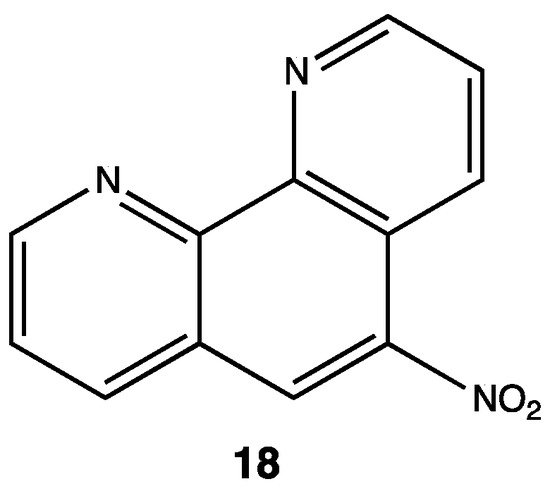
2.3. Antitubercular
This entry is adapted from the peer-reviewed paper 10.3390/ph15060717
References
- Kamila Morawska; Katarzyna Jedlińska; Sylwia Smarzewska; Radovan Metelka; Witold Ciesielski; Dariusz Guziejewski; Analysis and DNA interaction of the profluralin herbicide. Environmental Chemistry Letters 2019, 17, 1359-1365, 10.1007/s10311-019-00865-1.
- Sunil Ghatge; Youri Yang; Seonyun Moon; Woo- Young Song; Tae-Young Kim; Kwang- Hyeon Liu; Hor-Gil Hur; A novel pathway for initial biotransformation of dinitroaniline herbicide butralin from a newly isolated bacterium Sphingopyxis sp. strain HMH. Journal of Hazardous Materials 2020, 402, 123510, 10.1016/j.jhazmat.2020.123510.
- Rosario Tavera-Hernández; Manuel Jiménez-Estrada; Jesús J. Alvarado-Sansininea; Antonio Nieto-Camacho; Hugo López-Muñoz; Luis Sánchez-Sánchez; María L. Escobar; Synthesis of Chrysin, Quercetin and Naringin Nitroderivatives: Antiproliferative, Anti-inflammatory and Antioxidant Activity. Letters in Drug Design & Discovery 2021, 18, 795-805, 10.2174/1570180818666210122162313.
- Talita A. Ribeiro; Erik Machado-Ferreira; Lohaine F. Guimarães; Jéssica Cavaleiro; Alan Messala A. Britto; Nátaly Redua; Lucas Miguel Pereira de Souza; André S. Pimentel; Paulo H.S. Picciani; Osvaldo N. Oliveira; et al. Novel cytotoxic amphiphilic nitro-compounds derived from a synthetic route for paraconic acids. Colloids and Surfaces A: Physicochemical and Engineering Aspects 2021, 626, 126984, 10.1016/j.colsurfa.2021.126984.
- Aduragbenro D.A. Adedapo; Abayomi M. Ajayi; Nancy Losie Ekwunife; Olufunke O. Falayi; Ademola Oyagbemi; Temidayo Olutayo Omobowale; Adeolu A. Adedapo; Antihypertensive effect of Phragmanthera incana (Schum) Balle on NG-nitro-L-Arginine methyl ester (L-NAME) induced hypertensive rats. Journal of Ethnopharmacology 2020, 257, 112888, 10.1016/j.jep.2020.112888.
- Allison Rice; Yueming Long; S. King; Nitroaromatic Antibiotics as Nitrogen Oxide Sources. Biomolecules 2021, 11, 267, 10.3390/biom11020267.
- Patricia M. Toro; Alejandra Acuña; Mario Mallea; Michel Lapier; Mauricio Moncada; Jonathan Cisterna; Iván Brito; Hugo Klahn; Condensation and substitution products obtained in reactions of isomeric bromo-nitrofuraldehydes with ferrocenylamine: Electrochemistry and anti-parasitic evaluation. Journal of Organometallic Chemistry 2019, 901, 120946, 10.1016/j.jorganchem.2019.120946.
