Your browser does not fully support modern features. Please upgrade for a smoother experience.
Please note this is an old version of this entry, which may differ significantly from the current revision.
Subjects:
Others
Researchers are increasingly paying attention to sustainable methods for storing energy. Many researchers are now concentrating their efforts on the development and exploration of novel materials for use in energy storage devices due to the limited supply of existing energy sources such as oil, coal, and natural gas.
- Chalcogenides
- electrochemical
- energy storage
- Batteries
- Supercapacitors
1. Batteries
An electrochemical battery energy storage solution is required for our sustainable future. For decades, rechargeable batteries have been transforming the battery industry. These rechargeable batteries, including Li-ion, Pb–acid, Ni metal-hydride, and Ni-Cd batteries, dominate the global market. Lithium-ion batteries are currently the norm in the battery industry. Li-ion batteries are made up of four major components: a cathode, an anode, an electrolyte, and a separator. This is depicted in Figure 1.
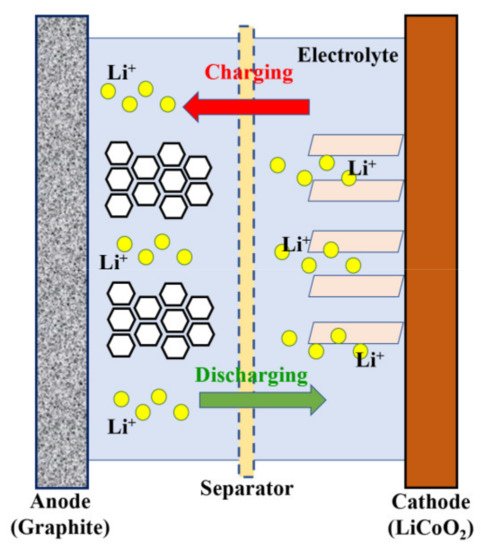
Figure 1. Schematic of the structure and working mechanism of Li-ion batteries.
While graphite is often used for the anode electrode, lithium metal oxides or phosphates, such as LiCoO2 and LiFePO4, are commonly used for the cathode electrode. In order to maintain charge balance, electrons migrate from the anode to the cathode during discharge, which implies Li-ions flow through the electrolyte and cross over the separator. On the other hand, the charging process is triggered by a charging power source, which causes the Li ions to migrate in the opposite direction [1]. As previously mentioned, one of the most important components of electrochemical Li-ion batteries is the electrode materials, which significantly impact battery performance. Therefore, it is important to review the role of chalcogenides in the synthesis and design of appropriate electrode material for high-performance Li-ion batteries. A closely related lithium element is sodium (group 1 element). Hence sodium-ion batteries have been investigated as an alternative sustainable energy storage solution. Because Na-ion has a larger atomic radius compared to Li-ion, they are expected to migrate at a slower rate [2]. However, reports demonstrated that the migration barrier could be shortened for a layered structured system [3][4][5].
Figure 2 illustrates the structure of Na-ion batteries showing the materials for the four major components: a cathode, an anode, an electrolyte, and binders. Investigating the potential of Na-ion batteries as an alternative electrochemical energy storage device is critical in the development of sustainable, innovative storage systems.
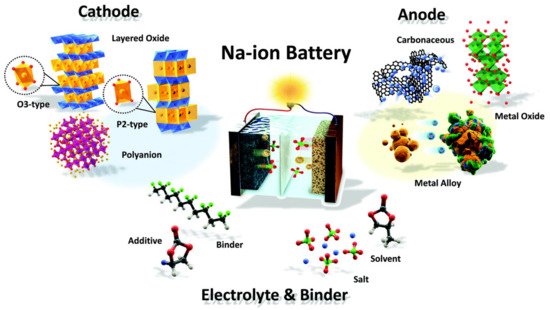
Figure 2. Schematic of the structure and illustration of a Na-ion battery system. Adapted with permission [6]. Copyright (2017) Royal Society of Chemistry. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
While investigating the electrochemical performance and environmental friendliness of several new electrode materials, most Na-ion batteries and, to a large extent, Li-ion batteries focused on transition metal sulfides-based materials such as cobalt sulfides, and iron sulfides, nickel sulfides, and tin sulfides. The storage of Na-ions in metal sulfide materials depends on the transition metal elements that are present in the metal sulfide materials. Developing new electrode materials with specific structures necessitates a thorough understanding of Na-ion transport properties in various electrode materials. The importance of understanding the role of chalcogenides in the synthesis and design of acceptable electrode materials for high-performance Na-ion batteries cannot be overstated.
