Lactadherin is a secreted glycoprotein associated with the milk fat globule membrane, which is highly present in the blood and in the mammary tissue of lactating women. Several biological functions have been associated with this protein, mainly attributable to its immunomodulatory role promoting phagocyte-mediated clearance of apoptotic cells. It has been shown that lactadherin also plays important roles in cell adhesion, promotion of angiogenesis, and tissue regeneration. On the other hand, this protein has been used as a marker of breast cancer and tumor progression. Recently, high levels of lactadherin has been associated with poor prognosis and decreased survival, not only in breast cancer, but also in melanoma, ovarian, colorectal, and other types of cancer. Although the mechanisms responsible for the tumor-promoting effects attributed to lactadherin have not been fully elucidated, a growing body of literature indicates that lactadherin could be a promising therapeutic target and/or biomarker for breast and other tumors. Moreover, recent studies have shown its presence in extracellular vesicles derived from cancer cell lines and cancer patients, which was associated with cancer aggressiveness and worse prognosis.
- lactadherin
- breast cancer
- metastasis
- exosomes
- extracellular vesicles
1. Introduction
Lactadherin: A Milk Fat Globule Protein Associated with Cancer Development
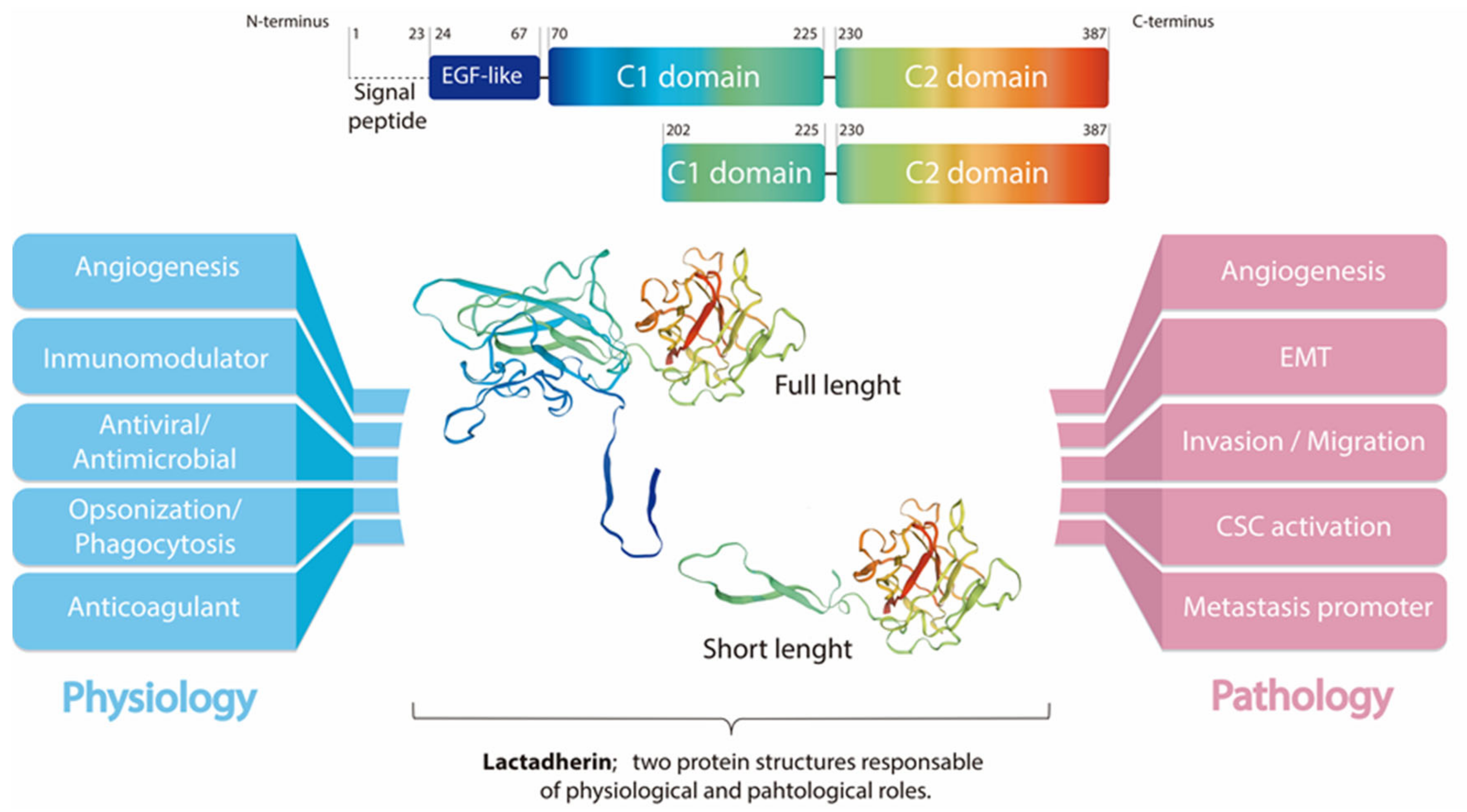
2. Lactadherin as a Biomarker of Tumor Subtype, Progression and Metastasis
3. Lactadherin Role as Promotor of Tumor Progression
3.1. Tumor Cell Survival/Proliferation and EMT
3.2. Lactadherin Immunomodulatory Role
4. Lactadherin as a Possible New Cancer Therapeutic Target
5. Extracellular Vesicles and Exosomes as Promotors of Breast Cancer Metastasis
6. Role and Use of Lactadherin in EVs and Exosomes
7. Conclusions
This entry is adapted from the peer-reviewed paper 10.3390/ijms23073855
References
