Nanotechnologies have attracted increasing attention in their application in medicine, especially in the development of new drug delivery systems. With the help of nano-sized carriers, drugs can reach specific diseased areas, prolonging therapeutic efficacy while decreasing undesired side-effects. In addition, recent nanotechnological advances, such as surface stabilization and stimuli-responsive functionalization have also significantly improved the targeting capacity and therapeutic efficacy of the nanocarrier assisted drug delivery system.
- nanomedicine
- nanocarriers
- drug delivery
1. Introduction
Nanotechnology has emerged to be an area of active investigation, especially in its applications in medicine [1]. The nanoscale manipulation allows optimal targeting and delivery as well as the controllable release of drugs or imaging agents [2]. Among all the applications of nanotechnology in medicine, nanocarrier assisted drug delivery system has attracted significant research interest due to its great translational value. The small size of the nanocarriers can help drugs overcome certain biological barriers to reach diseased areas [3][4]. Taking advantage of different nano-sized materials and various structures, nanocarriers can help poorly soluble drugs become more bioavailable and protect easily degraded therapeutics from degradation [5][6]. In addition, the modifiable surfaces of nanocarriers also expand their usability in different biomedical applications, especially in targeted therapy [7]. Indeed, their modification can not only stabilize but also functionalize them to be responsive to different stimuli, improving the therapeutic efficacy [7].
2. Strategies to Enhance Drug Delivery Efficiency
In nanomedicine, one common route for the carried-on drugs to reach the diseased area is to passively diffuse out of the nanocarriers that have accumulated in the diseased tissue [8]. As for adding targeting capacity to the nanocarriers, especially in cancer therapy, a common approach is surface addition of ligands that are specific to receptors overexpressed in certain cancer cells [9]. In an effort to develop smarter nanocarriers that can further improve the targeting efficiency and on-demand drug release, various stimuli-responsive nanocarriers are currently under development [10]. In the following sections, we will discuss the features and applications of some of the stimuli-responsive nanocarriers (Figure 1).
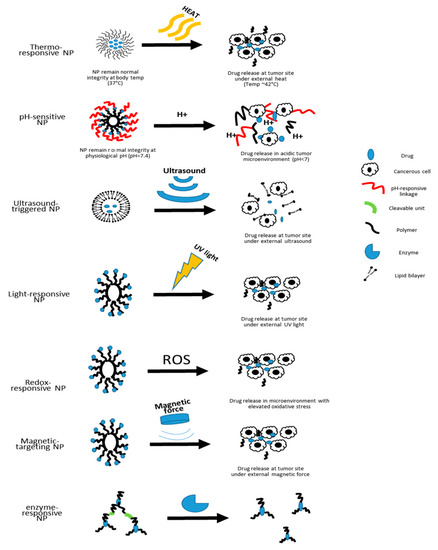
Figure 1. Schematic illustration of the drug-releasing mechanisms of different types of nanocarriers.
Currently, the stimuli-responsive nanocarriers can be activated by either exogenous stimuli, such as variations in temperature, magnetic field, ultrasound intensity, light or electric pulses, or endogenous stimuli, such as changes in pH, enzyme concentration or redox gradients [10]. The specific design of nanoscale stimuli-responsive systems enables the controlled drug biodistribution in response to specific exogenous or endogenous stimuli [11]. Through the stimuli responsiveness, on-demand drug release could be achievable [11][12][13].
2.1. Thermo-Responsive
Thermo-responsive nanocarriers are among the most investigated nanocarriers in cancer therapy for solid tumors [11]. The working concept is that thermo-responsive nanocarriers are able to retain their payload at 37 °C, the physiological body temperature, but rapidly release their payload at the heated tumor area (~40 to 42 °C) [11][14]. As nanotechnology advances, polymeric nanocarriers that exhibit lower critical solution temperature (LCST) and upper critical solution temperature (UCST) can have better control of drug release [15]. This strategy is helpful in the treatment of solid tumors as drugs can be loaded at 37 °C, and depending on the LCST or UCST, drugs can then be released by either cooling or heating the tumor after injection [15]. Based on the existing thermo-responsive technology, an additional pulsatile drug delivery system has been introduced by embedding thermo-responsive polymers to liquid crystals to achieve programmed drug release in concordance with the circadian rhythm [16].