As with so many other battery energy storage solutions for our sustainable future, there is a continuing desire for new technologies with higher storage capacities and fewer demands on critical minerals. Metal–Sulfur batteries have recently received significant attention [7]. In theory, lithium-sulfur batteries, for example, could store five times as much energy as lithium-ion batteries. Similar to other electrochemical batteries, lithium-sulfur batteries are made of two electrodes (lithium anode and sulfur cathode), a separator, and an electrolyte (either solid or liquid). The fundamental charging and discharging process is depicted in Figure 3, and it is governed by the equation S+2Li→Li2S. From Figure 3, lithium metal is oxidized to Li+ at the anode, and sulfur is reduced to lithium sulfide (Li2S) at the cathode during the discharge process. Lithium-sulfur batteries have several drawbacks, the most significant of which are their short cycle life, low utilization of active materials, early failure due to electrical shorting, and anode rechargeability. Another considerable drawback that has received more attention is the suppression of the polysulfide shuttle in lithium-sulfur batteries. According to a study, the effects of polysulfide shuttle can be mediated by including polysulfide absorbent and mediators as part of the cathode material [8]. Several strategies have been used to suppress polysulfide shuttle effects, including the use of electrolyte additives at the anode, the use of porous layers to impede diffusion, the use of a selective electrolyte separator, and the encapsulation of polysulfides at the cathode [9][10].
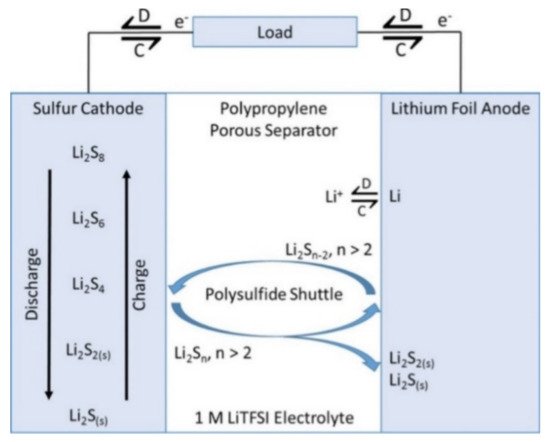
Figure 3. Schematic diagram of a Li–S cell structure with charge/discharge operations. Adapted with permission [9]. Copyright (2017) The Electrochemical Society. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
The ideal material for electrochemical energy storage must exhibit superior electrochemical properties. The electrochemical performance of electrode materials is mostly determined by the composition and structure/morphology of the materials. There has been a lot of interest in electrocatalysts of chalcogenides because of their intrinsic electrochemical activities [11][12]. Transition metal dichalcogenide (molybdenum disulfide) was utilized as a catalyst in a study to convert polysulfides into a sulfur cathode because of its inherent sulfur deficiencies. Nanocomposites of MoS2−x nanoflakes were created on thin films of reduced graphene oxide utilizing co-dispersion and filtration processes. The steps in the preparation of MoS2−x/rGO are depicted in Figure 4. Surface sulfur deficiency at the cathode had a favourable effect on the kinetics of polysulfide shuttling. As a result, the nanocomposite sulfur cathode with only a small amount of MoS2−x/rGO (4%) exhibited a high-rate capability and a long cycle life. This emphasizes the importance of designing cathode material composition and structure/morphology to improve the electrochemical performance of emerging technologies [13].
