Evidence has shown that gut microbiome plays a role in modulating the development of diseases beyond the gastrointestinal tract, including skin disorders such as psoriasis. The gut–skin axis refers to the bidirectional relationship between the gut microbiome and skin health. This is regulated through several mechanisms such as inflammatory mediators and the immune system. Dysregulation of microbiota has been seen in numerous inflammatory skin conditions such as atopic dermatitis, rosacea, and psoriasis. Understanding how gut microbiome are involved in regulating skin health may lead to development of novel therapies for these skin disorders through microbiome modulation, in particularly psoriasis.
- microbiota
- skin disease
- gut dysbiosis
1. Introduction
2. Psoriasis Pathogenesis and the Cutaneous Immune System
3. The Gut-Skin Axis and the Gut Microbiome
Effects of Gut Dysbiosis on Gut Microbial Metabolite and the Gut Immunoregulatory Characteristic
4. Alterations in the Alpha and Beta Diversity of Gut Microbiome in Psoriasis Patients
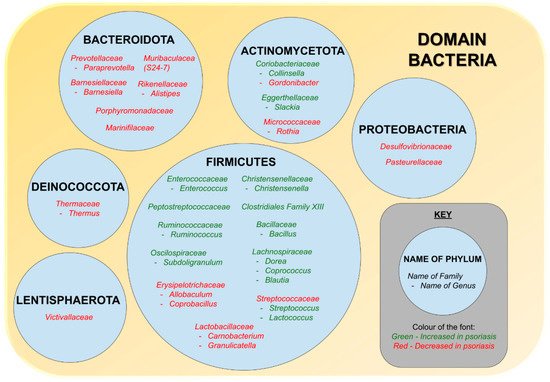
5. Alterations in the Relative Abundance of Gut Microbiome of Psoriasis Patients
6. Treatment
Benefits of Gut Microbiome Modulation on Skin Health
| Evidence of Benefits of Microbiome Modulation on General Skin Health | ||||
|---|---|---|---|---|
| Reference | Study Subjects | Probiotics Used | Duration of Intervention | Outcome of Treatment |
| [153] | Mice | Lactobacillus helveticus-fermented milk whey | 5 weeks | Reduced TEWL and areas of dermatitis |
| [156] | Mice | Lactobacillus reuteri ATCC-PTA-6475 |
12 days | Shorter wound healing time, increased Foxp3+ regulatory T cell |
| [154] | Ex vivo human abdominal skin explant model | Lactobacillus paracasei CNCM-I 2116 (ST11) | 24 h | Stronger skin barrier function, reduced neurogenic inflammatory skin diseases |
| [151] | Human | Lactobacillus paracasei NCC2461 | Two months | Reduced transepidermal water loss (TEWL) and skin sensitivity |
| Evidence of benefits of microbiome modulation on psoriasis | ||||
| Non-human studies | ||||
| [142] | Mice | Lactobacillus pentosus GMNL-77 |
6 days | Improved skin lesions, reduced Th-17 associated proinflammatory cytokines, reduced Th17 and Th22 T cells in spleen |
| [157] | Imiquimod (IMQ)-induced psoriasis in mice | Leuconostoc mesenteroides NTM048 | 21 days | Reduced erythema and scaling, increased plasma deoxycholic acid (DCA) level, reduced IL-17 production in murine spleen |
| [158] | Imiquimod (IMQ)-induced psoriasis in mice | Bifidobacterium adolescentis, Bifidobacterium breve, Bifidobacterium animalis, Lacticaseibacillus paracasei, Limosilactobacillus reuteri | 2 weeks | B. adolescentis CCFM667, B. breve CCFM1078, Lacticaseibacillus paracasei CCFM1074, and Limosilactobacillus reuteri CCFM1132 reduced psoriasis-like pathological characteristics and suppressed the release of IL-23/ Th17 related inflammatory cytokines |
| [159] | TNF-α-induced HaCaT cell hyperproliferation | 102 strains of probiotics | Not specified | Bifidobacterium animalis CCFM1148 and Lactobacillus paracasei CCFM1147 suppressed keratinocyte hyperproliferation by preventing NF-κB activation and downregulating the levels of IL-6 and IL-8 |
| Human studies | ||||
| [143] | Patients with psoriasis | Bifidobacteria infantis 35624 | 6–8 weeks | Reduced plasma C-Reactive Protein and TNF-α levels |
| [161] | Case study of a patient with pustular psoriasis | Lactobacillus sporogenes | 6 months | Pustular lesions improved in 2 weeks and lesion free by 6 months of intervention |
| [162] | 27 patients with psoriasis | Bacteroides fragillis BF839 | 12 weeks | Significant reduction in PASI score |
| [163] | 50 patients with plaque psoriasis | Multistrain probiotic including Lactobacillus acidophilus, Bifidobacterium bifidum, Bifidobacterium lactis and Bifidobacterium langum |
8 weeks | Reduced Beck’s Depression Inventory (BDI) and Dermatology Life Quality Index (DLQI) scores, reduced PASI and psoriasis symptom scale, increased Total Antioxidant Capacity (TAC), decreased CRP and IL-6 levels. |
| [164] | 64 patients with mild to moderate psoriasis | Lactocare® that contains seven strains (Lactobacillus casei, Lactobacillus acidophilus, Lactobacillus rhamnosus, Lactobacillus bulgaricus, Bifidobacterium breve, Bifidobacterium longum, Streptococcus thermophiles with prebiotic fructooligosaccharide) | 12 weeks | Increased serum levels of Fe, Zn, P, Mg, Ca, and Na in probiotic group, signifying better mineral absorption |
| [165] | 90 patients with plaque psoriasis | Bifidobacterium longum CECT 7347, B. lactis CECT 8145 and Lactobacillus rhamnosus CECT 8361 | 12 weeks | Greater reduction in PASI score, lower rate of relapse |
7. Discussion
8. Conclusions
This entry is adapted from the peer-reviewed paper 10.3390/biomedicines10051037
References
- Global Report on Psoriasis; 2016; p. 48. Available online: https://www.who.int/publications/i/item/global-report-on-psoriasis (accessed on 26 October 2016).
- Global Burden of Disease Study 2010: Results by Cause 1990–2010. Available online: https://ghdx.healthdata.org/record/ihme-data/gbd-2010-results-cause-1990-2010 (accessed on 20 July 2021).
- Barnas, J.L.; Ritchlin, C.T. Etiology and pathogenesis of psoriatic arthritis. Rheum. Dis. Clin. 2015, 41, 643–663.
- Boehncke, W.-H. Etiology and pathogenesis of psoriasis. Rheum. Dis. Clin. 2015, 41, 665–675.
- Kulig, P.; Musiol, S.; Freiberger, S.N.; Schreiner, B.; Gyülveszi, G.; Russo, G.; Pantelyushin, S.; Kishihara, K.; Alessandrini, F.; Kündig, T. IL-12 protects from psoriasiform skin inflammation. Nat. Commun. 2016, 7, 1–14.
- Roberson, E.D.; Bowcock, A.M. Psoriasis genetics: Breaking the barrier. Trends Genet. 2010, 26, 415–423.
- Sondermann, W.; Djeudeu Deudjui, D.; Körber, A.; Slomiany, U.; Brinker, T.; Erbel, R.; Moebus, S. Psoriasis, cardiovascular risk factors and metabolic disorders: Sex-specific findings of a population-based study. J. Eur. Acad. Dermatol. Venereol. 2020, 34, 779–786.