2.2. pH-Responsive
As pH values vary in different biological compartments, pH-responsive nanocarriers will allow better control of site-specific drug release [17]. There are two main strategies that exist for the development of pH-sensitive nanocarriers. One is using polymers functionalized with ionizable groups that can undergo a conformational change upon encountering environmental pH change, and the other is the incorporation of acid-sensitive bonds that break in an acidic environment for drug release [11][18]. Due to the acidic microenvironment of tumor sites, pH-responsive nanocarriers rise to be a useful strategy for targeted cancer therapy [19]. The intracellular pH values also differ from the ones in the extracellular matrix [20].
A wide range of nanocarriers can be equipped with pH-responsivity [21]. For example, vaccines for hepatitis B virus are under development using pH-responsive liposomes to achieve cytosolic drug release [22]. Polymeric micelles that are functionalized to be pH-responsive are under active investigation for their application in chemotherapy [23][24]. To overcome certain disadvantages of some pH-sensitive polymers, such as uncontrolled drug-loading or drug-releasing rate as well as undesired toxicity, non-polymer pH-sensitive carbon dots were also developed for cancer therapy [25]. In addition, recently surface modified pH-responsive SWCTs have also been developed to co-deliver anti-cancer drugs and genes [26].
2.3. Ultrasound-Triggered
The ultrasound-triggered drug-releasing approach is an appealing on-demand drug-releasing strategy because of their non-invasiveness as well as the controllable frequency and duration in order to regulate the depth of tissue penetration [11]. Ultrasound generated mechanical forces can transform nanodroplets (such as liposomes) to nano-bubbles [11]. These ultrasound-genic nanobubbles increase the efficiency of delivering payloads to neighboring cells [11]. Prabhakar et al. have recently developed a nanobubble liposome complex that can be ultrasound triggered to deliver both imaging agents and anti-cancer drugs, suggesting a promising future for the theranostic application of ultrasound-triggered nanocarriers [27]. However, despite the fact that ultrasound is non-invasive, the frequency applied in order to trigger drug release differs from conventional settings for clinical imaging purposes. Such differences raise the concern of potential mechanical induced cell damage [28]. Another limitation is that the ultrasound beam may be attenuated by the hard tissues and certain tissue associated movements [29].
2.4. Light-Responsive
Another non-invasive and controllable approach for drug release is to incorporate light-responsive materials in the nanocarriers. Under certain wavelengths of light, these nanocarriers can either disassemble for drug release or shrink in size for deeper tissue penetration [11]. Wang et al. developed a near-infrared (NIR) light-responsive polymeric nanocarrier by incorporating selenium that can rapidly dissociate within minutes post NIR light exposure due to reactive oxygen species (ROS)-mediated selenium oxidation [30]. Such irreversible dissociation of nanocarriers promotes continuous drug release [30]. With the help of light-responsive nanomaterials, a high degree of spatiotemporal precision can be achieved, but the safety of light-responsive nanocarriers are still not well defined [31][32]. Specifically, the irreversible change of these nanocarriers raises concerns for the safety of byproducts [31].
2.5. Redox-Responsive
Given that oxidative stress has been found to be elevated in the pathogenesis of many diseases, another type of stimuli-responsive nanocarriers that have attracted significant interest are the redox-responsive nanocarriers [33][34][35]. The tumor microenvironment has certain features that are different from healthy tissues. For example, there is a significant elevation in the concentrations of glutathione and ROS in tumor microenvironments than in normal tissues [36]. With the help of redox-responsive polymers, these nanocarriers can significantly increase the concentration of drugs released in the diseased area [36][37].