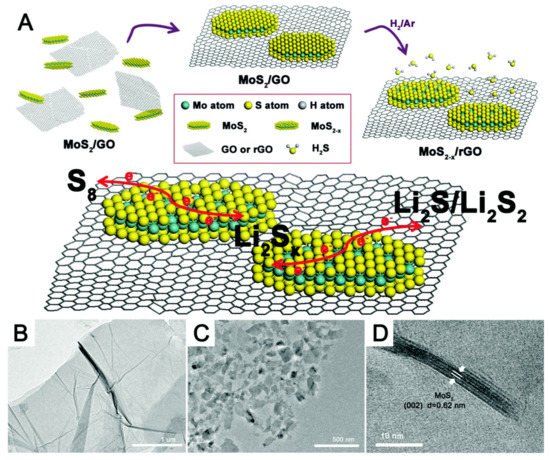
Figure 4. (A) Schematic of the synthesis of the MoS2−x/rGO composite and the conversion of Li2Sx on the MoS2−x/rGO surface. TEM images of (B) a thin GO film and (C) MoS2 nanoflakes. (D) HRTEM image of MoS2 nanoflakes. Adapted with permission [13]. Copyright (2017) The Royal Society of chemistry. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Metal–Air batteries have also demonstrated enormous potential in the field of electrochemical energy storage systems. A conventional metal-air battery consists of a metal electrode, an air-breathing cathode, and a lithium salt-containing electrolyte. The anode of metal-air batteries can be made using low-cost, environmentally friendly elements, such as Fe, Na, Al, and Zn. Metal–Air batteries are both safer and offer comparable energy density. Figure 5 shows the theoretical energy density and specific energy of commonly published metal-air batteries.
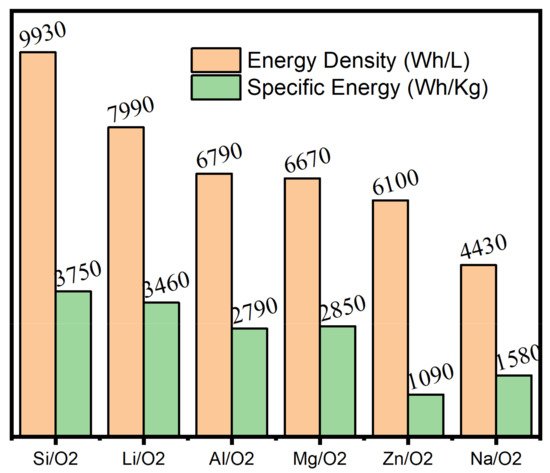
Figure 5. Theoretical energy density and specific energy (including oxygen) of commonly researched metal-air batteries. Adapted from [14]. Copyright (2021) MDPI. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
In a conventional metal-air battery structure (Figure 6), metal is converted to ions at the anode, while oxygen is transformed into hydroxide ions at the cathode. The utilization of air (ambient oxygen) has the extra benefit of lowering costs and simplifying cell construction. Metal–Air batteries such as Li-air battery [15][16], Na–air battery [17][18], Al–air battery [19][20], Mg–air battery [21][22], Zn–air battery [23][24], Fe–air battery [25][26][27]. Sn–air battery has widely been published, and the electrochemical performance and prospects of improved specific power and specific energy are well established. Indeed, the prospects of using metal chalcogenides as a possible anodic material offer a green and sustainable solution. In this vain, Lyu et al. [28] used a hybrid CoS2/rGO cathode catalyst to fabricate Li–O2 batteries. The electrodes exhibited high-rate performance. Furthermore, cobalt sulfide nanoparticles coated on graphene nanosheets have been shown to produce Zn–air batteries with outstanding cycling stability and high rechargeability [29].