- Meglio, P.D.; Villanova, F.; Nestle, F.O. Psoriasis. Cold Spring Harb. Perspect. Med. 2014, 4, a015354.
- Gupta, R.; Debbaneh, M.G.; Liao, W. Genetic epidemiology of psoriasis. Curr. Dermatol. Rep. 2014, 3, 61–78.
- Lee, E.J.; Do Han, K.; Han, J.H.; Lee, J.H. Smoking and risk of psoriasis: A nationwide cohort study. J. Am. Acad. Dermatol. 2017, 77, 573–575.
- Jensen, P.; Skov, L. Psoriasis and obesity. Dermatology 2016, 232, 633–639.
- Barrea, L.; Nappi, F.; Di Somma, C.; Savanelli, M.C.; Falco, A.; Balato, A.; Balato, N.; Savastano, S. Environmental risk factors in psoriasis: The point of view of the nutritionist. Int. J. Environ. Res. Public Health 2016, 13, 743.
- Fry, L.; Baker, B.S. Triggering psoriasis: The role of infections and medications. Clin. Dermatol. 2007, 25, 606–615.
- Kamiya, K.; Kishimoto, M.; Sugai, J.; Komine, M.; Ohtsuki, M. Risk Factors for the Development of Psoriasis. Int. J. Mol. Sci. 2019, 20, 4347.
- Danielsen, K.; Olsen, A.; Wilsgaard, T.; Furberg, A.S. Is the prevalence of psoriasis increasing? A 30-year follow-up of a population-based cohort. Br. J. Dermatol. 2013, 168, 1303–1310.
- Parisi, R.; Symmons, D.P.; Griffiths, C.E.; Ashcroft, D.M. Global epidemiology of psoriasis: A systematic review of incidence and prevalence. J. Invest. Dermatol. 2013, 133, 377–385.
- Rachakonda, T.D.; Schupp, C.W.; Armstrong, A.W. Psoriasis prevalence among adults in the United States. J. Am. Acad. Dermatol. 2014, 70, 512–516.
- Jacobson, C.C.; Kumar, S.; Kimball, A.B. Latitude and psoriasis prevalence. J. Am. Acad. Dermatol. 2011, 65, 870–873.
- Le, S.; Toussi, A.; Maverakis, N.; Marusina, A.I.; Barton, V.R.; Merleev, A.A.; Luxardi, G.; Raychaudhuri, S.P.; Maverakis, E. The cutaneous and intestinal microbiome in psoriatic disease. Clin. Immunol. 2020, 108537.
- Szántó, M.; Dózsa, A.; Antal, D.; Szabó, K.; Kemény, L.; Bai, P. Targeting the gut-skin axis—Probiotics as new tools for skin disorder management? Exp. Dermatol. 2019, 28, 1210–1218.
- Sikora, M.; Stec, A.; Chrabaszcz, M.; Knot, A.; Waskiel-Burnat, A.; Rakowska, A.; Olszewska, M.; Rudnicka, L. Gut microbiome in psoriasis: An updated review. Pathogens 2020, 9, 463.
- Zheng, D.; Liwinski, T.; Elinav, E. Interaction between microbiota and immunity in health and disease. Cell Res. 2020, 30, 492–506.
- Toor, D.; Wsson, M.K.; Kumar, P.; Karthikeyan, G.; Kaushik, N.K.; Goel, C.; Singh, S.; Kumar, A.; Prakash, H. Dysbiosis disrupts gut immune homeostasis and promotes gastric diseases. Int. J. Mol. Sci. 2019, 20, 2432.
- Nishida, A.; Inoue, R.; Inatomi, O.; Bamba, S.; Naito, Y.; Andoh, A. Gut microbiota in the pathogenesis of inflammatory bowel disease. Clin. J. Gastroenterol. 2018, 11, 1–10.
- Korman, N. Management of psoriasis as a systemic disease: What is the evidence? Br. J. Dermatol. 2020, 182, 840–848.
- Gerdes, S.; Mrowietz, U.; Boehncke, W.-H. Komorbidität bei Psoriasis vulgaris. Hautarzt. 2016, 67, 438–444.
- Kim, H.-N.; Han, K.; Park, Y.-G.; Lee, J.H. Metabolic syndrome is associated with an increased risk of psoriasis: A nationwide population-based study. Metabolism 2019, 99, 19–24.
- Gisondi, P.; Fostini, A.C.; Fossà, I.; Girolomoni, G.; Targher, G. Psoriasis and the metabolic syndrome. Clin. Dermatol. 2018, 36, 21–28.
- Singh, S.; Young, P.; Armstrong, A.W. Relationship between psoriasis and metabolic syndrome: A systematic review. G. Ital. Dermatol. Venereol. 2016, 151, 663–677.
- Ramessur, R.; Gill, D. The effect of statins on severity of psoriasis: A systematic review. Indian J. Dermatol. Venereol. Leprol. 2017, 83.
- Ogdie, A.; Gelfand, J. Clinical risk factors for the development of psoriatic arthritis among patients with psoriasis: A review of available evidence. Curr. Rheumatol. Rep. 2015, 17, 1–7.
- Alinaghi, F.; Tekin, H.G.; Burisch, J.; Wu, J.J.; Thyssen, J.P.; Egeberg, A. Global prevalence and bidirectional association between psoriasis and inflammatory bowel disease—a systematic review and meta-analysis. J. Crohn’s Colitis 2020, 14, 351–360.
- Komine, M. Recent advances in psoriasis research; the clue to mysterious relation to gut microbiome. Int. J. Mol. Sci. 2020, 21, 2582.
- O’Neill, C.A.; Monteleone, G.; McLaughlin, J.T.; Paus, R. The gut-skin axis in health and disease: A paradigm with therapeutic implications. Bioessays 2016, 38, 1167–1176.
- Levkovich, T.; Poutahidis, T.; Smillie, C.; Varian, B.J.; Ibrahim, Y.M.; Lakritz, J.R.; Alm, E.J.; Erdman, S.E. Probiotic bacteria induce a ‘glow of health’. PLoS ONE 2013, 8, e53867.
- Ramírez-Boscá, A.; Navarro-López, V.; Martínez-Andrés, A.; Such, J.; Francés, R.; de la Parte, J.H.; Asín-Llorca, M. Identification of bacterial DNA in the peripheral blood of patients with active psoriasis. JAMA Dermatol. 2015, 151, 670–671.
- Sacchetti, L.; Nardelli, C. Gut microbiome investigation in celiac disease: From methods to its pathogenetic role. Clin. Chem. Lab. Med. 2020, 58, 340–349.
- Baba, H.; Masuyama, A.; Yoshimura, C. Promoter of Differentiation and Keratinization of Epidermic Cell and Functional Beverage/Food for Promotion of Keratinization of Epidermis. Available online: https://scienceon.kisti.re.kr/srch/selectPORSrchPatent.do?cn=USP2012018097246 (accessed on 9 July 2021).
- Abhishek, S.; Krishnan, S.P. Epidermal differentiation complex: A review on its epigenetic regulation and potential drug targets. Cell J. 2016, 18, 1.