- Sabha, B.H.; Alzahrani, F.; Almehdar, H.A.; Uversky, V.N.; Redwan, E.M. Disorder in milk proteins: Lactadherin multifunctionality and structure. Curr. Protein Pept. Sci. 2018, 19, 983–997.
- Taylor, M.R.; Couto, J.R.; Scallan, C.D.; Ceriani, R.L.; Peterson, J.A. Lactadherin (Formerly BA46), a Membrane-Associated Glycoprotein Expressed in Human Milk and Breast Carcinomas, Promotes Arg-Gly-Asp (RGD)-Dependent Cell Adhesion. DNA Cell Biol. 1997, 16, 861–869.
- Larocca, D.; Peterson, J.A.; Urrea, R.; Kuniyoshi, J.; Bistrain, A.M.; Ceriani, R.L. A Mr 46,000 human milk fat globule protein that is highly expressed in human breast tumors contains Factor VHI-like domains. Cancer Res. 1991, 51, 4994.
- Couto, J.R.; Taylor, M.R.; Godwin, S.G.; Ceriani, R.L.; Peterson, J.A. Cloning and Sequence Analysis of Human Breast Epithelial Antigen BA46 Reveals an RGD Cell Adhesion Sequence Presented on an Epidermal Growth Factor-Like Domain. DNA Cell Biol. 1996, 15, 281–286.
- Andersen, M.H.; Graversen, H.; Fedosov, S.N.; Petersen, T.E.; Rasmussen, J.T. Functional analyses of two cellular binding domains of bovine lactadherin. Biochemistry 2000, 39, 6200–6206.
- Giuffrida, M.G.; Cavaletto, M.; Giunta, C.; Conti, A.; Godovac-Zimmermann, J. Isolation and characterization of full and truncated forms of human breast carcinoma protein BA46 from human milk fat globule membranes. J. Protein Chem. 1998, 17, 143–148.
- Hanayama, R.; Tanaka, M.; Miwa, K.; Shinohara, A.; Iwamatsu, A.; Nagata, S. Identification of a factor that links apoptotic cells to phagocytes. Nature 2002, 417, 182–187.
- Newburg, D.S.; Peterson, J.A.; Ruiz-Palacios, G.M.; Matson, D.O.; Morrow, A.L.; Shults, J.; Guerrero, M.L.; Chaturvedi, P.; Newburg, S.O.; Scallan, C.D.; et al. Role of human-milk lactadherin in protection against symptomatic rotavirus infection. Lancet 1998, 351, 1160–1164.
- Kvistgaard, A.; Pallesen, L.; Arias, C.; Lopez, S.; Petersen, T.; Heegaard, C.; Rasmussen, J.T. Inhibitory Effects of Human and Bovine Milk Constituents on Rotavirus Infections. J. Dairy Sci. 2004, 87, 4088–4096.