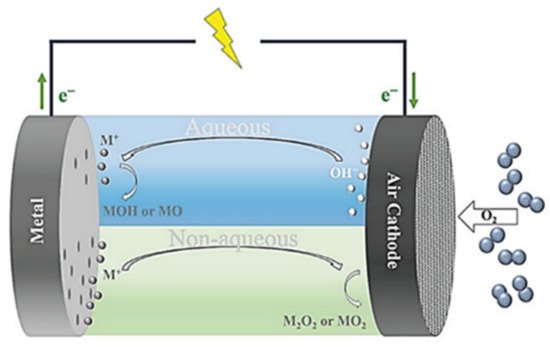
Figure 6. Schematic representation of metal-air batteries with aqueous (upper, blue) and nonaqueous (below, green) electrolytes. Adapted from [30]. Copyright (2021) MDPI. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
2. Supercapacitors
Electrochemical energy storage systems such as supercapacitors offer tremendous opportunities for clean energy storage. They comparatively offer high power density, long cycle life, and fast charging. It is expected that next-generation storage devices will be dominated by electrochemical storage systems. The development of electrochemical energy storage systems with superior energy requires novel electrode materials, and these electrode materials are critical to the electrochemical performance of the storage device. In this section, we will explore supercapacitors that are based on chalcogenides electrodes. A manufactured supercapacitor can be classified as symmetric or asymmetric based on the configuration of the electrodes. The anodic and cathodic electrode materials in symmetric supercapacitors are identical, whereas the anodic and cathodic electrode materials in asymmetric supercapacitors are distinct. Asymmetric supercapacitors have a significantly greater potential range. This has made it more appealing. When it comes to supercapacitors, the electrode is the most critical component. For a supercapacitor to have improved electrochemical performance, the materials that are utilized to construct the electrodes must be of high performance. Scientists are attempting to develop low-cost electrode materials that have a high specific surface area, good electronic conductivity, and are stable.
The electrochemical activity of TMCs significantly influences the effectiveness of electrodes used for supercapacitor electrodes. The electrochemical activity of the electrode material regulates electrochemical performance, such as energy density and power density. Many factors influence supercapacitor electrochemical performance, including nanostructure orientation and bond mechanisms. Even though several innovative materials (for example, metal oxides) have demonstrated considerable potential as supercapacitor electrodes, several of them have also demonstrated significant limitations [31][32].
Therefore, it is essential to investigate novel electrode materials that exhibit superior electrochemical performance. A possible approach in this regard is the employment of chalcogenides, which operate based on a pseudocapacitive (fast reversible redox mechanism) or hybrid (combination of charge accumulation and fast reversible redox mechanism) charge storage mechanism to store and release electrochemical energy. Chalcogenides, especially metal sulfides, metal selenides, metal tellurides, etc., have been widely applied in supercapacitor applications due to their excellent electrochemical activity. Despite this, there are a few drawbacks to using chalcogenides as an electrode material, including low conductivity, poor cyclability, a low surface area, and aggregation problems [33][34][35]. Several synthesis approaches have been used to create electrodes with high surface area by utilizing nanostructured features such as nanowires, nanorods, and nanobelts. However, to attain improved electronic conductivity and stability, the use of different active materials must be investigated.
In a reported attempt to improve the electrochemical activity by improving the surface area, chalcogenides based supercapacitors, ternary metal chalcogenides hybrid supercapacitors (MX; M = Zn–Co–Ni; X = S, Se, Te) were fabricated [36]. On nickel-based substrates, a well-organized nanostructured needlelike feature that looks similar to sea urchins was fabricated. The sample had a lot of voids and exhibited improved specific capacitance and stability. By using selenization, tellurization, and sulfurization of precursors, the morphology of the sample could be altered to affect the specific capacitance. Some chalcogenides-based electrode material exhibits inherently high electronic conductivity [37][38]. For example, MoS2 has a lower electronic conductivity compared to WS2; to tackle this, researchers have employed different synthesis methods to fabricate various nanostructured electrodes for supercapacitor applications.