- Hsu, D.K.; Fung, M.A.; Chen, H.-L. Role of skin and gut microbiota in the pathogenesis of psoriasis, an inflammatory skin disease. Med. Microecol. 2020, 4, 100016.
- Weaver, C.T.; Elson, C.O.; Fouser, L.A.; Kolls, J.K. The Th17 pathway and inflammatory diseases of the intestines, lungs, and skin. Annu. Rev. Pathol.: Mech. Dis. 2013, 8, 477–512.
- Gaur, M.; Dobke, M.; Lunyak, V.V. Mesenchymal stem cells from adipose tissue in clinical applications for dermatological indications and skin aging. Int. J. Mol. Sci. 2017, 18, 208.
- Kobayashi, T.; Naik, S.; Nagao, K. Choreographing immunity in the skin epithelial barrier. Immunity 2019, 50, 552–565.
- Matejuk, A. Skin immunity. Arch. Immunol. Ther. Exp. (Warsz.) 2018, 66, 45–54.
- Yazdi, A.S.; Röcken, M.; Ghoreschi, K. Cutaneous immunology: Basics and new concepts. Semin. Immunopathol. 2016, 38, 3–10.
- Liang, Y.; Sarkar, M.K.; Tsoi, L.C.; Gudjonsson, J.E. Psoriasis: A mixed autoimmune and autoinflammatory disease. Curr. Opin. Immunol. 2017, 49, 1–8.
- Blauvelt, A.; Chiricozzi, A. The immunologic role of IL-17 in psoriasis and psoriatic arthritis pathogenesis. Clin. Rev. Allergy Immunol. 2018, 55, 379–390.
- Wang, A.; Bai, Y. Dendritic cells: The driver of psoriasis. J. Dermatol 2020, 47, 104–113.
- Albanesi, C.; Madonna, S.; Gisondi, P.; Girolomoni, G. The interplay between keratinocytes and immune cells in the pathogenesis of psoriasis. Front. Immunol. 2018, 9, 1549.
- Luger, T.; Loser, K. Novel insights into the pathogenesis of psoriasis. Clin. Immunol. 2017, 186, 43–45.
- Harden, J.L.; Krueger, J.G.; Bowcock, A.M. The immunogenetics of psoriasis: A comprehensive review. J. Autoimmun. 2015, 64, 66–73.
- Dainichi, T.; Kitoh, A.; Otsuka, A.; Nakajima, S.; Nomura, T.; Kaplan, D.H.; Kabashima, K. The epithelial immune microenvironment (EIME) in atopic dermatitis and psoriasis. Nat. Immunol. 2018, 19, 1286–1298.
- Chimenti, M.S.; Perricone, C.; Novelli, L.; Caso, F.; Costa, L.; Bogdanos, D.; Conigliaro, P.; Triggianese, P.; Ciccacci, C.; Borgiani, P. Interaction between microbiome and host genetics in psoriatic arthritis. Autoimmun. Rev. 2018, 17, 276–283.
- Chandra, A.; Ray, A.; Senapati, S.; Chatterjee, R. Genetic and epigenetic basis of psoriasis pathogenesis. Mol. Immunol. 2015, 64, 313–323.
- Lowes, M.A.; Russell, C.B.; Martin, D.A.; Towne, J.E.; Krueger, J.G. The IL-23/T17 pathogenic axis in psoriasis is amplified by keratinocyte responses. Trends Immunol. 2013, 34, 174–181.
- Zeng, B.; Shi, S.; Ashworth, G.; Dong, C.; Liu, J.; Xing, F. ILC3 function as a double-edged sword in inflammatory bowel diseases. Cell Death Dis. 2019, 10, 1–12.
- Benhadou, F.; Mintoff, D.; Del Marmol, V. Psoriasis: Keratinocytes or immune cells–which is the trigger? Dermatology 2019, 235, 91–100.
- Ellis, S.R.; Nguyen, M.; Vaughn, A.R.; Notay, M.; Burney, W.A.; Sandhu, S.; Sivamani, R.K. The skin and gut microbiome and its role in common dermatologic conditions. Microorganisms 2019, 7, 550.
- Power, S.E.; O’Toole, P.W.; Stanton, C.; Ross, R.P.; Fitzgerald, G.F. Intestinal microbiota, diet and health. Br. J. Nutr. 2014, 111, 387–402.
- Ipci, K.; Altıntoprak, N.; Muluk, N.B.; Senturk, M.; Cingi, C. The possible mechanisms of the human microbiome in allergic diseases. Eur. Arch. Otorhinolaryngol. 2017, 274, 617–626.
- Mowat, A.M.; Agace, W.W. Regional specialization within the intestinal immune system. Nat. Rev. Immunol. 2014, 14, 667–685.
- Yatsunenko, T.; Rey, F.E.; Manary, M.J.; Trehan, I.; Dominguez-Bello, M.G.; Contreras, M.; Magris, M.; Hidalgo, G.; Baldassano, R.N.; Anokhin, A.P. Human gut microbiome viewed across age and geography. Nature 2012, 486, 222–227.
- Bäckhed, F.; Roswall, J.; Peng, Y.; Feng, Q.; Jia, H.; Kovatcheva-Datchary, P.; Li, Y.; Xia, Y.; Xie, H.; Zhong, H. Dynamics and stabilization of the human gut microbiome during the first year of life. Cell Host Microbe 2015, 17, 690–703.
- Li, M.; Wang, M.; Donovan, S.M. Early development of the gut microbiome and immune-mediated childhood disorders. Semin. Reprod. Med. 2014, 32, 74–86.
- Qin, J.; Li, R.; Raes, J.; Arumugam, M.; Burgdorf, K.S.; Manichanh, C.; Nielsen, T.; Pons, N.; Levenez, F.; Yamada, T. A human gut microbial gene catalogue established by metagenomic sequencing. Nature 2010, 464, 59–65.
- McKenzie, C.; Tan, J.; Macia, L.; Mackay, C.R. The nutrition-gut microbiome-physiology axis and allergic diseases. Immunol. Rev. 2017, 278, 277–295.
- Mazidi, M.; Rezaie, P.; Kengne, A.P.; Mobarhan, M.G.; Ferns, G.A. Gut microbiome and metabolic syndrome. Diabetes Metab. Syndr. Clin. Res. Rev. 2016, 10, S150–S157.
- Lin, S.; Wang, Z.; Lam, K.-L.; Zeng, S.; Tan, B.K.; Hu, J. Role of intestinal microecology in the regulation of energy metabolism by dietary polyphenols and their metabolites. Food Nutr. Res. 2019, 63.
- Spencer, S.P.; Fragiadakis, G.K.; Sonnenburg, J.L. Pursuing human-relevant gut microbiota-immune interactions. Immunity 2019, 51, 225–239.
- Chelakkot, C.; Ghim, J.; Ryu, S.H. Mechanisms regulating intestinal barrier integrity and its pathological implications. Exp. Mol. Med. 2018, 50, 1–9.
- Lee, J.K.-F.; Hern Tan, L.T.; Ramadas, A.; Ab Mutalib, N.-S.; Lee, L.-H. Exploring the Role of Gut Bacteria in Health and Disease in Preterm Neonates. Int. J. Environ. Res. Public Health 2020, 17, 6963.
- Boyle, R.J.; Lahtinen, S.J.; Tang, M.L. Probiotics and skin. In Nutrition and Skin; Springer: Berlin/Heidelberg, Germany, 2011; pp. 111–127.
