Two years have passed since WHO declared a pandemic state for SARS-CoV-2 infection. COVID-19 pathogenesis consists of a first viral phase responsible for early symptoms followed by an inflammatory phase, cytokine-mediated, responsible for late-onset manifestations up to ARDS. The dysregulated immune response has an outstanding role in the progression of pulmonary damage in COVID-19. IL-6, through the induction of pro-inflammatory chemokines and cytokines, plays a key role in the development and maintenance of inflammation, acting as a pioneer of the hyperinflammatory condition and cytokine storm in severe COVID-19. Therefore, drugs targeting both IL-6 and IL-6 receptors have been evaluated in order to blunt the abnormal SARS-CoV-2-induced cytokine release. Sarilumab, a high-affinity anti-IL-6 receptor antibody, may represent a promising weapon to treat the fearsome hyperinflammatory phase by improving the outcome of patients with moderate-to-severe COVID-19 pneumonia.
- COVID-19
- cytokines
- IL-6
- IL-6R
1. Considerations concerning IL-6, IL-6R, and Sarilumab
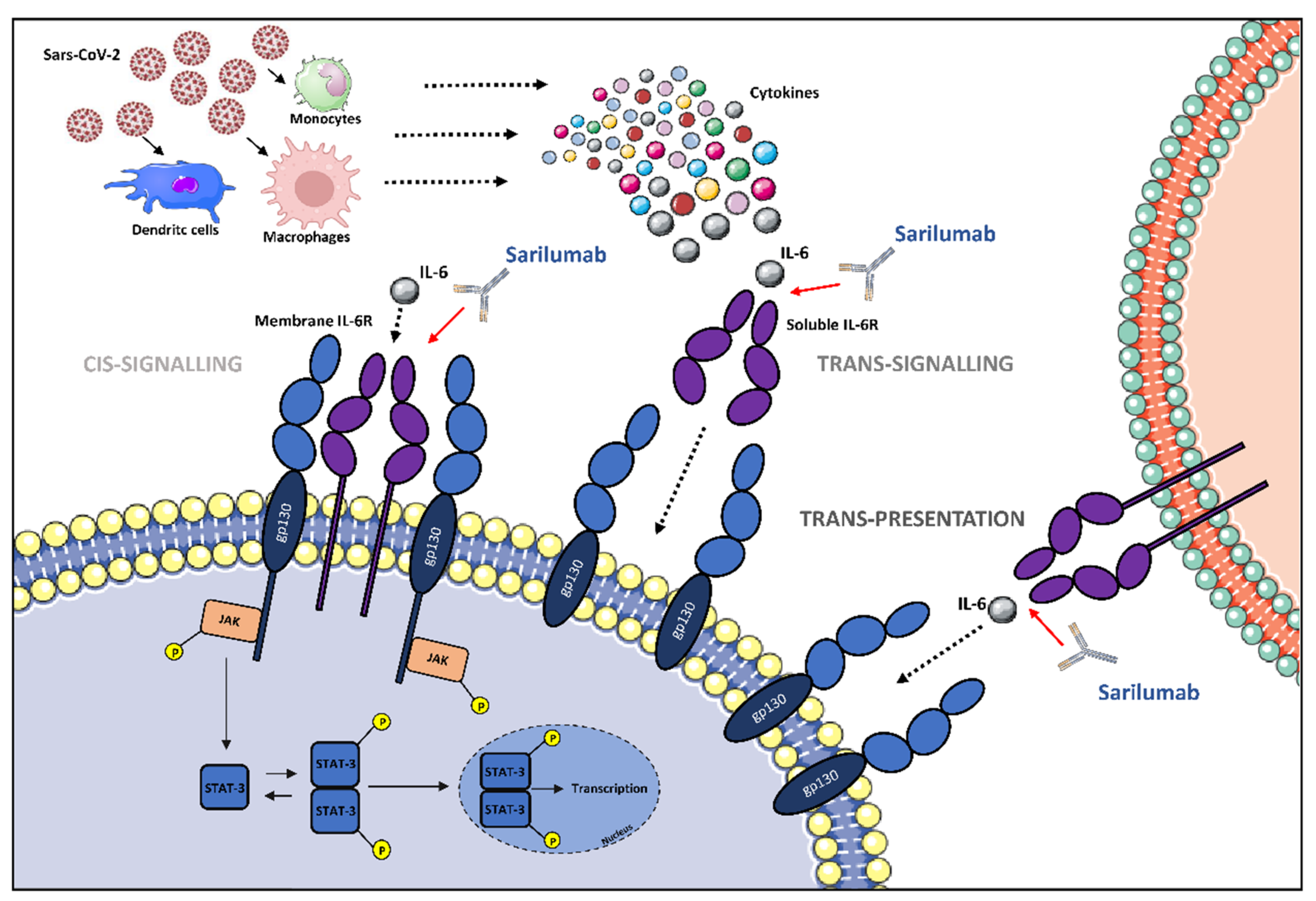 Figure 1. The potential role of IL-6 receptor antagonization by sarilumab in SARS-CoV-2-induced cytokine release syndrome (CRS)-like syndrome. After SARS-CoV-2 infection, immune cells release cytokines, including IL-6. IL-6 can either attach to its respective membrane-bound receptor (mIL-6R) or soluble receptor (sIL-6R), which activates JAK/STAT pathways. The different signaling modalities, including cis-signaling, trans-signaling, and trans-presentation are reported. Abbreviations: gp130, glycoprotein 130; IL-6, interleukin-6; JAK, Janus family tyrosine kinase; mIL-6R, membrane-bound interleukin-6 receptor; sIL-6R, soluble interleukin-6 receptor; STAT, signal transducer and activator of transcription; P, phosphoryl group. Illustrations use elements from Servier Medical Art (https://smart.servier.com/, accessed on 10 April 2022).