Furthermore, other nanocomposites have been investigated to improve electronic conductivity [39][40][41][42]. Teli et al. produced an interconnected crumpled-nanoflake structured MoS2 on Ni foam using a one-step electrodeposition method [37]. The fabricated electrodes exhibited diffusion-controlled or capacitive-controlled charge storage kinetics and excellent coulombic efficiency. When employed as an electrode material for supercapacitor applications, WS2/α-NiMoO4 composite electrodes with nano/micro cavity-like hierarchical architecture exhibited high specific areas with increased pore size, superior electrical conductivity, and improved stability [43]. Cobalt sulfide (CoS) has generated much research attention because they are low-cost and possess a weak Co–S bond that exhibits theoretically high capacitance. As a result of these properties, transition metal sulfides for use in supercapacitor applications have been developed. For example, Zhang’s research group fabricated CoS nanowires on layered Ti3C2Tx using the solvothermal method [43]. With the Ti3C2Tx layer providing large interlayer spacing, improved conductivity, and chemical diversity on the surface, the samples exhibited remarkably improved electrochemical performance. In another research, 2D hierarchical MoS2 lamella inserted in the CoS2 flake was fabricated. The 2D MoS2 multilayers provided support preventing interlayer expansion and structural collapse. The electrochemical performance and cyclic stability of these chalcogenides can be improved with secondary support [44]. Other typical chalcogenides [45][46][47][48][49][50][51][52][53] also demonstrated great potential for supercapacitor applications.
This entry is adapted from the peer-reviewed paper 10.3390/en15114052
References
- Zhang, H.; Li, X.; Liu, W.; Yue, H.; Shi, Z.; Yin, Y.; Yang, S. Olivine LiFePO4 as an additive into LiCoO2 electrodes for LIBs to improve high-voltage performances. J. Alloys Compd. 2021, 869, 159188.
- Abraham, K.M. Intercalation positive electrodes for rechargeable sodium cells. Solid State Ionics 1982, 7, 199–212.
- Rajabathar, J.R.; Al-lohedan, H.A.; Arunachalam, P.; Issa, Z.A.; Gnanamani, M.K.; Appaturi, J.N.; Ibrahim, S.N.; Mohammed Dahan, W. Unexpected discovery of low-cost maricite NaFePO4 as a high-performance electrode for Na-ion batteries. J. Alloys Compd. 2021, 850, 540–545.
- Rajabathar, J.R.; Al-lohedan, H.A.; Arunachalam, P.; Issa, Z.A.; Gnanamani, M.K.; Appaturi, J.N.; Ibrahim, S.N.; Mohammed Dahan, W. Challenges for Na-ion negative electrodes. J. Alloys Compd. 2021, 850, A1011.
- Rajabathar, J.R.; Al-lohedan, H.A.; Arunachalam, P.; Issa, Z.A.; Gnanamani, M.K.; Appaturi, J.N.; Ibrahim, S.N.; Mohammed Dahan, W. Sodium-Ion Batteries. J. Alloys Compd. 2021, 850, 947–958.
- Hwang, J.-Y.; Myung, S.-T.; Sun, Y.-K. Sodium-ion batteries: Present and future. Chem. Soc. Rev. 2017, 46, 3529–3614.
- Nagde, K.R.; Dhoble, S.J. Li-S ion batteries: A substitute for Li-ion storage batteries. In Energy Materials; Elsevier: Amsterdam, The Netherlands, 2021; pp. 335–371.
- Evers, S.; Yim, T.; Nazar, L.F. Understanding the nature of absorption/adsorption in nanoporous polysulfide sorbents for the Li–S battery. J. Phys. Chem. C 2012, 116, 19653–19658.
- Moy, D.; Narayanan, S.R. Mixed Conduction Membranes Suppress the Polysulfide Shuttle in Lithium-Sulfur Batteries. J. Electrochem. Soc. 2017, 164, A560–A566.
- Moy, D.; Manivannan, A.; Narayanan, S.R. Direct Measurement of Polysulfide Shuttle Current: A Window into Understanding the Performance of Lithium-Sulfur Cells. J. Electrochem. Soc. 2014, 162, A1–A7.
- Li, H.; Tsai, C.; Koh, A.L.; Cai, L.; Contryman, A.W.; Fragapane, A.H.; Zhao, J.; Han, H.S.; Manoharan, H.C.; Abild-Pedersen, F.; et al. Activating and optimizing MoS2 basal planes for hydrogen evolution through the formation of strained sulphur vacancies. Nat. Mater. 2016, 15, 48–53.