- Anna Jezuita; Krzysztof Ejsmont; Halina Szatylowicz; Substituent effects of nitro group in cyclic compounds. Structural Chemistry 2020, 32, 179-203, 10.1007/s11224-020-01612-x.
- Kunal Nepali; Hsueh-Yun Lee; Jing-Ping Liou; Nitro-Group-Containing Drugs. Journal of Medicinal Chemistry 2018, 62, 2851-2893, 10.1021/acs.jmedchem.8b00147.
- J. Squella; S. Bollo; L. Nunez-Vergara; Recent Developments in the Electrochemistry of Some Nitro Compounds of Biological Significance. Current Organic Chemistry 2005, 9, 565-581, 10.2174/1385272053544380.
- Dorota Olender; Justyna Żwawiak; Lucjusz Zaprutko; Multidirectional Efficacy of Biologically Active Nitro Compounds Included in Medicines. Pharmaceuticals 2018, 11, 54, 10.3390/ph11020054.
- Caroline G. Sanz; Kevin A. Dias; Raphael P. Bacil; Ricardo A.M. Serafim; Leandro H. Andrade; Elizabeth I. Ferreira; Silvia H.P. Serrano; Electrochemical characterization of para- and meta-nitro substituents in aqueous media of new antichagasic pharmaceutical leaders. Electrochimica Acta 2020, 368, 137582, 10.1016/j.electacta.2020.137582.
- Alexandre A. Oliveira; Ana P. A. Oliveira; Lucas L. Franco; Micael O. Ferencs; João F. G. Ferreira; Sofia M. P. S. Bachi; Nivaldo L. Speziali; Luiz M. Farias; Paula P. Magalhães; Heloisa Beraldo; et al. 5-Nitroimidazole-derived Schiff bases and their copper(II) complexes exhibit potent antimicrobial activity against pathogenic anaerobic bacteria. BioMetals 2018, 31, 571-584, 10.1007/s10534-018-0106-6.
- Jadriane A. Xavier; Thaissa L. Silva; Eduardo Caio Torres-Santos; Camila Calado de Vasconcelos; Anastacio Boane; Ricardo Alexandre dos Santos; Andre Felippe A. Xavier; Marília O.F. Goulart; Unveiling the relevance of the redox character of nitroaromatic and nitroheteroaromatic compounds as potential medicines. Current Opinion in Electrochemistry 2021, 29, 100740, 10.1016/j.coelec.2021.100740.
- Rodolfo Rodrigo Florido França; Cheyene Almeida Celestino Menozzi; Frederico Silva Castelo-Branco; Lucas Villas Bôas Hoelz; Nubia Boechat; The Medicinal Chemistry of 3-nitro-1,2,4-triazoles: Focus on Infectious Diseases. Current Topics in Medicinal Chemistry 2021, 21, 2072-2100, 10.2174/1568026621999210902124524.
- Kuldeep Chauhan; Moni Sharma; Priyanka Trivedi; Vinita Chaturvedi; Prem M.S. Chauhan; New class of methyl tetrazole based hybrid of (Z)-5-benzylidene-2-(piperazin-1-yl)thiazol-4(%H)-one as potent antitubercular agents. Bioorganic & Medicinal Chemistry Letters 2014, 24, 4166-4170, 10.1016/j.bmcl.2014.07.061.
- Rajan Sharma; Jacques Joubert; Recent Developments in Drug Design of NO-donor Hybrid Compounds. Mini-Reviews in Medicinal Chemistry 2018, 18, 1175-1198, 10.2174/1389557518666180416150005.
- Hammerich, O. Reduction of Nitro Compounds and Related Substrates. In Organic Electrochemistry: Revised and Expanded, 5th ed.; CRC Press: Boca Raton, FL, USA, 2015; pp. 1149–1200.
- Nagaraju Kerru; Lalitha Gummidi; Suresh Maddila; Kranthi Kumar Gangu; Sreekantha B. Jonnalagadda; A Review on Recent Advances in Nitrogen-Containing Molecules and Their Biological Applications. Molecules 2020, 25, 1909, 10.3390/molecules25081909.