- Jinushi, M.; Chiba, S.; Yoshiyama, H.; Masutomi, K.; Kinoshita, I.; Dosaka-Akita, H.; Yagita, H.; Takaoka, A.; Tahara, H. Tumor-associated macrophages regulate tumorigenicity and anticancer drug responses of cancer stem/initiating cells. Proc. Natl. Acad. Sci. USA 2011, 108, 12425–12430.
- Silvestre, J.-S.; Théry, C.; Hamard, G.; Boddaert, J.; Aguilar, B.; Delcayre, A.; Houbron, C.; Tamarat, R.; Blanc-Brude, O.; Heeneman, S.; et al. Lactadherin promotes VEGF-dependent neovascularization. Nat. Med. 2005, 11, 499–506.
- Zhou, Y.; Bond, A.M.; Shade, J.E.; Zhu, Y.; Davis, C.-H.O.; Wang, X.; Su, Y.; Yoon, K.-J.; Phan, A.T.; Chen, W.J.; et al. Autocrine Mfge8 Signaling Prevents Developmental Exhaustion of the Adult Neural Stem Cell Pool. Cell Stem Cell 2018, 23, 444–452.e4.
- Kamińska, A.; Enguita, F.J.; Stępień, E. Lactadherin: An unappreciated haemostasis regulator and potential therapeutic agent. Vasc. Pharmacol. 2018, 101, 21–28.
- Uchiyama, A.; Yamada, K.; Perera, B.; Ogino, S.; Yokoyama, Y.; Takeuchi, Y.; Ishikawa, O.; Motegi, S.-I. Protective Effect of MFG-E8 after Cutaneous Ischemia–Reperfusion Injury. J. Investig. Dermatol. 2015, 135, 1157–1165.
- Motegi, S.-I.; Leitner, W.; Lu, M.; Tada, Y.; Sárdy, M.; Wu, C.; Chavakis, T.; Udey, M.C. Pericyte-Derived MFG-E8 Regulates Pathologic Angiogenesis. Arter. Thromb. Vasc. Biol. 2011, 31, 2024–2034.
- Motegi, S.; Ishikawa, O. Mesenchymal stem cells: The roles and functions in cutaneous wound healing and tumor growth. J. Dermatol. Sci. 2017, 86, 83–89.
- Nakashima, Y.; Miyagi-Shiohira, C.; Noguchi, H.; Omasa, T. The Healing Effect of Human Milk Fat Globule-EGF Factor 8 Protein (MFG-E8) in A Rat Model of Parkinson’s Disease. Brain Sci. 2018, 8, 167.
- Bocca, S.; Anderson, S.; Amaker, B.; Swanson, R.; Franchi, A.; Lattanzio, F.; Oehninger, S. Milk fat globule epidermal growth factor 8 (MFG-E8): A novel protein in the mammalian endometrium with putative roles in implantation and placentation. Placenta 2012, 33, 795–802.
- Arklie, J.; Taytor-Papadimitrou, J.; Bodmer, W.; Egar, M.; Millis, R. Differentiation antigens expressed by epithelial cells in the lactating breast are also detectable in breast cancer. Int. J. Cancer 1981, 28, 23–29.
- Ceriani, R.L.; Peterson, J.A.; Blank, E.W. Breast Cancer Diagnosis with Human Mammary Epithelial Antigens and the Prospective Use of Antibodies against Them in Therapy. In Mechanisms of Cancer Metastasis. Developments in Oncology; Honn, K.V., Powers, W.E., Sloane, B.F., Eds.; Elsevier: Amsterdam, The Netherlands, 1986; pp. 235–257.
- UCSC Xena Open Access. Available online: https://xena.ucsc.edu (accessed on 13 May 2020).
- Human Protein Atlas. (Shows Expression of Lactadherin mRNA and Protein in All Female Reproductive Organs). Available online: https://www.proteinatlas.org/ENSG00000140545-MFGE8/tissue (accessed on 13 May 2020).
- Taylor-Papadimitriou, J.; Peterson, J.A.; Arklie, J.; Burchell, J.; Ceriani, R.L.; Bodmer, W.F. Monoclonal antibodies to epithelial-specific components of the human milk fat globule membrane: Production and reaction with cells in culture. Int. J. Cancer 1981, 28, 17–28.