- Ji, G.; Yu, Y.; Yao, Q.; Qu, B.; Chen, D.; Chen, W.; Xie, J.; Lee, J.Y. Promotion of reversible Li+ storage in transition metal dichalcogenides by Ag nanoclusters. NPG Asia Mater. 2016, 8, e247.
- Lin, H.; Yang, L.; Jiang, X.; Li, G.; Zhang, T.; Yao, Q.; Zheng, G.W.; Lee, J.Y. Electrocatalysis of polysulfide conversion by sulfur-deficient MoS2 nanoflakes for lithium–sulfur batteries. Energy Environ. Sci. 2017, 10, 1476–1486.
- Clark, S.; Latz, A.; Horstmann, B. A Review of Model-Based Design Tools for Metal-Air Batteries. Batteries 2018, 4, 5.
- Zou, X.; Lu, Q.; Liao, K.; Shao, Z. Towards practically accessible aprotic Li-air batteries: Progress and challenges related to oxygen-permeable membranes and cathodes. Energy Storage Mater. 2022, 45, 869–902.
- Lu, S.-H.; Lu, H.-C. Pouch-type hybrid Li-air battery enabled by flexible composite lithium-ion conducting membrane. J. Power Source 2021, 489, 229431.
- Parveen, N.; Khan, Z.; Ansari, S.A.; Park, S.; Senthilkumar, S.T.; Kim, Y.; Ko, H.; Cho, M.H. Feasibility of using hollow double walled Mn2O3 nanocubes for hybrid Na-air battery. Chem. Eng. J. 2019, 360, 415–422.
- Wang, H.; Xu, Q. Materials Design for Rechargeable Metal-Air Batteries. Matter 2019, 1, 565–595.
- Hou, X.; Zhang, Y.; Cui, C.; Lin, C.; Li, Y.; Bu, D.; Yan, G.; Liu, D.; Wu, Q.; Song, X.-M. Photo-assisted Al-air batteries based on gel-state electrolyte. J. Power Source 2022, 533, 231377.
- Wu, P.; Zhao, Q.; Yu, H.; Tang, Z.; Li, Y.; Huang, D.; Sun, D.; Wang, H.; Tang, Y. Modification on water electrochemical environment for durable Al-Air Battery: Achieved by a Low-Cost sucrose additive. Chem. Eng. J. 2022, 438, 135538.
- Zhang, L.; Shao, Q.; Zhang, J. An overview of non-noble metal electrocatalysts and their associated air cathodes for Mg-air batteries. Mater. Rep. Energy 2021, 1, 100002.
- Vaghefinazari, B.; Snihirova, D.; Wang, C.; Wang, L.; Deng, M.; Höche, D.; Lamaka, S.V.; Zheludkevich, M.L. Exploring the effect of sodium salt of Ethylenediaminetetraacetic acid as an electrolyte additive on electrochemical behavior of a commercially pure Mg in primary Mg-air batteries. J. Power Source 2022, 527, 231176.
- Leong, K.W.; Wang, Y.; Ni, M.; Pan, W.; Luo, S.; Leung, D.Y.C. Rechargeable Zn-air batteries: Recent trends and future perspectives. Renew. Sustain. Energy Rev. 2022, 154, 111771.
- Qian, M.; Guo, M.; Qu, Y.; Xu, M.; Liu, D.; Hou, C.; Isimjan, T.T.; Yang, X. Energy barrier engineering of oxygen reduction reaction synergistically promoted by binary Zn-Cu pair sites for advanced Zn–air batteries. J. Alloys Compd. 2022, 907, 164527.
- Hang, B.T.; Watanabe, T.; Egashira, M.; Watanabe, I.; Okada, S.; Yamaki, J. The effect of additives on the electrochemical properties of Fe/C composite for Fe/air battery anode. J. Power Source 2006, 155, 461–469.
- Hang, B.T.; Hayashi, H.; Yoon, S.-H.; Okada, S.; Yamaki, J. Fe2O3-filled carbon nanotubes as a negative electrode for an Fe–air battery. J. Power Source 2008, 178, 393–401.