Similarly, in diabetes, hyperglycemia induces cellular hypoxia through mitochondrial ROS production [37][38]. In order to benefit diabetic patients, Gu’s group has been advancing the painless microneedle patch using hypoxia-responsive nanoparticles that release insulin in a hypoxemic microenvironment. They first developed microneedle-array patches loaded with hypoxia-sensitive vesicles to provide fast glucose-responsive insulin delivery [39]. Subsequently, they improved their nanoparticles using H2O2-responsive polymeric vesicles integrated with transcutaneous patches for glucose-mediated insulin delivery [40]. Combining both technologies, they developed a new nanovesicle that is responsive to both hypoxic environment and H2O2 in order to enhance insulin delivery [41].
As ROS and redox signaling also play important roles in ischemia/reperfusion (I/R) injury, redox-responsive nanocarriers delivering anti-oxidant to the injured site could also potentially ameliorate I/R injury in the tissues [42][43][44]. For this purpose, Kang and colleagues have developed ROS-responsive nanocarriers for their application in various I/R injuries [45][46][47][48][49]. Recently, Elkassih et al. also developed degradable redox-responsive disulfide cross-linked nanogel drug carriers to decrease cytotoxicity and to increase drug uptake in areas with increased oxidative stress [50].
2.6. Magnetic Targeting
Nanocarriers can also be modified to be responsive to magnetic force. In this approach, the therapeutic agents are attached to or encapsulated in magnetic nanocarriers, which are often made of functionalized polymers [51]. Among all the candidate nanomaterials, the biocompatible superparamagnetic iron oxide nanoparticles (SPIONs) with modifications are the most widely used as part of the magnetic nanocarriers [52]. These nanocarriers are then injected into the bloodstream near the target site [51]. When the magnetic fields are applied over the target site, the magnetic force will drive the accumulation and release of the payloads [51]. Previous studies have shown that magnetophoresis can enhance the accumulation and penetration of nanocarriers into solid tumors [53]. However, the clinical translation of this approach is difficult due to its low efficacy and uncontrollable magnetic nanoparticle distribution [54][55]. One of the biggest challenges with the use of magnetic fields is that because the magnetic force falls off significantly with distance, the target sites are limited to the near-surface of the body [51]. Some groups attempted to overcome this challenge by implanting magnets within the body in order to reach deep tissue penetration [51]. However, the use of permanent magnets itself has become a limitation for the clinical translation of this approach [56][57]. In addition, the lack of real-time imaging and the difficulties in controlling magnetic force for precise delivery are also factors that limit its clinical use [56][57]. Using the oppositely polarized magnets, Liu et al. aimed to improve the weak magnetic force in older generations of magnetophoresis that can only be used to treat superficial tumors [53]. This method increased the penetration by five-fold and accumulation by three-fold of magnetic nanoparticles within solid tumors compared to passive enhanced permeability and retention effect [53].
2.7. Enzyme-Responsive Nanocarriers
In addition to the environmental stimuli such as pH and oxidative stress, enzymes that present within the cellular system can also be utilized as a trigger for targeted drug release [58]. Nanocarriers such as polymeric micelles, liposomes, and dendrimers are often functionalized by attaching cleavable peptides to the surface that are tailored to specific enzymes present in the targeting tissues [59][60][61][62]. In order to achieve the goal of targeted cargo delivery, several releasing mechanisms can be employed. In functionalized enzyme-responsive liposomes, the enzymes can either directly perturb the lipid bilayer structure, or cleave a lipopeptide or lipopolymer incorporated in the bilayer to achieve destabilization of the nanocarrier [60]. In addition, targeted enzymes can also remove the shielding polymers from the surface to increase cellular uptake, or activate a prodrug in the nanocarriers [60].