- Kosiewicz, M.M.; Dryden, G.W.; Chhabra, A.; Alard, P. Relationship between gut microbiota and development of T cell associated disease. FEBS Lett. 2014, 588, 4195–4206.
- Lee, L.-H.; Wong, S.H.; Chin, S.-F.; Singh, V.; Ab Mutalib, N.-S. Editorial: Human Microbiome: Symbiosis to Pathogenesis. Front. Microbiol. 2021, 12.
- Lau, A.; Teng Hern, T.; Ab Mutalib, N.-S.; Wong, S.; Letchumanan, V.; Lee, L.H. The chemistry of gut microbiome in health and diseases. Prog. Microbes Mol. Biol. 2021, 4.
- Shen, T.-C.D. Diet and gut microbiota in health and disease. Nestle Nutr. Inst. Workshop Ser. 2017, 88, 117–126.
- Vallianou, N.; Stratigou, T.; Christodoulatos, G.S.; Dalamaga, M. Understanding the role of the gut microbiome and microbial metabolites in obesity and obesity-associated metabolic disorders: Current evidence and perspectives. Curr. Obes. Rep. 2019, 8, 317–332.
- Scher, J.U.; Littman, D.R.; Abramson, S.B. Microbiome in inflammatory arthritis and human rheumatic diseases. Arthritis Rheumatol. 2016, 68, 35.
- Stevens, B.R.; Roesch, L.; Thiago, P.; Russell, J.T.; Pepine, C.J.; Holbert, R.C.; Raizada, M.K.; Triplett, E.W. Depression phenotype identified by using single nucleotide exact amplicon sequence variants of the human gut microbiome. Mol. Psychiatry 2021, 26, 4277–4287.
- Kitai, T.; Tang, W.W. Gut microbiota in cardiovascular disease and heart failure. Clin. Sci. 2018, 132, 85–91.
- Shah, K.R.; Boland, C.R.; Patel, M.; Thrash, B.; Menter, A. Cutaneous manifestations of gastrointestinal disease: Part I. J. Am. Acad. Dermatol. 2013, 68, 189.e1–189.e21.
- Thrash, B.; Patel, M.; Shah, K.R.; Boland, C.R.; Menter, A. Cutaneous manifestations of gastrointestinal disease: Part II. J. Am. Acad. Dermatol. 2013, 68, 211.e1–211.e33.
- Gloster, H.; Gebauer, L.; Mistur, R. Cutaneous manifestations of gastrointestinal disease. In Absolute Dermatology Review; Springer: Cham, Switzerland, 2016; pp. 171–179.
- Kim, H.-J.; Lee, S.-H.; Hong, S.-J. Antibiotics-induced dysbiosis of intestinal microbiota aggravates atopic dermatitis in mice by altered short-chain fatty acids. Allergy Asthma Immunol. Res. 2020, 12, 137–148.
- Polkowska-Pruszyńska, B.; Gerkowicz, A.; Krasowska, D. The gut microbiome alterations in allergic and inflammatory skin diseases–an update. J. Eur. Acad. Dermatol. Venereol. 2020, 34, 455–464.
- Purchiaroni, F.; Tortora, A.; Gabrielli, M.; Bertucci, F.; Gigante, G.; Ianiro, G.; Ojetti, V.; Scarpellini, E.; Gasbarrini, A. The role of intestinal microbiota and the immune system. Eur. Rev. Med. Pharmacol. Sci. 2013, 17, 323–333.
- Rajilić-Stojanović, M.; De Vos, W.M. The first 1000 cultured species of the human gastrointestinal microbiota. FEMS Microbiol. Rev. 2014, 38, 996–1047.
- Sirisinha, S. The potential impact of gut microbiota on your health: Current status and future challenges. Asian Pac. J. Allergy Immunol. 2016, 34, 249–264.
- Huttenhower, C.; Gevers, D.; Knight, R.; Abubucker, S.; Badger, J.H.; Chinwalla, A.T.; Creasy, H.H.; Earl, A.M.; FitzGerald, M.G.; Fulton, R.S.; et al. Structure, function and diversity of the healthy human microbiome. Nature 2012, 486, 207–214.
- Samuelson, D.R.; Welsh, D.A.; Shellito, J.E. Regulation of lung immunity and host defense by the intestinal microbiota. Front. Microbiol. 2015, 6, 1085.
- Dawson, L.F.; Donahue, E.H.; Cartman, S.T.; Barton, R.H.; Bundy, J.; McNerney, R.; Minton, N.P.; Wren, B.W. The analysis of para-cresol production and tolerance in Clostridium difficile 027 and 012 strains. BMC Microbiol. 2011, 11, 1–10.
- Miyazaki, K.; Masuoka, N.; Kano, M.; Iizuka, R. Bifidobacterium fermented milk and galacto-oligosaccharides lead to improved skin health by decreasing phenols production by gut microbiota. Benef. Microbes 2014, 5, 121–128.
- Zákostelská, Z.; Málková, J.; Klimešová, K.; Rossmann, P.; Hornová, M.; Novosádová, I.; Stehlíková, Z.; Kostovčík, M.; Hudcovic, T.; Štepánková, R. Intestinal microbiota promotes psoriasis-like skin inflammation by enhancing Th17 response. PLoS One 2016, 11, e0159539.
- Stehlikova, Z.; Kostovcikova, K.; Kverka, M.; Rossmann, P.; Dvorak, J.; Novosadova, I.; Kostovcik, M.; Coufal, S.; Srutkova, D.; Prochazkova, P. Crucial role of microbiota in experimental psoriasis revealed by a gnotobiotic mouse model. Front. Microbiol. 2019, 10, 236.
- Suzuki, K.; Kawamoto, S.; Maruya, M.; Fagarasan, S. GALT: Organization and dynamics leading to IgA synthesis. Adv. Immunol. 2010, 107, 153–185.
- Adarsh, M.; Dogra, S.; Vaiphei, K.; Vaishnavi, C.; Sinha, S.; Sharma, A. Evaluation of subclinical gut inflammation using faecal calprotectin levels and colonic mucosal biopsy in patients with psoriasis and psoriatic arthritis. Br. J. Dermatol. 2019, 181, 401–402.
- Sikora, M.; Chrabąszcz, M.; Maciejewski, C.; Zaremba, M.; Waśkiel, A.; Olszewska, M.; Rudnicka, L. Intestinal barrier integrity in patients with plaque psoriasis. J. Dermatol 2018, 45, 1468–1470.
- Vlachos, C.; Gaitanis, G.; Katsanos, K.H.; Christodoulou, D.K.; Tsianos, E.; Bassukas, I.D. Psoriasis and inflammatory bowel disease: Links and risks. Psoriasis (Auckl) 2016, 6, 73.
- Visser, M.J.; Kell, D.B.; Pretorius, E. Bacterial dysbiosis and translocation in psoriasis vulgaris. Front. Cell. Infect. Microbiol. 2019, 9, 7.
- Alesa, D.I.; Alshamrani, H.M.; Alzahrani, Y.A.; Alamssi, D.N.; Alzahrani, N.S.; Almohammadi, M.E. The role of gut microbiome in the pathogenesis of psoriasis and the therapeutic effects of probiotics. J. Family Med. Prim. Care 2019, 8, 3496.