Figure 1. The potential role of IL-6 receptor antagonization by sarilumab in SARS-CoV-2-induced cytokine release syndrome (CRS)-like syndrome. After SARS-CoV-2 infection, immune cells release cytokines, including IL-6. IL-6 can either attach to its respective membrane-bound receptor (mIL-6R) or soluble receptor (sIL-6R), which activates JAK/STAT pathways. The different signaling modalities, including cis-signaling, trans-signaling, and trans-presentation are reported. Abbreviations: gp130, glycoprotein 130; IL-6, interleukin-6; JAK, Janus family tyrosine kinase; mIL-6R, membrane-bound interleukin-6 receptor; sIL-6R, soluble interleukin-6 receptor; STAT, signal transducer and activator of transcription; P, phosphoryl group. Illustrations use elements from Servier Medical Art (https://smart.servier.com/, accessed on 10 April 2022).2. Cytokine Storm and Rationale for IL-6 Inhibitor Administration in COVID-19 Patients
4. Evidence
5. Conclusions
This entry is adapted from the peer-reviewed paper 10.3390/idr14030040
References
- Kishimoto, T. IL-6: From its discovery to clinical applications. Int. Immunol. 2010, 22, 347–352.
- Mihara, M.; Hashizume, M.; Yoshida, H.; Suzuki, M.; Shiina, M. IL-6/IL-6 receptor system and its role in physiological and pathological conditions. Clin. Sci. 2012, 122, 143–159.
- Hunter, C.A.; Jones, S.A. IL-6 as a keystone cytokine in health and disease. Nat. Immunol. 2015, 16, 448–457.
- Hwang, S.Y.; Kim, J.Y.; Kim, K.W.; Park, M.K.; Moon, Y.; Kim, W.U.; Kim, H.Y. IL-17 induces production of IL-6 and IL-8 in rheumatoid arthritis synovial fibroblasts via NF-kappaB- and PI3-kinase/Akt-dependent pathways. Arthritis Res. Ther. 2004, 6, R120.
- Sallenave, J.M.; Guillot, L. Innate Immune Signaling and Proteolytic Pathways in the Resolution or Exacerbation of SARS-CoV-2 in COVID-19: Key Therapeutic Targets? Front. Immunol. 2020, 11, 1229.
- Mino, T.; Murakawa, Y.; Fukao, A.; Vandenbon, A.; Wessels, H.H.; Ori, D.; Uehata, T.; Tartey, S.; Akira, S.; Suzuki, Y.; et al. Regnase-1 and roquin regulate a common element in inflammatory mRNAs by spatiotemporally distinct mechanisms. Cell 2015, 161, 1058–1073.