Based on the purpose of the treatment, a variety of enzymes can be utilized to trigger local drug delivery. For example, one unique feature of the cancerous tissues is the overexpression of a type of extracellular proteolytic enzyme, called the matrix metalloproteinases (MMPs) [63]. Based on this feature, several MMP-responsive nanocarriers have been developed as one approach for the targeted cancer treatment [64]. For targeted anti-inflammatory treatment, a protease secreted by neutrophils, called human neutrophil elastase (HNE) is exploited as a biological cue for controlled drug release from nanocarriers equipped with HNE-sensitive peptide linkers [65][66]. This approach can significantly increase the sensitivity of targeted anti-inflammatory treatment, as neutrophils are the first cells recruited to inflammatory sites [67]. Enzyme-responsive nanocarriers can also be applied to regulate coagulation locally. For this purpose, Bhat et al. developed a thrombin-responsive mesoporous silica nanoparticle (MSN) that is loaded with an anticoagulant drug and capped with a peptide containing a thrombin-specific cleavage site [68]. When the coagulation cascade is triggered, active thrombin can degrade the capping peptide sequence on the nanocarrier and release the anticoagulant locally [68]. Due to the site-specificity of thrombin, thrombin-responsive nanocarriers possess the advantage of spatiotemporal specificity in anti-thrombotic drug delivery [69].
While significant progress has been made in the development of enzyme-responsive nanocarriers, several challenges remain to be addressed for the wider application of this approach. For example, there are various enzyme subtypes existing in the biological system that share similar cleavage sites [61]. In addition, current imaging technologies are yet to satisfy the need for the confirmation of controlled drug release at the targeted areas [61].
2.8. Multimodal Nanocarriers
Despite the fact that stimuli-responsive nanocarriers have already significantly improved targeted drug delivery, they still face certain limitations. For example, stimuli-responsive nanocarriers utilizing external factors, such as ultrasound, light, heat, and magnetic forces, are limited to targets with known target localization. In addition, human physiology is a complex system. Nanocarriers that are responsive to pH and oxidative stress might potentially release cargoes at areas other than the diseased area because those non-targeted areas also share similarly elevated pH or oxidative stress, due to conditions such as metabolic acidosis, vascular occlusion, or other inflammations. By combining different stimuli-responsive properties, multimodal nanocarriers can improve the efficacy of nanomedicine, especially in cancer therapy [70]. Recently, nanocarriers that possess both thermo- and pH-responsiveness are being developed to treat certain cancers [71]. Using this dual stimuli-responsive system, Hiruta et al. developed a polymeric micelle for anti-cancer drug delivery that can be selectively up-taken with external thermal stimulation and effectively release its cargo at endosomal pH [72]. As each stimuli-responsive nanocarrier has different advantages and limitations (Table 1), with additional stimuli responsiveness, nanocarriers can be more attuned to the stimuli changes and exert therapeutic effects in a more precise fashion. Recently, a programmable polymer library for stimuli-responsive nanocarriers containing logic gates has also been developed to gather systemic information in order to achieve precision medicine [73][74].
Table 1. Summary of different stimuli-responsive nanocarriers.
| Stimuli Type | Advantages | Challenges/Limitations | References |
|---|---|---|---|
| Thermo-Responsive |
|
|
[11][14][15] |
| pH-Sensitive |
|
|
[11][17][18][19][21][22][23][24][25][26] |
| Ultrasound-Triggered |
|
|
[11][27][28][29] |
| Light-Responsive |
|
|
[30][31][32] |
| Redox-Responsive |
|
|
[34][36][37] |
| Magnetic-Targeting |
|
|
[53][54][55][56][57] |
| Enzyme-Responsive |
|
|
[58][59][60][61][62][64] |
Another example in the anticancer drug delivery system is the nanocarriers designed to respond to the tumor microenvironment which can switch its size and morphology in response to the acidic tumor microenvironment and near-infrared laser irradiation [75]. Jia et al. have recently developed a smart nanodrug that can switch its size and morphology in response to the acidic tumor microenvironment and near-infrared laser irradiation to effectively ablate a tumor, inhibiting tumor metastasis [75]. This nanodrug is assembled by a cytolytic peptide, an NIR-absorbing molecule, and a tumor-targeting polymer [75]. Under normal physiological environment, the assembly is a negatively charged nanosphere about 50 nm in size [75]. The acidic tumor microenvironment triggers the transformation of the nanodrug into net-like nanofibers [75]. The net-like structure helps to limit the mobility of tumor cells and also prolongs the drug retention time [75]. During photothermal therapy, the nanocomplex can be photodegraded into smaller nanospheres about 25 nm in size to allow deeper tumor penetration of the drug [75].