- Forbes, J.D.; Van Domselaar, G.; Bernstein, C.N. The gut microbiota in immune-mediated inflammatory diseases. Front. Microbiol. 2016, 7, 1081.
- Shapiro, J.; Cohen, N.A.; Shalev, V.; Uzan, A.; Koren, O.; Maharshak, N. Psoriatic patients have a distinct structural and functional fecal microbiota compared with controls. J. Dermatol 2019, 46, 595–603.
- Kim, Y.-G.; Udayanga, K.G.S.; Totsuka, N.; Weinberg, J.B.; Núñez, G.; Shibuya, A. Gut dysbiosis promotes M2 macrophage polarization and allergic airway inflammation via fungi-induced PGE2. Cell Host Microbe 2014, 15, 95–102.
- Vinolo, M.A.; Rodrigues, H.G.; Nachbar, R.T.; Curi, R. Regulation of inflammation by short chain fatty acids. Nutrients 2011, 3, 858–876.
- Plöger, S.; Stumpff, F.; Penner, G.B.; Schulzke, J.D.; Gäbel, G.; Martens, H.; Shen, Z.; Günzel, D.; Aschenbach, J.R. Microbial butyrate and its role for barrier function in the gastrointestinal tract. Ann. N. Y. Acad. Sci. 2012, 1258, 52–59.
- Lopez-Siles, M.; Khan, T.M.; Duncan, S.H.; Harmsen, H.J.; Garcia-Gil, L.J.; Flint, H.J. Cultured representatives of two major phylogroups of human colonic Faecalibacterium prausnitzii can utilize pectin, uronic acids, and host-derived substrates for growth. Appl. Environ. Microbiol. 2012, 78, 420–428.
- Sitkin, S.; Pokrotnicks, J. Clinical Potential of Anti-inflammatory Effects of Faecalibacterium prausnitzii and Butyrate in Inflammatory Bowel Disease. Inflamm. Bowel Dis. 2019, 25, e40–e41.
- Codoñer, F.M.; Ramírez-Bosca, A.; Climent, E.; Carrión-Gutierrez, M.; Guerrero, M.; Pérez-Orquín, J.M.; De La Parte, J.H.; Genovés, S.; Ramón, D.; Navarro-López, V. Gut microbial composition in patients with psoriasis. Sci. Rep. 2018, 8, 1–7.
- Staiano-Coico, L.; Khandke, L.; Krane, J.F.; Sharif, S.; Gottlieb, A.B.; Krueger, J.G.; Heim, L.; Rigas, B.; Higgins, P.J. TGF-α and TGF-β expression during sodium-N-butyrate-induced differentiation of human keratinocytes: Evidence for subpopulation-specific up-regulation of TGF-β mRNA in suprabasal cells. Exp. Cell Res. 1990, 191, 286–291.
- Scepanovic, P.; Hodel, F.; Mondot, S.; Partula, V.; Byrd, A.; Hammer, C.; Alanio, C.; Bergstedt, J.; Patin, E.; Touvier, M. A comprehensive assessment of demographic, environmental, and host genetic associations with gut microbiome diversity in healthy individuals. Microbiome 2019, 7, 1–15.
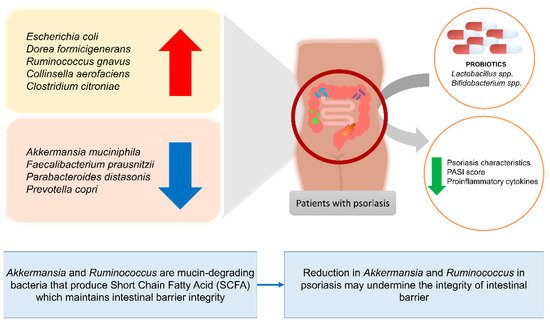
- Tan, L.; Zhao, S.; Zhu, W.; Wu, L.; Li, J.; Shen, M.; Lei, L.; Chen, X.; Peng, C. The Akkermansia muciniphila is a gut microbiota signature in psoriasis. Exp. Dermatol. 2018, 27, 144–149.
- Chen, Y.J.; Ho, H.J.; Tseng, C.H.; Lai, Z.L.; Shieh, J.J.; Wu, C.Y. Intestinal microbiota profiling and predicted metabolic dysregulation in psoriasis patients. Exp. Dermatol. 2018, 27, 1336–1343.
- Yeh, N.-L.; Hsu, C.-Y.; Tsai, T.-F.; Chiu, H.-Y. Gut microbiome in psoriasis is perturbed differently during secukinumab and ustekinumab therapy and associated with response to treatment. Clin. Drug Investig. 2019, 39, 1195–1203.
- Scher, J.U.; Ubeda, C.; Artacho, A.; Attur, M.; Isaac, S.; Reddy, S.M.; Marmon, S.; Neimann, A.; Brusca, S.; Patel, T. Decreased bacterial diversity characterizes the altered gut microbiota in patients with psoriatic arthritis, resembling dysbiosis in inflammatory bowel disease. Arthritis Rheumatol. 2015, 67, 128–139.
- Hidalgo-Cantabrana, C.; Gomez, J.; Delgado, S.; Requena-López, S.; Queiro-Silva, R.; Margolles, A.; Coto, E.; Sanchez, B.; Coto-Segura, P. Gut microbiota dysbiosis in a cohort of patients with psoriasis. Br. J. Dermatol. 2019, 181, 1287–1295.
- Huang, L.; Gao, R.; Yu, N.; Zhu, Y.; Ding, Y.; Qin, H. Dysbiosis of gut microbiota was closely associated with psoriasis. Sci. China Life Sci. 2019, 62, 807–815.
- Wagner, B.D.; Grunwald, G.K.; Zerbe, G.O.; Mikulich-Gilbertson, S.K.; Robertson, C.E.; Zemanick, E.T.; Harris, J.K. On the use of diversity measures in longitudinal sequencing studies of microbial communities. Front. Microbiol. 2018, 9, 1037.
- Masallat, D.; Moemen, D. Gut bacterial microbiota in psoriasis: A case control study. Afr. J. Microbiol. Res. 2016, 10, 1337–1343.
- Calcinaro, F.; Dionisi, S.; Marinaro, M.; Candeloro, P.; Bonato, V.; Marzotti, S.; Corneli, R.; Ferretti, E.; Gulino, A.; Grasso, F. Oral probiotic administration induces interleukin-10 production and prevents spontaneous autoimmune diabetes in the non-obese diabetic mouse. Diabetologia 2005, 48, 1565–1575.
- Lavasani, S.; Dzhambazov, B.; Nouri, M.; Fåk, F.; Buske, S.; Molin, G.; Thorlacius, H.; Alenfall, J.; Jeppsson, B.; Weström, B. A novel probiotic mixture exerts a therapeutic effect on experimental autoimmune encephalomyelitis mediated by IL-10 producing regulatory T cells. PLoS One 2010, 5, e9009.
- De Preter, V.; Machiels, K.; Joossens, M.; Arijs, I.; Matthys, C.; Vermeire, S.; Rutgeerts, P.; Verbeke, K. Faecal metabolite profiling identifies medium-chain fatty acids as discriminating compounds in IBD. Gut 2015, 64, 447–458.