- Sasaki, M.; Peterson, J.A.; Ceriani, R.L. Monoclonal antibodies against breast epithelial cell surface antigens in breast cancer therapy. Hybridoma 1983, 2, 120.
- Peterson, J.A.; Zava, D.T.; Duwe, A.K.; Blank, E.W.; Battifora, H.; Ceriani, R.L. Biochemical and histological characterization of antigens preferentially expressed on the surface and cytoplasm of breast carcinoma cells identified by monoclonal antibodies against the human milk fat globule. Hybridoma 1990, 9, 221–235.
- Ceriani, R.L.; Sasaki, M.; Sussman, H.; Wara, W.M.; Blank, E.W. Circulating human mammary epithelial antigens in breast cancer. Proc. Natl. Acad. Sci. USA 1982, 79, 5420–5424.
- Neutzner, M.; Lopez, T.; Feng, X.; Bergmann-Leitner, E.S.; Leitner, W.W.; Udey, M.C. MFG-E8/lactadherin promotes tumor growth in an angiogenesis-dependent transgenic mouse model of multistage carcinogenesis. Cancer Res. 2007, 67, 6777–6785.
- Sugano, G.; Bernard-Pierrot, I.; Laé, M.; Battail, C.; Allory, Y.; Stransky, N.; Krumeich, S.; Lepage, M.-L.; Maille, P.; Donnadieu, M.-H.; et al. Milk fat globule–epidermal growth factor–factor VIII (MFGE8)/lactadherin promotes bladder tumor development. Oncogene 2010, 30, 642–653.
- Na, L.; Dai, C.; Yang, Y.; Wu, X.; Wang, L.; Wang, P. The expression levels and clinical significance of MFG-E8 and CD133 in epithelial ovarian cancer. Gynecol. Endocrinol. 2020, 36, 803–807.
- Okamoto, A.; Sakakura, K.; Takahashi, H.; Motegi, S.I.; Kaira, K.; Yokobori-Kuwabara, Y.; Ishikawa, O.; Chikamatsu, K. Immunological and clinicopathological significance of MFG-E8 expression in patients with oral squamous cell carcinoma. Pathol. Oncol. Res. 2019, 26, 1263–1268.
- Kanemura, T.; Miyata, H.; Makino, T.; Tanaka, K.; Sugimura, K.; Hamada-Uematsu, M.; Mizote, Y.; Uchida, H.; Miyazaki, Y.; Takahashi, T.; et al. Immunoregulatory influence of abundant MFG -E8 expression by esophageal cancer treated with chemotherapy. Cancer Sci. 2018, 109, 3393–3402.
- Fujiwara, C.; Motegi, S.-I.; Ohira, A.; Yamaguchi, S.; Sekiguchi, A.; Yasuda, M.; Nakamura, H.; Makiguchi, T.; Yokoo, S.; Hoshina, D.; et al. The significance of tumor cells-derived MFG-E8 in tumor growth of angiosarcoma. J. Dermatol. Sci. 2019, 96, 18–25.
- Yang, W.; Lai, Z.; Li, Y.; Mu, J.; Yang, M.; Xie, J.; Xu, J. Immune signature profiling identified prognostic factors for gastric cancer. Chin. J. Cancer Res. 2019, 31, 463–470.
- Carrascosa, C.; Obula, R.G.; Missiaglia, E.; Lehr, H.A.; Delorenzi, M.; Frattini, M.; Rüegg, C.; Mariotti, A. MFG-E8/lactadherin regulates cyclins D1/D3 expression and enhances the tumorigenic potential of mammary epithelial cells. Oncogene 2012, 31, 1521–1532.
- Yang, C.; Hayashida, T.; Forster, N.; Li, C.; Shen, D.; Maheswaran, S.; Chen, L.; Anderson, K.S.; Ellisen, L.W.; Sgroi, D.; et al. The integrin alpha(v)beta(3–5) ligand MFG-E8 is a p63/p73 target gene in triple-negative breast cancers but exhibits suppressive functions in ER(+) and erbB2(+) breast cancers. Cancer Res. 2011, 71, 937–945.