When combining gene and photothermal therapy, synergistic therapeutic effects have been observed, suggesting the advantages of multimodal nanomedicine [76]. In a similar effort for on-demand drug release, Deng et al. developed a new liposomal drug delivery platform that can control payload release only when triggered by x-ray radiation [77]. This liposome incorporates gold nanoparticles with a photosensitizer called verteporfin [77]. Under radiation, the photosensitizer produces singlet oxygen to destabilize the liposomal membrane, allowing payload to be released from the liposome, while the gold nanoparticles are used for radiation enhancement [77]. This platform could provide synergistic therapeutic effects in chemotherapy when combined with radiotherapy [77]. The main drawback of this platform design, however, is that the photosensitizer, as well as the gold nanoparticle incorporated, generate a certain level of ROS in the tissues that could be damaging [77].
Continued efforts are being made to advance theranostic nanocarriers. By functionalizing the surface of FDA-approved iron oxide nanoparticles with an imaging contrast agent and a peptide activatable by a tumor-specific enzyme new theranostic nanocarriers can achieve enzyme-specific drug delivery at the site of the tumor and simultaneous MRI imaging [78]. Conjugated polymer nanosystems are developed to combine diagnostic imaging together with photothermal therapy and drug delivery in cancer therapy [79]. Li et al. have also functionalized the carbon quantum dots so that they structurally mimic large amino acids that can selectively accumulate at tumor sites for both imaging and drug delivery purposes [80]. A recent review by Riccardi et al. summarizes the development of different nanocarriers with various decorations (or functionalization) for the improvement in bioavailability, pharmacokinetics, and specificity of anticancer ruthenium-based drugs [81].
2.9. Bioinspired Nanocarriers
Besides using biodegradable and biocompatible synthetic materials for the development of nanocarriers, other bioinspired natural materials are also being explored for their application in drug delivery systems. One strategy is to develop biomimetic nanoparticles by using the cell membrane as camouflage [82]. For example, utilizing the concept of biomimetic functionalization of the nanocarriers, Liu et al. integrated a red blood cell (RBC) membrane vesicle with near-infrared persistent luminescence nanophosphors to ensure the nanocarriers can bypass macrophage uptake and systemic clearance to improve circulation time for bio-imaging and drug delivery [83]. Santos’ group further engineered the isolated RBC membranes to form nanoerythrosomes (NERs), i.e., derivatives of RBCs with an average diameter of 100 nm, for drug delivery [84]. Similarly, using a cancer membrane as camouflage, a tumor homing nanocarrier that carries imaging and/or therapeutic moieties can also provide a new platform for targeted drug delivery [85][86]. Compared to RBC membranes, cancer cell membranes alone are unstable and have insufficient drug entrapment, thus cannot act as an autonomous drug delivery system without the support of other nanomaterials [84]. Therefore, Balasubramanian et al. combined cancer cell membrane material with porous silicon nanoparticles to develop nanocarriers that serve as artificial organelles in order to supplement cellular functions under oxidative stress [87].
In addition to using bioinspired nanomaterials to develop new nanocarriers, existing nanocarriers can also be modified in a bioinspired fashion. Zhang et al. recently developed a liposomal nanocarrier with a modified surface that has a short nontoxic peptide derived from Aβ1-42 that specifically interacts with the lipid-binding domain of apolipoproteins [88]. These nanocarriers absorb plasma apolipoproteins A1, E, and J, resulting in the exposure of the receptor-binding domain of apolipoproteins to achieve brain-targeting drug delivery [88].