- Kostic, A.D.; Xavier, R.J.; Gevers, D. The microbiome in inflammatory bowel disease: Current status and the future ahead. Gastroenterology 2014, 146, 1489–1499.
- Scher, J.U. The microbiome in psoriasis and psoriatic arthritis: Joints. J. Rheumatol. Suppl. 2018, 94, 32–35.
- Eppinga, H.; Sperna Weiland, C.J.; Thio, H.B.; van der Woude, C.J.; Nijsten, T.E.; Peppelenbosch, M.P.; Konstantinov, S.R. Similar depletion of protective Faecalibacterium prausnitzii in psoriasis and inflammatory bowel disease, but not in hidradenitis suppurativa. J. Crohn’s Colitis 2016, 10, 1067–1075.
- Cani, P.D.; de Vos, W.M. Next-generation beneficial microbes: The case of Akkermansia muciniphila. Front. Microbiol. 2017, 8, 1765.
- Li, J.; Lin, S.; Vanhoutte, P.M.; Woo, C.W.; Xu, A. Akkermansia muciniphila protects against atherosclerosis by preventing metabolic endotoxemia-induced inflammation in Apoe−/− mice. Circulation 2016, 133, 2434–2446.
- Schneeberger, M.; Everard, A.; Gómez-Valadés, A.G.; Matamoros, S.; Ramírez, S.; Delzenne, N.M.; Gomis, R.; Claret, M.; Cani, P.D. Akkermansia muciniphila inversely correlates with the onset of inflammation, altered adipose tissue metabolism and metabolic disorders during obesity in mice. Sci. Rep. 2015, 5, 1–14.
- Png, C.W.; Lindén, S.K.; Gilshenan, K.S.; Zoetendal, E.G.; McSweeney, C.S.; Sly, L.I.; McGuckin, M.A.; Florin, T.H. Mucolytic bacteria with increased prevalence in IBD mucosa augmentin vitroutilization of mucin by other bacteria. Am. J. Gastroenterol. 2010, 105, 2420–2428.
- Reunanen, J.; Kainulainen, V.; Huuskonen, L.; Ottman, N.; Belzer, C.; Huhtinen, H.; de Vos, W.M.; Satokari, R. Akkermansia muciniphila adheres to enterocytes and strengthens the integrity of the epithelial cell layer. Appl. Environ. Microbiol. 2015, 81, 3655–3662.
- Bian, X.; Wu, W.; Yang, L.; Lv, L.; Wang, Q.; Li, Y.; Ye, J.; Fang, D.; Wu, J.; Jiang, X. Administration of Akkermansia muciniphila ameliorates dextran sulfate sodium-induced ulcerative colitis in mice. Front. Microbiol. 2019, 10, 2259.
- Gaffen, S.L.; Jain, R.; Garg, A.V.; Cua, D.J. The IL-23–IL-17 immune axis: From mechanisms to therapeutic testing. Nat. Rev. Immunol. 2014, 14, 585–600.
- Warren, R.; Menter, A. Handbook of Psoriasis and Psoriatic Arthritis; Springer: Berlin/Heidelberg, Germany, 2016.
- Salem, I.; Ramser, A.; Isham, N.; Ghannoum, M.A. The gut microbiome as a major regulator of the gut-skin axis. Front. Microbiol. 2018, 9, 1459.
- Song, C.; Yang, C.; Meng, S.; Li, M.; Wang, X.; Zhu, Y.; Kong, L.; Lv, W.; Qiao, H.; Sun, Y. Deciphering the mechanism of Fang-Ji-Di-Huang-Decoction in ameliorating psoriasis-like skin inflammation via the inhibition of IL-23/Th17 cell axis. J. Ethnopharmacol. 2021, 281, 114571.
- Yao, X.; Zhu, Z.; Manandhar, U.; Liao, H.; Yu, T.; Wang, Y.; Bian, Y.; Zhang, B.; Zhang, X.; Xie, J.; et al. RNA-seq reveal RNA binding protein GNL3 as a key mediator in the development of psoriasis vulgaris by regulating the IL23/IL17 axis. Life Sci. 2022, 293, 119902.
- Haugh, I.M.; Preston, A.K.; Kivelevitch, D.N.; Menter, A.M. Risankizumab: An anti-IL-23 antibody for the treatment of psoriasis. Drug Des. Devel. Ther. 2018, 12, 3879.
- Dong, J.; Goldenberg, G. New biologics in psoriasis: An update on IL-23 and IL-17 inhibitors. Cutis 2017, 99, 123–127.
- Kamata, M.; Tada, Y. Safety of biologics in psoriasis. J. Dermatol 2018, 45, 279–286.
- Molinelli, E.; Campanati, A.; Brisigotti, V.; Offidani, A. Biologic therapy in psoriasis (Part II): Efficacy and safety of new treatment targeting IL23/IL-17 pathways. Curr. Pharm. Biotechnol. 2017, 18, 964–978.
- Yan, D.; Issa, N.; Afifi, L.; Jeon, C.; Chang, H.-W.; Liao, W. The role of the skin and gut microbiome in psoriatic disease. Curr. Dermatol. Rep. 2017, 6, 94–103.
- Tang, W.W.; Wang, Z.; Levison, B.S.; Koeth, R.A.; Britt, E.B.; Fu, X.; Wu, Y.; Hazen, S.L. Intestinal microbial metabolism of phosphatidylcholine and cardiovascular risk. N. Engl. J. Med. 2013, 368, 1575–1584.
- Chen, Y.-H.; Wu, C.-S.; Chao, Y.-H.; Lin, C.-C.; Tsai, H.-Y.; Li, Y.-R.; Chen, Y.-Z.; Tsai, W.-H.; Chen, Y.-K. Lactobacillus pentosus GMNL-77 inhibits skin lesions in imiquimod-induced psoriasis-like mice. J. Food Drug Anal. 2017, 25, 559–566.
- Groeger, D.; O’Mahony, L.; Murphy, E.F.; Bourke, J.F.; Dinan, T.G.; Kiely, B.; Shanahan, F.; Quigley, E.M. Bifidobacterium infantis 35624 modulates host inflammatory processes beyond the gut. Gut Microbes 2013, 4, 325–339.
- Huang, Y.J.; Marsland, B.J.; Bunyavanich, S.; O’Mahony, L.; Leung, D.Y.; Muraro, A.; Fleisher, T.A. The microbiome in allergic disease: Current understanding and future opportunities—2017 PRACTALL document of the American Academy of Allergy, Asthma & Immunology and the European Academy of Allergy and Clinical Immunology. J. Allergy Clin. Immunol. 2017, 139, 1099–1110.
- Gasbarrini, G.; Bonvicini, F.; Gramenzi, A. Probiotics history. J. Clin. Gastroenterol. 2016, 50, S116–S119.
- George Kerry, R.; Patra, J.K.; Gouda, S.; Park, Y.; Shin, H.-S.; Das, G. Benefaction of probiotics for human health: A review. J. Food Drug Anal. 2018, 26, 927–939.
- Yamashiro, Y. Probiotics to Prebiotics and Their Clinical Use. In Encyclopedia of Infection and Immunity; Rezaei, N., Ed.; Elsevier: Oxford, UK, 2022; pp. 674–688.
- Hui, M.L.; Tan, L.T.; Letchumanan, V.; He, Y.W.; Fang, C.M.; Chan, K.G.; Law, J.W.; Lee, L.H. The Extremophilic Actinobacteria: From Microbes to Medicine. Antibiotics 2021, 6, 682.