- Yu, L.; Zhao, L.; Jia, Z.; Bi, J.; Wei, Q.; Song, X.; Jiang, L.; Lin, S.; Wei, M. MFG-E8 overexpression is associated with poor prognosis in breast cancer patients. Pathol. Res. Pr. 2018, 215, 490–498.
- Wang, M.-Y.; Huang, M.; Wang, C.-Y.; Tang, X.-Y.; Wang, J.-G.; Yang, Y.-D.; Xiong, X.; Gao, C.-W. Transcriptome Analysis Reveals MFGE8-HAPLN3 Fusion as a Novel Biomarker in Triple-Negative Breast Cancer. Front. Oncol. 2021, 11, 682021.
- Tibaldi, L.; Leyman, S.; Nicolas, A.; Notebaert, S.; Dewulf, M.; Ngo, T.H.; Zuany-Amorim, C.; Amzallag, N.; Bernard-Pierrot, I.; Sastre-Garau, X.; et al. New Blocking Antibodies Impede Adhesion, Migration and Survival of Ovarian Cancer Cells, Highlighting MFGE8 as a Potential Therapeutic Target of Human Ovarian Carcinoma. PLoS ONE 2013, 8, e72708.
- Jinushi, M.; Nakazaki, Y.; Carrasco, D.R.; Draganov, D.; Souders, N.; Johnson, M.; Mihm, M.C.; Dranoff, G. Milk Fat Globule EGF-8 Promotes Melanoma Progression through Coordinated Akt and Twist Signaling in the Tumor Microenvironment. Cancer Res. 2008, 68, 8889–8898.
- Oba, J.; Moroi, Y.; Nakahara, T.; Abe, T.; Hagihara, A.; Furue, M. Expression of milk fat globule epidermal growth factor-VIII may be an indicator of poor prognosis in malignant melanoma. Br. J. Dermatol. 2011, 165, 506–512.
- Yamada, K.; Uchiyama, A.; Uehara, A.; Perera, B.; Ogino, S.; Yokoyama, Y.; Takeuchi, Y.; Udey, M.C.; Ishikawa, O.; Motegi, S.-I. MFG-E8 Drives Melanoma Growth by Stimulating Mesenchymal Stromal Cell–Induced Angiogenesis and M2 Polarization of Tumor-Associated Macrophages. Cancer Res. 2016, 76, 4283–4292.
- Zhao, Q.; Xu, L.; Sun, X.; Zhang, K.; Shen, H.; Tian, Y.; Sun, F.; Li, Y. MFG-E8 overexpression promotes colorectal cancer progression via AKT/MMPs signalling. Tumor Biol. 2017, 39, 1010428317707881.
- Yang, Y.; Li, J.; Song, Q.; Zhu, K.; Yu, X.; Tian, Y.; Zhang, J. Reduction in milk fat globule-EGF factor 8 inhibits triple-negative breast cancer cell viability and migration. Oncol. Lett. 2019, 17, 3457–3465.
- Xu, X.; Zhang, A.; Zhu, Y.; He, W.; Di, W.; Fang, Y.; Shi, X. MFG-E8 reverses microglial-induced neurotoxic astrocyte (A1) via NF-κB and PI3K-Akt pathways. J. Cell. Physiol. 2018, 234, 904–914.
- Li, H.; Xu, W.; Ma, Y.; Zhou, S.; Xiao, R. Milk fat globule membrane protein promotes C2C12 cell proliferation through the PI3K/Akt signaling pathway. Int. J. Biol. Macromol. 2018, 114, 1305–1314.
- Gao, Y.-Y.; Zhang, Z.-H.; Zhuang, Z.; Lu, Y.; Wu, L.-Y.; Ye, Z.N.; Zhang, X.-S.; Chen, C.-L.; Li, W.; Hang, C.-H. Recombinant milk fat globule-EGF factor-8 reduces apoptosis via integrin β3/FAK/PI3K/AKT signaling pathway in rats after traumatic brain injury. Cell Death Dis. 2018, 9, 845.
- Wu, J.; Yang, H.; Cheng, J.; Zhang, L.; Ke, Y.; Zhu, Y.; Wang, C.; Zhang, X.; Zhen, X.; Zheng, L.T. Knockdown of milk-fat globule EGF factor-8 suppresses glioma progression in GL261 glioma cells by repressing microglial M2 polarization. J. Cell. Physiol. 2019, 235, 8679–8690.