- Higa, M.; Oka, M.; Fujihara, Y.; Masuda, K.; Yoneda, Y.; Kishimoto, T. Regulation of inflammatory responses by dynamic subcellular localization of RNA-binding protein Arid5a. Proc. Natl. Acad. Sci. USA 2018, 115, E1214–E1220.
- Garbers, C.; Heink, S.; Korn, T.; Rose-John, S. Interleukin-6: Designing specific therapeutics for a complex cytokine. Nat. Rev. Drug Discov. 2018, 17, 395–412.
- Rose-John, S. Il-6 trans-signaling via the soluble IL-6 receptor: Importance for the proinflammatory activities of IL-6. Int. J. Biol. Sci. 2012, 8, 1237–1247.
- Narazaki, M.; Kishimoto, T. The two-faced cytokine IL-6 in host defense and diseases. Int. J. Mol. Sci. 2018, 19, 3528.
- Xu, H.; Liu, J.; Niu, M.; Song, S.; Wei, L.; Chen, G.; Ding, Y.; Wang, Y.; Su, Z.; Wang, H. Soluble IL-6R-mediated IL-6 trans-signaling activation contributes to the pathological development of psoriasis. J. Mol. Med. 2021, 99, 1009–1020.
- Heinrich, P.C.; Behrmann, I.; Haan, S.; Hermanns, H.M.; Müller-Newen, G.; Schaper, F. Principles of interleukin (IL)-6-type cytokine signalling and its regulation. Biochem. J. 2003, 374, 1–20.
- Atal, S.; Fatima, Z. IL-6 Inhibitors in the Treatment of Serious COVID-19: A Promising Therapy? Pharmaceut. Med. 2020, 34, 223–231.
- Kim, G.W.; Lee, N.R.; Pi, R.H.; Lim, Y.S.; Lee, Y.M.; Lee, J.M.; Jeong, H.S.; Chung, S.H. IL-6 inhibitors for treatment of rheumatoid arthritis: Past, present, and future. Arch. Pharm. Res. 2015, 38, 575–584.
- Kitamura, H.; Ohno, Y.; Toyoshima, Y.; Ohtake, J.; Homma, S.; Kawamura, H.; Takahashi, N.; Taketomi, A. Interleukin-6/STAT3 signaling as a promising target to improve the efficacy of cancer immunotherapy. Cancer Sci. 2017, 108, 1947–1952.
- Calabrese, L.H.; Rose-John, S. IL-6 biology: Implications for clinical targeting in rheumatic disease. Nat. Rev. Rheumatol. 2014, 10, 720–727.
- Genovese, M.C.; Fleischmann, R.M.; Fiore, S.; Radin, A.; Fan, C.; Huizinga, T.W. SAT0117 Sarilumab, a Subcutaneously-Administered, Fully-Human Monoclonal Antibody Inhibitor of the IL-6 Receptor: Relationship between Eular Responses and Change from Baseline of Selected Clinical Parameters. Ann. Rheum. Dis. 2013, 72, A620.
- Khiali, S.; Rezagholizadeh, A.; Entezari-Maleki, T. A comprehensive review on sarilumab in COVID-19. Exp. Opin. Biol. Ther. 2021, 21, 615–626.
- Bae, S.C.; Lee, Y.H. Comparative efficacy and tolerability of sarilumab 150 and 200 mg in patients with active rheumatoid arthritis: A Bayesian network meta-analysis of randomized controlled trials. Z. Rheumatol. 2018, 77, 421–428.
- Zizzo, G.; Tamburello, A.; Castelnovo, L.; Laria, A.; Mumoli, N.; Faggioli, P.M.; Stefani, I.; Mazzone, A. Immunotherapy of COVID-19: Inside and Beyond IL-6 Signalling. Front. Immunol. 2022, 13, 795315.
- Channappanavar, R.; Perlman, S. Pathogenic human coronavirus infections: Causes and consequences of cytokine storm and immunopathology. Semin. Immunopathol. 2017, 39, 529–539.
- Al-Hajeri, H.; Baroun, F.; Abutiban, F.; Al-Mutairi, M.; Ali, Y.; Alawadhi, A.; Albasri, A.; Aldei, A.; AlEnizi, A.; Alhadhood, N.; et al. Therapeutic role of immunomodulators during the COVID-19 pandemic—A narrative review. Postgrad. Med. 2022, 134, 160–179.