- Inoishi, A.; Ida, S.; Uratani, S.; Okano, T.; Ishihara, T. High capacity of an Fe–air rechargeable battery using LaGaO3-based oxide ion conductor as an electrolyte. Phys. Chem. Chem. Phys. 2012, 14, 12818–12822.
- Lyu, Z.; Zhou, Y.; Dai, W.; Cui, X.; Lai, M.; Wang, L.; Huo, F.; Huang, W.; Hu, Z.; Chen, W. Recent advances in understanding of the mechanism and control of Li 2 O 2 formation in aprotic Li–O2 batteries. Chem. Soc. Rev. 2017, 46, 6046–6072.
- Geng, D.; Ding, N.-N.; Hor, T.S.A.; Chien, S.W.; Liu, Z.; Zong, Y. Cobalt sulfide nanoparticles impregnated nitrogen and sulfur co-doped graphene as bifunctional catalyst for rechargeable Zn–air batteries. RSC Adv. 2015, 5, 7280–7284.
- Olabi, A.G.; Sayed, E.T.; Wilberforce, T.; Jamal, A.; Alami, A.H.; Elsaid, K.; Rahman, S.M.; Shah, S.K.; Abdelkareem, M.A. Metal-Air Batteries—A Review. Energies 2021, 14, 7373.
- Bao, S.-J.; Li, C.M.; Guo, C.-X.; Qiao, Y. Biomolecule-assisted synthesis of cobalt sulfide nanowires for application in supercapacitors. J. Power Source 2008, 180, 676–681.
- Subramanian, A.; Punnoose, D.; Raman, V.; Gopi, C.V.V.M.; Rao, S.S.; Khan, M.A.; Kim, H.-J. Layer by layer approach to enhance capacitance using metal sulfides for supercapacitor applications. Mater. Lett. 2018, 231, 64–67.
- Sajedi-Moghaddam, A.; Saievar-Iranizad, E.; Pumera, M. Two-dimensional transition metal dichalcogenide/conducting polymer composites: Synthesis and applications. Nanoscale 2017, 9, 8052–8065.
- Kim, Y.; Park, T.; Na, J.; Yi, J.W.; Kim, J.; Kim, M.; Bando, Y.; Yamauchi, Y.; Lin, J. Layered transition metal dichalcogenide/carbon nanocomposites for electrochemical energy storage and conversion applications. Nanoscale 2020, 12, 8608–8625.
- Cherusseri, J.; Choudhary, N.; Sambath Kumar, K.; Jung, Y.; Thomas, J. Recent trends in transition metal dichalcogenide based supercapacitor electrodes. Nanoscale Horiz. 2019, 4, 840–858.
- Ahmad, M.; Hussain, I.; Nawaz, T.; Li, Y.; Chen, X.; Ali, S.; Imran, M.; Ma, X.; Zhang, K. Comparative study of ternary metal chalcogenides (MX.; M= Zn–Co–Ni; X= S, Se, Te): Formation process, charge storage mechanism and hybrid supercapacitor. J. Power Source 2022, 534, 231414.
- Teli, A.M.; Beknalkar, S.A.; Mane, S.M.; Bhat, T.S.; Kambale, B.B.; Patil, S.B.; Sadale, S.B.; Shin, J.C. Electrodeposited crumpled MoS2 nanoflakes for asymmetric supercapacitor. Ceram. Int. 2022, in press.
- Sharma, G.K.; Ranjan, B.; Kaur, D. Electrochemical kinetics of 2D-MoS2 sputtered over stainless-steel mesh: Insights into the Na+ ions storage for flexible supercapacitors. Ceram. Int. 2022, in press.
- Gao, Y.-P.; Huang, K.-J.; Wu, X.; Hou, Z.-Q.; Liu, Y.-Y. MoS2 nanosheets assembling three-dimensional nanospheres for enhanced-performance supercapacitor. J. Alloys Compd. 2018, 741, 174–181.
- Thanh, T.D.; Chuong, N.D.; Van Hien, H.; Kshetri, T.; Kim, N.H.; Lee, J.H. Recent advances in two-dimensional transition metal dichalcogenides-graphene heterostructured materials for electrochemical applications. Prog. Mater. Sci. 2018, 96, 51–85.