- Tan, L.T.-H.; Chan, K.-G.; Lee, L.-H.; Goh, B.-H. Streptomyces Bacteria as Potential Probiotics in Aquaculture. Front. Microbiol. 2016, 7.
- Law, J.; Letchumanan, V.; Teng Hern, T.; Ser, H.L.; Goh, B.H.; Lee, L.H. The Rising of “Modern Actinobacteria” Era. Prog. Microbes Mol. Biol. 2020, 3.
- Gueniche, A.; Philippe, D.; Bastien, P.; Reuteler, G.; Blum, S.; Castiel-Higounenc, I.; Breton, L.; Benyacoub, J. Randomised double-blind placebo-controlled study of the effect of Lactobacillus paracasei NCC 2461 on skin reactivity. Benef. Microbes 2014, 5, 137–145.
- Benyacoub, J.; Bosco, N.; Blanchard, C.; Demont, A.; Philippe, D.; Castiel-Higounenc, I.; Guéniche, A. Immune modulation property of Lactobacillus paracasei NCC2461 (ST11) strain and impact on skin defences. Benef. Microbes 2014, 5, 129–136.
- Baba, H.; Masuyama, A.; Yoshimura, C.; Aoyama, Y.; Takano, T.; Ohki, K. Oral intake of Lactobacillus helveticus-fermented milk whey decreased transepidermal water loss and prevented the onset of sodium dodecylsulfate-induced dermatitis in mice. Biosci. Biotechnol. Biochem. 2010, 74, 18–23.
- Gueniche, A.; Benyacoub, J.; Philippe, D.; Bastien, P.; Kusy, N.; Breton, L.; Blum, S.; Castiel-Higounenc, I. Lactobacillus paracasei CNCM I-2116 (ST11) inhibits substance P-induced skin inflammation and accelerates skin barrier function recovery in vitro. Eur. J. Dermatol. 2010, 20, 731–737.
- Philippe, D.; Stephanie, B.; Benyacoub, J. Oral Lactobacillus paracasei improves skin barrier function recovery and reduces local skin inflammation. Eur. J. Dermatol. 2011, 21, 279–280.
- Poutahidis, T.; Kearney, S.M.; Levkovich, T.; Qi, P.; Varian, B.J.; Lakritz, J.R.; Ibrahim, Y.M.; Chatzigiagkos, A.; Alm, E.J.; Erdman, S.E. Microbial symbionts accelerate wound healing via the neuropeptide hormone oxytocin. PLoS One 2013, 8, e78898.
- Ogawa, C.; Inoue, R.; Yonejima, Y.; Hisa, K.; Yamamoto, Y.; Suzuki, T. Supplemental Leuconostoc mesenteroides strain NTM048 attenuates imiquimod-induced psoriasis in mice. J. Appl. Microbiol. 2021, 131, 3043–3055.
- Lu, W.; Deng, Y.; Fang, Z.; Zhai, Q.; Cui, S.; Zhao, J.; Chen, W.; Zhang, H. Potential role of probiotics in ameliorating psoriasis by modulating gut microbiota in imiquimod-induced psoriasis-like mice. Nutrients 2021, 13, 2010.
- Deng, Y.D.; Fang, Z.F.; Cui, S.M.; Zhao, J.X.; Zhang, H.; Chen, W.; Lu, W.W. Evaluation of probiotics for inhibiting hyperproliferation and inflammation relevant to psoriasis in vitro. J. Funct. Foods 2021, 81.
- Thio, H.B. The microbiome in psoriasis and psoriatic arthritis: The skin perspective. J. Rheumatol. Suppl. 2018, 94, 30–31.
- Vijayashankar, M.; Raghunath, N. Pustular psoriasis responding to probiotics–A new insight. Our Dermatol. Online 2012, 3, 326.
- Lin, C.; Zeng, T.; Deng, Y.; Yang, W.; Xiong, J. Treatment of psoriasis vulgaris using Bacteroides fragilis BF839: A single-arm, open preliminary clinical study. Shengwu Gongcheng Xuebao/Chin. J. Biotechnol. 2021, 37, 3828–3835.
- Moludi, J.; Khedmatgozar, H.; Saiedi, S.; Razmi, H.; Alizadeh, M.; Ebrahimi, B. Probiotic supplementation improves clinical outcomes and quality of life indicators in patients with plaque psoriasis: A randomized double-blind clinical trial. Clin. Nutr. ESPEN 2021, 46, 33–39.
- Akbarzadeh, A.; Taheri, M.; Ebrahimi, B.; Alirezaei, P.; Doosti-Irani, A.; Soleimani, M.; Nouri, F. Evaluation of Lactocare® Synbiotic Administration on the Serum Electrolytes and Trace Elements Levels in Psoriasis Patients: A Randomized, Double-Blind, Placebo-Controlled Clinical Trial Study. Biol. Trace Elem. Res. 2021.
- Navarro-López, V.; Martínez-Andrés, A.; Ramírez-Boscá, A.; Ruzafa-Costas, B.; Núñez-Delegido, E.; Carrión-Gutiérrez, M.A.; Prieto-Merino, D.; Codoñer-Cortés, F.; Ramón-Vidal, D.; Genovés-Martínez, S.; et al. Efficacy and Safety of Oral Administration of a Mixture of Probiotic Strains in Patients with Psoriasis: A Randomized Controlled Clinical Trial. Acta Derm. Venereol. 2019, 99, 1078–1084.
- Davidovici, B.B.; Sattar, N.; Jörg, P.C.; Puig, L.; Emery, P.; Barker, J.N.; Van De Kerkhof, P.; Ståhle, M.; Nestle, F.O.; Girolomoni, G. Psoriasis and systemic inflammatory diseases: Potential mechanistic links between skin disease and co-morbid conditions. J. Invest. Dermatol. 2010, 130, 1785–1796.
- Dowlatshahi, E.; Van Der Voort, E.; Arends, L.; Nijsten, T. Markers of systemic inflammation in psoriasis: A systematic review and meta-analysis. Br. J. Dermatol. 2013, 169, 266–282.
- Suárez-Farinas, M.; Li, K.; Fuentes-Duculan, J.; Hayden, K.; Brodmerkel, C.; Krueger, J.G. Expanding the psoriasis disease profile: Interrogation of the skin and serum of patients with moderate-to-severe psoriasis. J. Invest. Dermatol. 2012, 132, 2552–2564.
- El Dib, R.; Periyasamy, A.G.; de Barros, J.L.; França, C.G.; Senefonte, F.L.; Vesentini, G.; Alves, M.G.O.; Rodrigues, J.V.d.S.; Gomaa, H.; Gomes Júnior, J.R.; et al. Probiotics for the treatment of depression and anxiety: A systematic review and meta-analysis of randomized controlled trials. Clin. Nutr. ESPEN 2021, 45, 75–90.
- McFarland, L.V.; Karakan, T.; Karatas, A. Strain-specific and outcome-specific efficacy of probiotics for the treatment of irritable bowel syndrome: A systematic review and meta-analysis. EClinicalMedicine 2021, 41, 101154.