- Zhuowang, G.; Chen, Y.; Wang, B.; Zhang, X.; Yan, Y.; Zhou, L.; Zhang, Y.; Xie, Y. MFGE8 attenuates Ang-II-induced atrial fibrosis and vulnerability to atrial fibrillation through inhibition of TGF-β1/Smad2/3 pathway. J. Mol. Cell. Cardiol. 2020, 139, 164–175.
- Matsuda, A.; Jacob, A.; Wu, R.; Zhou, M.; Aziz, M.; Wang, P. Milk fat globule–EGF factor VIII ameliorates liver injury after hepatic ischemia-reperfusion. J. Surg. Res. 2012, 180, e37–e46.
- Deng, K.-Q.; Li, J.; She, Z.-G.; Gong, J.; Cheng, W.-L.; Gong, F.-H.; Zhu, X.-Y.; Zhang, Y.; Wang, Z.; Li, H. Restoration of Circulating MFGE8 (Milk Fat Globule-EGF Factor 8) Attenuates Cardiac Hypertrophy Through Inhibition of Akt Pathway. Hypertension 2017, 70, 770–779.
- Ceriani, R.L.; Blank, E.W.; Peterson, J.A. Experimental immunotherapy of human breast carcinomas implanted in nude mice with a mixture of monoclonal antibodies against human milk fat globule components. Cancer Res. 1987, 47, 532–540.
- Kotari, C.; Osseni, M.A.; Agbo, L.; Ouellette, G.; Déraspe, M.; Laviolette, F.; Corbeil, J.; Lambert, J.-P.; Diorio, C.; Durocher, F. Machine learning analysis identifies genes differentiating triple negative breast cancers. Sci. Rep. 2020, 10, 10464.
- Tian, J.; Wang, V.; Wang, N.; Khadang, B.; Boudreault, J.; Bakdounes, K.; Ali, S.; Lebrun, J.-J. Identification of MFGE8 and KLK5/7 as mediators of breast tumorigenesis and resistance to COX-2 inhibition. Breast Cancer Res. 2021, 23, 23.
- Thery, C.; Zitvogel, L.; Amigorena, S. Exosomes: Composition, biogenesis and function. Nat. Rev. Immunol. 2002, 2, 569–579.
- Andaloussi, S.; Mäger, I.; Breakefield, X.O.; Wood, M.J. Extracellular vesicles: Biology and emerging therapeutic opportunities. Nat. Rev. Drug Discov. 2013, 12, 347–357.
- Kowal, J.; Arras, G.; Colombo, M.; Jouve, M.; Morath, J.P.; Primdal-Bengtson, B.; Dingli, F.; Loew, D.; Tkach, M.; Théry, C. Proteomic comparison defines novel markers to characterize heterogeneous populations of extracellular vesicle subtypes. Proc. Natl. Acad. Sci. USA 2016, 113, E968–E977.
- Mathieu, M.; Martin-Jaular, L.; Lavieu, G.; Théry, C. Specificities of secretion and uptake of exosomes and other extracellular vesicles for cell-to-cell communication. Nat. Cell Biol. 2019, 21, 9–17.
- Lee, K.W.; Cho, J.A.; Park, H.; Lim, E.H. Exosomes from breast cancer cells can convert adipose tissue-derived mesenchymal stem cells into myofibroblast-like cells. Int. J. Oncol. 2011, 40, 130–138.
- Gorczynski, R.M.; Erin, N.; Zhu, F. Serum-derived exosomes from mice with highly metastatic breast cancer transfer increased metastatic capacity to a poorly metastatic tumor. Cancer Med. 2016, 5, 325–336.
- Harris, D.A.; Patel, S.H.; Gucek, M.; Hendrix, A.; Westbroek, W.; Taraska, J.W. Exosomes Released from Breast Cancer Carcinomas Stimulate Cell Movement. PLoS ONE 2015, 10, e0117495.
- Feng, Q.; Zhang, C.; Lum, D.; Druso, J.E.; Blank, B.; Wilson, K.F.; Welm, A.; Antonyak, M.A.; Cerione, R.A. A class of extracellular vesicles from breast cancer cells activates VEGF receptors and tumour angiogenesis. Nat. Commun. 2017, 8, 14450.