- Yang, X.; Zhao, L.; Lian, J. Arrays of hierarchical nickel sulfides/MoS2 nanosheets supported on carbon nanotubes backbone as advanced anode materials for asymmetric supercapacitor. J. Power Source 2017, 343, 373–382.
- Sun, T.; Li, Z.; Liu, X.; Ma, L.; Wang, J.; Yang, S. Facile construction of 3D graphene/MoS2 composites as advanced electrode materials for supercapacitors. J. Power Source 2016, 331, 180–188.
- Ray, S.K.; Pant, B.; Park, M.; Hur, J.; Lee, S.W. Cavity-like hierarchical architecture of WS2/α-NiMoO4 electrodes for supercapacitor application. Ceram. Int. 2020, 46, 19022–19027.
- Li, Y.; Wang, H.; Shu, T.; Yuan, J.; Lu, G.; Lin, B.; Gao, Z.; Wei, F.; Ma, C.; Qi, J.; et al. Two-dimensional hierarchical MoS2 lamella inserted in CoS2 flake as an advanced supercapacitor electrode. J. Energy Storage 2022, 51, 104299.
- Bhol, P.; Swain, S.; Altaee, A.; Saxena, M.; Samal, A.K. Cobalt–iron decorated tellurium nanotubes for high energy density supercapacitor. Mater. Today Chem. 2022, 24, 100871.
- Rathore, H.K.; Hariram, M.; Ganesha, M.K.; Singh, A.K.; Das, D.; Kumar, M.; Awasthi, K.; Sarkar, D. Charge storage mechanism in vanadium telluride/carbon nanobelts as electroactive material in an aqueous asymmetric supercapacitor. J. Colloid Interface Sci. 2022, 621, 110–118.
- Theerthagiri, J.; Karuppasamy, K.; Durai, G.; Rana, A.U.H.S.; Arunachalam, P.; Sangeetha, K.; Kuppusami, P.; Kim, H.-S. Recent Advances in Metal Chalcogenides (MX; X = S, Se) Nanostructures for Electrochemical Supercapacitor Applications: A Brief Review. Nanomaterials 2018, 8, 256.
- Miao, C.; Xia, G.; Zhu, K.; Ye, K.; Wang, Q.; Yan, J.; Cao, D.; Gong, F.; Wang, G. Enhanced supercapacitor performance of bimetallic metal selenides via controllable synergistic engineering of composition. Electrochim. Acta 2021, 370, 137802.
- Lei, H.; Zhou, J.; Zhao, R.; Peng, H.; Xu, Y.; Wang, F.; Hamouda, H.A.; Zhang, W.; Ma, G. Design and assembly of a novel asymmetric supercapacitor based on all-metal selenides electrodes. Electrochim. Acta 2020, 363, 137206.
- Amiri, M.; Saeed Hosseiny Davarani, S.; Ebrahim Moosavifard, S.; Fu, Y.-Q. Cobalt-molybdenum selenide double-shelled hollow nanocages derived from metal-organic frameworks as high performance electrodes for hybrid supercapacitor. J. Colloid Interface Sci. 2022, 616, 141–151.
- Liu, Q.; Hong, X.; You, X.; Zhang, X.; Zhao, X.; Chen, X.; Ye, M.; Liu, X. Designing heterostructured metal sulfide core-shell nanoneedle films as battery-type electrodes for hybrid supercapacitors. Energy Storage Mater. 2020, 24, 541–549.
- Wang, Q.; Qu, Z.; Chen, S.; Zhang, D. Metal organic framework derived P-doping with sulfide defect to boost high-performance asymmetric supercapacitors. J. Colloid Interface Sci. 2022, in press.
- Lu, L.; Xu, Q.; Chen, Y.; Zhou, Y.; Jiang, T.; Zhao, Q. Preparation of metal sulfide electrode materials derived based on metal organic framework and application of supercapacitors. J. Energy Storage 2022, 49, 104073.
This entry is offline, you can click here to edit this entry!