- Joachim Müller; Andrew Hemphill; Norbert Müller; Physiological aspects of nitro drug resistance in Giardia lamblia. International Journal for Parasitology: Drugs and Drug Resistance 2018, 8, 271-277, 10.1016/j.ijpddr.2018.04.008.
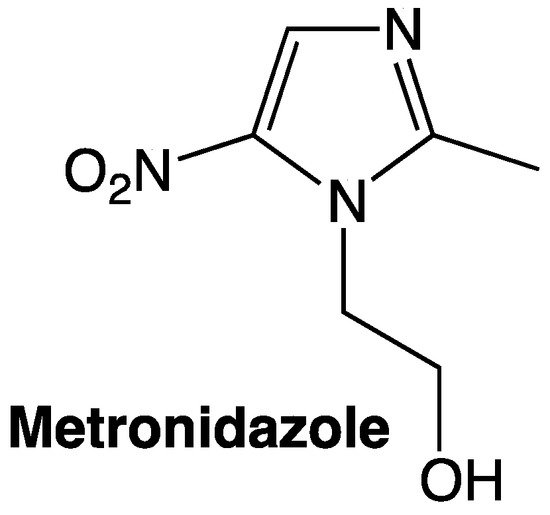
- Uzma Salar; Khalid Mohammed Khan; Muhammad Taha; Nor Hadiani Ismail; Basharat Ali; Qurat- Ul Ain; Shahnaz Perveen; Mehreen Ghufran; Abdul Wadood; Biology-oriented drug synthesis (BIODS): In vitro β-glucuronidase inhibitory and in silico studies on 2-(2-methyl-5-nitro-1H-imidazol-1-yl)ethyl aryl carboxylate derivatives. European Journal of Medicinal Chemistry 2016, 125, 1289-1299, 10.1016/j.ejmech.2016.11.031.
- C. Dhivya; S. Anbu Anjugam Vandarkuzhali; N. Radha; Antimicrobial activities of nanostructured polyanilines doped with aromatic nitro compounds. Arabian Journal of Chemistry 2019, 12, 3785-3798, 10.1016/j.arabjc.2015.12.005.
- Elham Ghobadi; Zahra Ghanbarimasir; Saeed Emami; A review on the structures and biological activities of anti-Helicobacter pylori agents. European Journal of Medicinal Chemistry 2021, 223, 113669, 10.1016/j.ejmech.2021.113669.
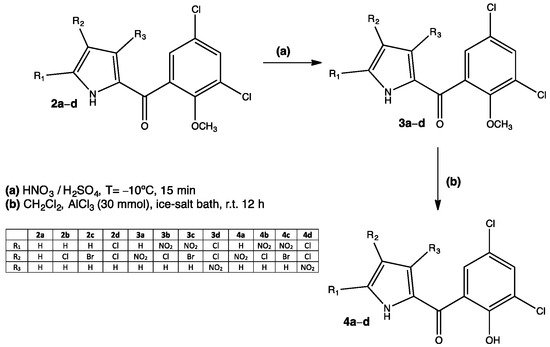
- Maria Valeria Raimondi; Alessandro Presentato; Giovanna Li Petri; Miriam Buttacavoli; Agnese Ribaudo; Viviana De Caro; Rosa Alduina; Patrizia Cancemi; New Synthetic Nitro-Pyrrolomycins as Promising Antibacterial and Anticancer Agents. Antibiotics 2020, 9, 292, 10.3390/antibiotics9060292.
- Katherine Valderrama; Elizabeth Pradel; Alexander M. Firsov; Hervé Drobecq; Hélène Bauderlique-Le Roy; Baptiste Villemagne; Yuri N. Antonenko; Ruben Christiaan Hartkoorn; Pyrrolomycins Are Potent Natural Protonophores. Antimicrobial Agents and Chemotherapy 2019, 63, e01450-19, 10.1128/aac.01450-19.