- Cazzato, G.; Colagrande, A.; Cimmino, A.; Cicco, G.; Scarcella, V.S.; Tarantino, P.; Lospalluti, L.; Romita, P.; Foti, C.; Demarco, A.; et al. HMGB1-TIM3-HO1: A new pathway of inflammation in skin of SARS-CoV-2 patients? A retrospective pilot study. Biomolecules 2021, 11, 1219.
- Cazzato, G.; Foti, C.; Colagrande, A.; Cimmino, A.; Scarcella, S.; Cicco, G.; Sablone, S.; Arezzo, F.; Romita, P.; Lettini, T.; et al. Skin manifestation of SARS-CoV-2: The italian experience. J. Clin. Med. 2021, 10, 1566.
- Cazzato, G.; Mazzia, G.; Cimmino, A.; Colagrande, A.; Sablone, S.; Lettini, T.; Rossi, R.; Santarella, N.; Elia, R.; Nacchiero, E.; et al. SARS-CoV-2 and skin: The pathologist’s point of view. Biomolecules 2021, 11, 838.
- Buszko, M.; Nita-Lazar, A.; Park, J.H.; Schwartzberg, P.L.; Verthelyi, D.; Young, H.A.; Rosenberg, A.S. Lessons learned: New insights on the role of cytokines in COVID-19. Nat. Immunol. 2021, 22, 404–411.
- Zhou, Y.; Fu, B.; Zheng, X.; Wang, D.; Zhao, C.; Qi, Y.; Sun, R.; Tian, Z.; Xu, X.; Wei, H. Pathogenic T-cells and inflammatory monocytes incite inflammatory storms in severe COVID-19 patients. Natl. Sci. Rev. 2020, 7, 998–1002.
- Lee, D.W.; Gardner, R.; Porter, D.L.; Louis, C.U.; Ahmed, N.; Jensen, M.; Grupp, S.A.; Mackall, C.L. Current concepts in the diagnosis and management of cytokine release syndrome. Blood 2014, 124, 188–195.
- Coomes, E.A.; Haghbayan, H. Interleukin-6 in COVID-19: A systematic review and meta-analysis. Rev. Med. Virol. 2020, 30, 1–9.
- Kayesh, M.E.H.; Kohara, M.; Tsukiyama-kohara, K. An overview of recent insights into the response of tlr to SARS-CoV-2 infection and the potential of tlr agonists as SARS-CoV-2 vaccine adjuvants. Viruses 2021, 13, 2302.
- Conti, P.; Ronconi, G.; Caraffa, A.; Gallenga, C.E.; Ross, R.; Frydas, I.; Kritas, S.K. Induction of pro-inflammatory cytokines (IL-1 and IL-6) and lung inflammation by Coronavirus-19 (COVI-19 or SARS-CoV-2): Anti-inflammatory strategies. J. Biol. Regul. Homeost. Agents 2020, 34, 327–331.
- Han, H.; Ma, Q.; Li, C.; Liu, R.; Zhao, L.; Wang, W.; Zhang, P.; Liu, X.; Gao, G.; Liu, F.; et al. Profiling serum cytokines in COVID-19 patients reveals IL-6 and IL-10 are disease severity predictors. Emerg. Microbes Infect. 2020, 9, 1123–1130.
- Diehl, S.; Anguita, J.; Hoffmeyer, A.; Zapton, T.; Ihle, J.N.; Fikrig, E.; Rincón, M. Inhibition of Th1 differentiation by IL-6 is mediated by SOCS1. Immunity 2000, 13, 805–815.
- Samavati, L.; Uhal, B.D. ACE2, Much More Than Just a Receptor for SARS-CoV-2. Front. Cell. Infect. Microbiol. 2020, 10, 317.
- Cifaldi, L.; Prencipe, G.; Caiello, I.; Bracaglia, C.; Locatelli, F.; De Benedetti, F.; Strippoli, R. Inhibition of natural killer cell cytotoxicity by interleukin-6: Implications for the pathogenesis of macrophage activation syndrome. Arthritis Rheumatol. 2015, 67, 3037–3046.