3. Strategies to Enhance Therapeutic Efficacy Using Different Payloads
3.1. Cell Replacement
Nanocarriers can also deliver other payloads such as cells to the diseased area. Such an approach can be applied in cell replacement therapies. For example, for type I diabetic patients, islet transplantation is a promising treatment [89]. However, it is limited by the shortage of donors and the significant side effect of immunosuppression [89]. In order to overcome this challenge, recent advancements in nanotechnology enables the encapsulation of the islet in immune-isolating membranes with chemical modifications for transplantation [90][89][91]. Using mesenchymal stem cells (MSCs) as cell-based drug delivery vectors for tumor-homing cancer treatment has also shown some promising results [92]. However, the broad biodistribution of MSCs also raises concerns for toxicity to non-target peripheral tissues [92]. A wider application of nanocarrier assisted cell replacement therapy still requires more investigation.
3.2. Gene Therapy
Another area of investigation that is currently on the horizon is the concept of nanocarrier assisted gene therapy. One important strategy in gene therapy is the use of small interfering RNA (siRNA) to silence disease-causing genes [93]. Currently, there are more than 20 siRNA based therapies in clinical trials [93]. However, there are two main concerns of RNA interference (RNAi) potency and specificity [94]. Transporting siRNA across the cell membrane is challenging due to its anionic property [95]. In addition, naked siRNA has immunostimulatory effects and is easily degraded in the bloodstream [95]. Viral vectors have long been used to deliver siRNA, however, they have drawbacks, such as being immunogenic and cytotoxic [96]. Being non-viral and equipped with targeting capacity, nanocarriers have drastically helped the emergence of RNAi therapeutics [93]. Different nanocarriers are being applied in the delivery of siRNA, such as nucleotides, lipids, and polymers [93][95]. The biocompatibility and design flexibility of nanocarriers allow better control of siRNA delivery to achieve desired gene knockdown efficiency [94][95][96].
Among all the nanocarriers under development for their potential to assist in gene delivery, lipid nanocarriers have shown the most promising results in the clinical translation of siRNA therapy [97][98][99][100][101]. Lipid nanoparticles can protect siRNA from degradation, and facilitate endocytosis and endosomal escape [100]. The first nanoparticle assisted targeted RNAi delivery in humans was reported in 2009 [102]. In this study, the nanocarriers were designed to passively accumulate and permeate in solid tumors [102]. With the help of CRISPR-Cas9 technology, new lipid nanocarriers capable of selective organ targeting (SORT) have been developed [103]. These nanocarriers can target extrahepatic tissues with the aid of targeting molecules for selective organs, revolutionizing tissue-specific gene editing [103]. Lipid nanocarrier assisted nucleic acid delivery is also under active investigation for its potential use in the development of prophylactic vaccines [104]. In addition, recently another type of nanocarrier similar to the liposome, called noisome, has also been developed [105]. Niosomes are self-assembled vesicles made up of single-chain non-ionic surfactants combined with appropriate amounts of cholesterol or other lipids [105]. Similar to liposomes, niosomes are capable of carrying hydrophilic or lipophilic drugs but are more stable, less expensive, and easier to manipulate [105]. They have the potential to be an alternative gene delivery system.
4. Conclusions
In this review, we discussed different types of nanocarriers applied in the drug delivery system. The use of nanocarriers, whether with single-featured nanomaterials and/or structures or in a hybrid fashion, has largely expanded the platform for drug delivery. In addition, the functionalization of nanocarriers that enables them to be sensitive to different stimuli (such as pH, heat, light, or oxidative stress) has further broadened their capacities for the delivery of different therapeutics. While tremendous advancements have been achieved in this field over the years, challenges remain to overcome in order to improve the translational value of current nanomedicine research. Specifically, the long-term safety profiles of these nanocarriers need to be carefully evaluated. Looking ahead, the nanocarrier assisted-delivery system holds great potential in improving therapeutic efficacy.