- Hoshino, A.; Costa-Silva, B.; Shen, T.-L.; Rodrigues, G.; Hashimoto, A.; Mark, M.T.; Molina, H.; Kohsaka, S.; Di Giannatale, A.; Ceder, S.; et al. Tumour exosome integrins determine organotropic metastasis. Nature 2015, 527, 329–335.
- Galindo-Hernandez, O.; Serna-Marquez, N.; Castillo-Sanchez, R.; Salazar, E.P. Extracellular vesicles from MDA-MB-231 breast cancer cells stimulated with linoleic acid promote an EMT-like process in MCF10A cells. Prostaglandins Leukot. Essent. Fat. Acids 2014, 91, 299–310.
- Luga, V.; Zhang, L.; Viloria-Petit, A.M.; Ogunjimi, A.A.; Inanlou, M.R.; Chiu, E.; Buchanan, M.; Hosein, A.N.; Basik, M.; Wrana, J.L. Exosomes Mediate Stromal Mobilization of Autocrine Wnt-PCP Signaling in Breast Cancer Cell Migration. Cell 2012, 151, 1542–1556.
- Novikova, S.; Shushkova, N.; Farafonova, T.; Tikhonova, O.; Kamyshinsky, R.; Zgoda, V. Proteomic Approach for Searching for Universal, Tissue-Specific, and Line-Specific Markers of Extracellular Vesicles in Lung and Colorectal Adenocarcinoma Cell Lines. Int. J. Mol. Sci. 2020, 21, 6601.
- Pathan, M.; Fonseka, P.; Chitti, S.V.; Kang, T.; Sanwlani, R.; Van Deun, J.; Hendrix, A.; Mathivanan, S. Vesiclepedia 2019: A compendium of RNA, proteins, lipids and metabolites in extracellular vesicles. Nucleic Acids Res. 2019, 47, D516–D519.
- Véron, P.; Segura, E.; Sugano, G.; Amigorena, S.; Théry, C. Accumulation of MFG-E8/lactadherin on exosomes from immature dendritic cells. Blood Cells Mol. Dis. 2005, 35, 81–88.
- Kenari, A.N.; Cheng, L.; Hill, A.F. Methods for loading therapeutics into extracellular vesicles and generating extracellular vesicles mimetic-nanovesicles. Methods 2020, 177, 103–113.
- Miyasaka, K.; Hanayama, R.; Tanaka, M.; Nagata, S. Expression of milk fat globule epidermal growth factor?8 in immature dendritic cells for engulfment of apoptotic cells. Eur. J. Immunol. 2004, 34, 1414–1422.
- Zeelenberg, I.S.; Ostrowski, M.; Krumeich, S.; Bobrie, A.; Jancic, C.; Boissonnas, A.; Delcayre, A.; Le Pecq, J.-B.; Combadière, B.; Amigorena, S.; et al. Targeting Tumor Antigens to Secreted Membrane Vesicles In vivo Induces Efficient Antitumor Immune Responses. Cancer Res. 2008, 68, 1228–1235.
- Komuro, H.; Kawai-Harada, Y.; Aminova, S.; Pascual, N.; Malik, A.; Contag, C.H.; Harada, M. Engineering Extracellular Vesicles to Target Pancreatic Tissue In Vivo. Nanotheranostics 2021, 5, 378–390.
- Wang, J.H.; Forterre, A.V.; Zhao, J.; Frimannsson, D.O.; Delcayre, A.; Antes, T.J.; Efron, B.; Jeffrey, S.S.; Pegram, M.D.; Matin, A.C. Anti-HER2 scFv-Directed Extracellular Vesicle-Mediated mRNA-Based Gene Delivery Inhibits Growth of HER2-Positive Human Breast Tumor Xenografts by Prodrug Activation. Mol. Cancer Ther. 2018, 17, 1133–1142.
- Kooijmans, S.A.A.; Gitz-Francois, J.J.J.M.; Schiffelers, R.M.; Vader, P. Recombinant phosphatidylserine-binding nanobodies for targeting of extracellular vesicles to tumor cells: A plug-and-play approach. Nanoscale 2018, 10, 2413–2426.