- Zizzo, G.; Cohen, P.L. Imperfect storm: Is interleukin-33 the Achilles heel of COVID-19? Lancet Rheumatol. 2020, 2, e779–e790.
- McGonagle, D.; Sharif, K.; O’Regan, A.; Bridgewood, C. The Role of Cytokines including Interleukin-6 in COVID-19 induced Pneumonia and Macrophage Activation Syndrome-Like Disease. Autoimmun. Rev. 2020, 19, 102537.
- Santa Cruz, A.; Mendes-Frias, A.; Oliveira, A.I.; Dias, L.; Matos, A.R.; Carvalho, A.; Capela, C.; Pedrosa, J.; Castro, A.G.; Silvestre, R. Interleukin-6 Is a Biomarker for the Development of Fatal Severe Acute Respiratory Syndrome Coronavirus 2 Pneumonia. Front. Immunol. 2021, 12, 263.
- Bovijn, J.; Lindgren, C.M.; Holmes, M.V. Genetic variants mimicking therapeutic inhibition of IL-6 receptor signaling and risk of COVID-19. Lancet Rheumatol. 2020, 2, e658–e659.
- Shekhawat, J.; Gauba, K.; Gupta, S.; Purohit, P.; Mitra, P.; Garg, M.; Misra, S.; Sharma, P.; Banerjee, M. Interleukin-6 Perpetrator of the COVID-19 Cytokine Storm. Indian J. Clin. Biochem. 2021, 36, 440–450.
- Niemand, C.; Nimmesgern, A.; Haan, S.; Fischer, P.; Schaper, F.; Rossaint, R.; Heinrich, P.C.; Müller-Newen, G. Activation of STAT3 by IL-6 and IL-10 in Primary Human Macrophages Is Differentially Modulated by Suppressor of Cytokine Signaling 3. J. Immunol. 2003, 170, 3263–3272.
- Veiga, V.C.; Prats, J.A.G.G.; Farias, D.L.C.; Rosa, R.G.; Dourado, L.K.; Zampieri, F.G.; MacHado, F.R.; Lopes, R.D.; Berwanger, O.; Azevedo, L.C.P.; et al. Effect of tocilizumab on clinical outcomes at 15 days in patients with severe or critical coronavirus disease 2019: Randomised controlled trial. BMJ 2021, 372, n84.
- Sivapalasingam, S.; Lederer, D.; Bhore, R.; Hajizadeh, N.; Criner, G.; Hosain, R.; Sarilumab-COVID-19 Study Team. A randomized placebo-controlled trial of sarilumab in hospitalized patients with COVID-19. MedRxiv 2021.
- Que, Y.; Hu, C.; Wan, K.; Hu, P.; Wang, R.; Luo, J.; Li, T.; Ping, R.; Hu, Q.; Sun, Y.; et al. Cytokine release syndrome in COVID-19: A major mechanism of morbidity and mortality. Int. Rev. Immunol. 2022, 41, 217–230.
- Cosentino, F.; Moscatt, V.; Marino, A.; Pampaloni, A.; Scuderi, D.; Ceccarelli, M.; Benanti, F.; Gussio, M.; Larocca, L.; Boscia, V.; et al. Clinical characteristics and predictors of death among hospitalized patients infected with SARS-CoV-2 in Sicily, Italy: A retrospective observational study. Biomed. Rep. 2022, 16, 34.
- Cheng, V.C.; Edwards, K.M.; Gandhi, R.; Muller, W.J. IDSA Guidelines on the Treatment and Management of Patients with COVID-19. Available online: www.idsociety.org/COVID19guidelines (accessed on 8 May 2022).
- COVID-19 Treatments|NIH COVID-19 Research. Available online: https://covid19.nih.gov/treatments-and-vaccines/COVID-19-treatments#covid19-treatments (accessed on 26 March 2022).
- Della-Torre, E.; Campochiaro, C.; Cavalli, G.; De Luca, G.; Napolitano, A.; La Marca, S.; Boffini, N.; Da Prat, V.; di Terlizzi, G.; Lanzillotta, M.; et al. Interleukin-6 blockade with sarilumab in severe COVID-19 pneumonia with systemic hyperinflammation: An open-label cohort study. Ann. Rheum. Dis. 2020, 79, 1277–1285.