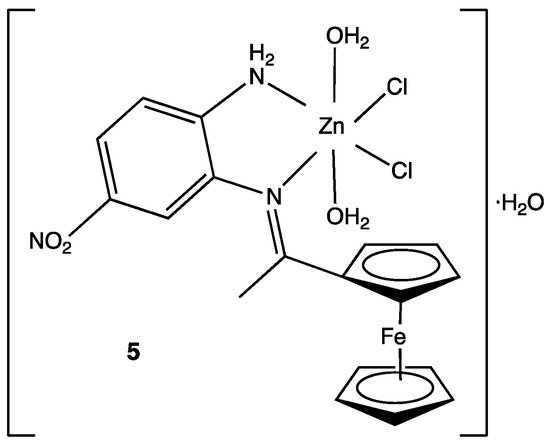
- Walaa H. Mahmoud; Reem G. Deghadi; Gehad G. Mohamed; Metal complexes of novel Schiff base derived from iron sandwiched organometallic and 4-nitro-1,2-phenylenediamine: Synthesis, characterization, DFT studies, antimicrobial activities and molecular docking. Applied Organometallic Chemistry 2018, 32, e4289, 10.1002/aoc.4289.
- Jéssica Tauany Andrade; Silmara Lucia Grego Alves; William Gustavo Lima; Carla Daiane Ferreira Sousa; Lucas Fernandes Carmo; Nívea Pereira De Sá; Fernanda Barbara Morais; Susana Johann; José Augusto Ferreira Perez Villar; Jaqueline Maria Siqueira Ferreira; et al. Pharmacologic potential of new nitro-compounds as antimicrobial agents against nosocomial pathogens: design, synthesis, and in vitro effectiveness. Folia Microbiologica 2019, 65, 393-405, 10.1007/s12223-019-00747-7.
- Akhilesh Gupta; Synthesis of Novel Nitro Substituted Benzothiazole Derivatives and Antibacterial activity against Pseudomonas aeruginosa. Research Journal of Pharmacy and Technology 2018, 12, 4663, 10.5958/0974-360x.2019.00803.5.
- Hossein Sadeghpour; Soghra Khabnadideh; Kamiar Zomorodian; Keyvan Pakshir; Khadijeh Hoseinpour; Nabiollah Javid; Ehsan Faghih-Mirzaei; Zahra Rezaei; Design, Synthesis, and Biological Activity of New Triazole and Nitro-Triazole Derivatives as Antifungal Agents. Molecules 2017, 22, 1150, 10.3390/molecules22071150.
- Jakub Suchodolski; Jakub Muraszko; Przemysław Bernat; Anna Krasowska; Lactate Like Fluconazole Reduces Ergosterol Content in the Plasma Membrane and Synergistically Kills Candida albicans. International Journal of Molecular Sciences 2021, 22, 5219, 10.3390/ijms22105219.
- Saqib Kidwai; Chan-Yong Park; Shradha Mawatwal; Prabhakar Tiwari; Myung Geun Jung; Tannu Priya Gosain; Pradeep Kumar; David Alland; Sandeep Kumar; Avinash Bajaj; et al. Dual Mechanism of Action of 5-Nitro-1,10-Phenanthroline against Mycobacterium tuberculosis. Antimicrobial Agents and Chemotherapy 2017, 61, e00969-17, 10.1128/aac.00969-17.
- Lia D'ambrosio; Rosella Centis; Giovanni Sotgiu; Emanuele Pontali; Antonio Spanevello; Giovanni Battista Migliori; New anti-tuberculosis drugs and regimens: 2015 update. ERJ Open Research 2015, 1, 00010-2015, 10.1183/23120541.00010-2015.
- Maria V. Papadopoulou; William D. Bloomer; Howard S. Rosenzweig; Alexander Arena; Francisco Arrieta; Joseph C. J. Rebolledo; Diane K. Smith; Nitrotriazole- and Imidazole-Based Amides and Sulfonamides as Antitubercular Agents. Antimicrobial Agents and Chemotherapy 2014, 58, 6828-6836, 10.1128/aac.03644-14.
- Maria V. Papadopoulou; William D. Bloomer; Howard S. Rosenzweig; The antitubercular activity of various nitro(triazole/imidazole)-based compounds. Bioorganic & Medicinal Chemistry 2017, 25, 6039-6048, 10.1016/j.bmc.2017.09.037.