- Kano, M.; Masuoka, N.; Kaga, C.; Sugimoto, S.; Iizuka, R.; Manabe, K.; Sone, T.; Oeda, K.; Nonaka, C.; Miyazaki, K. Consecutive intake of fermented milk containing Bifidobacterium breve strain Yakult and galacto-oligosaccharides benefits skin condition in healthy adult women. Biosci. Microbiota Food Health 2013, 32, 33–39.
- Lee, J.; Suk, J.; Kang, S. Effect of Lactobacillus rhamnosus KCTC 5033 on the Appearance of Facial Skin due to the Ingestion of Probiotics and Paraprobiotics. J. Investig. Cosmetol 2018, 14, 287–296.
- Sabbagh, F.; Kim, B.S. Recent advances in polymeric transdermal drug delivery systems. J. Control. Release 2022, 341, 132–146.
- Crovesy, L.; Masterson, D.; Rosado, E.L. Profile of the gut microbiota of adults with obesity: A systematic review. Eur. J. Clin. Nutr. 2020, 74, 1251–1262.
- Sobhonslidsuk, A.; Chanprasertyothin, S.; Pongrujikorn, T.; Kaewduang, P.; Promson, K.; Petraksa, S.; Ongphiphadhanakul, B. The association of gut microbiota with nonalcoholic steatohepatitis in Thais. Biomed Res. Int. 2018, 2018, 9340316.
- Yoshida, N.; Yamashita, T.; Hirata, K.-i. Gut microbiome and cardiovascular diseases. Diseases 2018, 6, 56.
- Moreno-Indias, I.; Sánchez-Alcoholado, L.; García-Fuentes, E.; Cardona, F.; Queipo-Ortuño, M.I.; Tinahones, F.J. Insulin resistance is associated with specific gut microbiota in appendix samples from morbidly obese patients. Am. J. Transl. Res 2016, 8, 5672.
- Wang, Z.; Klipfell, E.; Bennett, B.J.; Koeth, R.; Levison, B.S.; DuGar, B.; Feldstein, A.E.; Britt, E.B.; Fu, X.; Chung, Y.-M. Gut flora metabolism of phosphatidylcholine promotes cardiovascular disease. Nature 2011, 472, 57–63.
- Rath, S.; Heidrich, B.; Pieper, D.H.; Vital, M. Uncovering the trimethylamine-producing bacteria of the human gut microbiota. Microbiome 2017, 5, 54.
- Koeth, R.A.; Wang, Z.; Levison, B.S.; Buffa, J.A.; Org, E.; Sheehy, B.T.; Britt, E.B.; Fu, X.; Wu, Y.; Li, L. Intestinal microbiota metabolism of L-carnitine, a nutrient in red meat, promotes atherosclerosis. Nat. Med. 2013, 19, 576–585.
- Valguarnera, E.; Wardenburg, J.B. Good gone bad: One toxin away from disease for Bacteroides fragilis. J. Mol. Biol. 2020, 432, 765–785.
- Guerreiro, C.S.; Calado, Â.; Sousa, J.; Fonseca, J.E. Diet, microbiota, and gut permeability—the unknown triad in rheumatoid arthritis. Front. Med. 2018, 5, 349.
- Zhou, C.; Zhao, H.; Xiao, X.-y.; Guo, R.-j.; Wang, Q.; Chen, H.; Zhao, L.-d.; Zhang, C.-C.; Jiao, Y.-H.; Ju, Y.-m. Metagenomic profiling of the pro-inflammatory gut microbiota in ankylosing spondylitis. J. Autoimmun. 2020, 107, 102360.
- De Filippis, F.; Pasolli, E.; Tett, A.; Tarallo, S.; Naccarati, A.; De Angelis, M.; Neviani, E.; Cocolin, L.; Gobbetti, M.; Segata, N. Distinct genetic and functional traits of human intestinal Prevotella copri strains are associated with different habitual diets. Cell Host Microbe 2019, 25, 444–453.e443.
- Tett, A.; Huang, K.D.; Asnicar, F.; Fehlner-Peach, H.; Pasolli, E.; Karcher, N.; Armanini, F.; Manghi, P.; Bonham, K.; Zolfo, M. The Prevotella copri complex comprises four distinct clades underrepresented in westernized populations. Cell Host Microbe 2019, 26, 666–679.e667.
- Miquel, S.; Martin, R.; Rossi, O.; Bermúdez-Humarán, L.; Chatel, J.; Sokol, H.; Thomas, M.; Wells, J.; Langella, P. Faecalibacterium prausnitzii and human intestinal health. Curr. Opin. Microbiol. 2013, 16, 255–261.
- Stefia, L.V.; Lee, J.; Patel, J.; de Sousa, S.R.; Legrand, J.; Rodero, M.; Burman, S.; Linedale, R.; Morrison, M.; Khosrotehrani, K. Secretome components from Faecalibacterium prausnitzii strains A2-165 and AHMP21 Modulate Cutaneous Wound Inflammation. J. Invest. Dermatol. 2020, 140, 2312–2315.e2316.
- Hansen, R.; Russell, R.K.; Reiff, C.; Louis, P.; McIntosh, F.; Berry, S.H.; Mukhopadhya, I.; Bisset, M.W.; Barclay, A.R.; Bishop, J. Microbiota of De-NovoPediatric IBD: Increased Faecalibacterium Prausnitziiand Reduced Bacterial Diversity in Crohn’s But Not in Ulcerative Colitis. Am. J. Gastroenterol. 2012, 107, 1913–1922.
- Zheng, H.; Liang, H.; Wang, Y.; Miao, M.; Shi, T.; Yang, F.; Liu, E.; Yuan, W.; Ji, Z.-S.; Li, D.-K. Altered gut microbiota composition associated with eczema in infants. PLoS One 2016, 11, e0166026.
- Everard, A.; Belzer, C.; Geurts, L.; Ouwerkerk, J.P.; Druart, C.; Bindels, L.B.; Guiot, Y.; Derrien, M.; Muccioli, G.G.; Delzenne, N.M. Cross-talk between Akkermansia muciniphila and intestinal epithelium controls diet-induced obesity. Proc. Natl. Acad. Sci. USA 2013, 110, 9066–9071.
- Toya, T.; Corban, M.T.; Marrietta, E.; Horwath, I.E.; Lerman, L.O.; Murray, J.A.; Lerman, A. Coronary artery disease is associated with an altered gut microbiome composition. PLoS One 2020, 15, e0227147.
- Sikora, M.; Chrabąszcz, M.; Waśkiel-Burnat, A.; Rakowska, A.; Olszewska, M.; Rudnicka, L. Claudin-3–a new intestinal integrity marker in patients with psoriasis: Association with disease severity. J. Eur. Acad. Dermatol. Venereol. 2019, 33, 1907–1912.
- Sikora, M.; Stec, A.; Chrabaszcz, M.; Waskiel-Burnat, A.; Zaremba, M.; Olszewska, M.; Rudnicka, L. Intestinal fatty acid binding protein, a biomarker of intestinal barrier, is associated with severity of psoriasis. J. Clin. Med. 2019, 8, 1021.
- Mottawea, W.; Butcher, J.; Li, J.; Abujamel, T.; Manoogian, J.; Mack, D.; Stintzi, A. The mucosal–luminal interface: An ideal sample to study the mucosa-associated microbiota and the intestinal microbial biogeography. Pediatr. Res. 2019, 85, 895–903.