- Gremese, E.; Cingolani, A.; Bosello, S.L.; Alivernini, S.; Tolusso, B.; Perniola, S.; Landi, F.; Pompili, M.; Murri, R.; Santoliquido, A.; et al. Sarilumab use in severe SARS-CoV-2 pneumonia. EClinicalMedicine 2020, 27, 100553.
- Benucci, M.; Giannasi, G.; Cecchini, P.; Gobbi, F.L.; Damiani, A.; Grossi, V.; Infantino, M.; Manfredi, M. COVID-19 pneumonia treated with Sarilumab: A clinical series of eight patients. J. Med. Virol. 2020, 92, 2368–2370.
- The WHO Rapid Evidence Appraisal for COVID-19 Therapies (REACT) Working Group. Association Between Administration of IL-6 Antagonists and Mortality Among Patients Hospitalized for COVID-19: A Meta-analysis. JAMA 2021, 326, 499–518.
- Gordon, A.C.; Mouncey, P.R.; Al-Beidh, F.; Rowan, K.M.; Nichol, A.D.; Arabi, Y.M.; Annane, D.; Beane, A.; Van Bentum-Puijk, W.; Berry, L.R.; et al. Interleukin-6 Receptor Antagonists in Critically Ill Patients with COVID-19. N. Engl. J. Med. 2021, 384, 1491–1502.
- Lescure, F.X.; Honda, H.; Fowler, R.A.; Lazar, J.S.; Shi, G.; Wung, P.; Patel, N.; Hagino, O.; Bazzalo, I.J.; Casas, M.M.; et al. Sarilumab in patients admitted to hospital with severe or critical COVID-19: A randomised, double-blind, placebo-controlled, phase 3 trial. Lancet Respir. Med. 2021, 9, 522–532.
- Mariette, X.; Hermine, O.; Tharaux, P.L.; Resche-Rigon, M.; Porcher, R.; Ravaud, P.; Bureau, S.; Dougados, M.; Tibi, A.; Azoulay, E.; et al. Sarilumab in adults hospitalised with moderate-to-severe COVID-19 pneumonia (CORIMUNO-SARI-1): An open-label randomised controlled trial. Lancet Rheumatol. 2022, 4, e24–e32.
- Berardicurti, O.; Ruscitti, P.; Ursini, F.; D’Andrea, S.; Ciaffi, J.; Meliconi, R.; Iagnocco, A.; Cipriani, P.; Giacomelli, R. Mortality in tocilizumab-treated patients with COVID-19: A systematic review and meta-analysis. Clin. Exp. Rheumatol. 2020, 38, 1247–1254.
- Lok, L.S.C.; Farahi, N.; Juss, J.K.; Loutsios, C.; Solanki, C.K.; Peters, A.M.; Donaldson, F.; Porter-Brown, B.; Chilvers, E.R. Effects of tocilizumab on neutrophil function and kinetics. Eur. J. Clin. Investig. 2017, 47, 736–745.
- Wright, H.L.; Cross, A.L.; Edwards, S.W.; Moots, R.J. Effects of IL-6 and IL-6 blockade on neutrophil function in vitro and in vivo. Rheumatology 2014, 53, 1321–1331.
- Chamlagain, R.; Shah, S.; Sharma Paudel, B.; Dhital, R.; Kandel, B. Efficacy and Safety of Sarilumab in COVID-19: A Systematic Review. Interdiscip. Perspect. Infect. Dis. 2021, 2021, 8903435.
- Han, Q.; Guo, M.; Zheng, Y.; Zhang, Y.; De, Y.; Xu, C.; Zhang, L.; Sun, R.; Lv, Y.; Liang, Y.; et al. Current Evidence of Interleukin-6 Signaling Inhibitors in Patients With COVID-19: A Systematic Review and Meta-Analysis. Front. Pharmacol. 2020, 11, 2119.
- Erdem, H.; Hargreaves, S.; Ankarali, H.; Caskurlu, H.; Ceviker, S.A.; Bahar-Kacmaz, A.; Meric-Koc, M.; Altindis, M.; Yildiz-Kirazaldi, Y.; Kizilates, F.; et al. Managing adult patients with infectious diseases in emergency departments: International ID-IRI study. J. Chemother. 2021, 33, 302–318.
- El-Sokkary, R.; Uysal, S.; Erdem, H.; Kullar, R.; Pekok, A.U.; Amer, F.; Grgić, S.; Carevic, B.; El-Kholy, A.; Liskova, A.; et al. Profiles of multidrug-resistant organisms among patients with bacteremia in intensive care units: An international ID-IRI survey. Eur. J. Clin. Microbiol. Infect. Dis. 2021, 40, 2323–2334.
- Celesia, B.M.; Marino, A.; Del Vecchio, R.F.; Bruno, R.; Palermo, F.; Gussio, M.; Nunnari, G.; Cacopardo, B. Is it safe and cost saving to defer the CD4+ cell count monitoring in stable patients on ART with more than 350 or 500 cells/µL? Mediterr. J. Hematol. Infect. Dis. 2019, 11, e2019063.
- Marino, A.; Zafarana, G.; Ceccarelli, M.; Cosentino, F.; Moscatt, V.; Bruno, G.; Bruno, R.; Benanti, F.; Cacopardo, B.; Celesia, B.M. Immunological and Clinical Impact of DAA-Mediated HCV Eradication in a Cohort of HIV/HCV Coinfected Patients: Monocentric Italian Experience. Diagnostics 2021, 11, 2336.
- Silvestris, N.; Munafò, A.; Brunetti, O.; Burgaletto, C.; Scucces, L.; Bernardini, R. On the Management of Drug Interactions in the Course of Concomitant Treatments for COVID-19 and Antineoplastic Agents. Front. Oncol. 2020, 10, 1340.
- Boyce, E.G.; Rogan, E.L.; Vyas, D.; Prasad, N.; Mai, Y. Sarilumab: Review of a Second IL-6 Receptor Antagonist Indicated for the Treatment of Rheumatoid Arthritis. Ann. Pharmacother. 2018, 52, 780–791.
- KEVZARA® (Sarilumab) for Rheumatoid Arthritis. Available online: https://www.kevzara.com/ (accessed on 7 May 2022).
- Lee, E.B.; Daskalakis, N.; Xu, C.; Paccaly, A.; Miller, B.; Fleischmann, R.; Bodrug, I.; Kivitz, A. Disease–Drug Interaction of Sarilumab and Simvastatin in Patients with Rheumatoid Arthritis. Clin. Pharmacokinet. 2017, 56, 607–615.
- Marino, A.; Munafò, A.; Zagami, A.; Ceccarelli, M.; Di Mauro, R.; Cantarella, G.; Bernardini, R.; Nunnari, G.; Cacopardo, B. Ampicillin plus ceftriaxone regimen against enterococcus faecalis endocarditis: A literature review. J. Clin. Med. 2021, 10, 4594.
- Burgaletto, C.; Brunetti, O.; Munafò, A.; Bernardini, R.; Silvestris, N.; Cantarella, G.; Argentiero, A. Lights and shadows on managing immune checkpoint inhibitors in oncology during the COVID-19 era. Cancers 2021, 13, 1906.
- Marino, A.; Cosentino, F.; Ceccarelli, M.; Moscatt, V.; Pampaloni, A.; Scuderi, D.; D’andrea, F.; Venanzi Rullo, E.; Nunnari, G.; Benanti, F.; et al. Entecavir resistance in a patient with treatment-naïve hbv: A case report. Mol. Clin. Oncol. 2021, 14, 113.
- Celesia, B.M.; Marino, A.; Borracino, S.; Arcadipane, A.F.; Pantò, G.; Gussio, M.; Coniglio, S.; Pennisi, A.; Cacopardo, B.; Panarello, G. Successful extracorporeal membrane oxygenation treatment in an acquired immune deficiency syndrome (AIDS) patient with acute respiratory distress syndrome (ARDS) complicating pneumocystis jirovecii pneumonia: A challenging case. Am. J. Case Rep. 2020, 21, e919570.
- Koritala, T.; Pattan, V.; Tirupathi, R.; Rabaan, A.A.; Al Mutair, A.; Alhumaid, S.; Adhikari, R.; Deepika, K.; Jain, N.K.; Bansal, V.; et al. Infection risk with the use of interleukin inhibitors in hospitalized patients with COVID-19: A narrative review. Infez. Med. 2021, 29, 495–503.