This entry is adapted from the peer-reviewed paper 10.3390/pharmaceutics12090837
References
- Zhang, L.; Gu, F.X.; Chan, J.M.; Wang, A.Z.; Langer, R.S.; Farokhzad, O.C. Nanoparticles in Medicine: Therapeutic Applications and Developments. Clin. Pharmacol. Ther. 2008, 83, 761–769.
- Shi, J.; Votruba, A.R.; Farokhzad, O.C.; Langer, R. Nanotechnology in Drug Delivery and Tissue Engineering: From Discovery to Applications. Nano Lett. 2010, 10, 3223–3230.
- Hoshyar, N.; Gray, S.; Han, H.; Bao, G. The Effect of Nanoparticle Size on in Vivo Pharmacokinetics and Cellular Interaction. Nanomedicine 2016, 11, 673–692.
- Chenthamara, D.; Subramaniam, S.; Ramakrishnan, S.G.; Krishnaswamy, S.; Essa, M.M.; Lin, F.H.; Qoronfleh, M.W. Therapeutic Efficacy of Nanoparticles and Routes of Administration. Biomater. Res. 2019, 23, 22.
- Gunasekaran, T.; Haile, T.; Nigusse, T.; Dhanaraju, M.D. Nanotechnology: An Effective Tool for Enhancing Bioavailability and Bioactivity of Phytomedicine. Asian Pac. J. Trop. Biomed. 2014, 4, S1–S7.
- Anselmo, A.C.; Gokarn, Y.; Mitragotri, S. Non-Invasive Delivery Strategies for Biologics. Nat. Rev. Drug Discov. 2018, 18, 19–40.
- Shen, Z.; Nieh, M.P.; Li, Y. Decorating Nanoparticle Surface for Targeted Drug Delivery: Opportunities and Challenges. Polymers 2016, 8, 83.
- Rizvi, S.A.A.; Saleh, A.M. Applications of Nanoparticle Systems in Drug Delivery Technology. Saudi Pharm. J. 2018, 26, 64–70.
- Pardo, J.; Peng, Z.; Leblanc, R.M. Cancer Targeting and Drug Delivery Using Carbon-Based Quantum Dots and Nanotubes. Molecules 2018, 23, 378.
- Li, F.; Qin, Y.; Lee, J.; Liao, H.; Wang, N.; Davis, T.P.; Qiao, R.; Ling, D. Stimuli-Responsive Nano-Assemblies for Remotely Controlled Drug Delivery. J. Control. Release 2020, 322, 566–592.
- Mura, S.; Nicolas, J.; Couvreur, P. Stimuli-Responsive Nanocarriers for Drug Delivery. Nat. Mater. 2013, 12, 991–1003.
- Torchilin, V.P. Multifunctional, Stimuli-Sensitive Nanoparticulate Systems for Drug Delivery. Nat. Rev. Drug Discov. 2014, 13, 813–827.
- Karimi, M.; Ghasemi, A.; Sahandi Zangabad, P.; Rahighi, R.; Moosavi Basri, S.M.; Mirshekari, H.; Amiri, M.; Shafaei Pishabad, Z.; Aslani, A.; Bozorgomid, M.; et al. Smart Micro/Nanoparticles in Stimulus-Responsive Drug/Gene Delivery Systems. Chem. Soc. Rev. 2016, 45, 1457–1501.
- Sánchez-Moreno, P.; de Vicente, J.; Nardecchia, S.; Marchal, J.A.; Boulaiz, H. Thermo-Sensitive Nanomaterials: Recent Advance in Synthesis and Biomedical Applications. Nanomaterials 2018, 8, 935.
- Bordat, A.; Boissenot, T.; Nicolas, J.; Tsapis, N. Thermoresponsive Polymer Nanocarriers for Biomedical Applications. Adv. Drug Deliv. Rev. 2019, 138, 167–192.
- Lin, S.Y. Thermoresponsive Gating Membranes Embedded with Liquid Crystal(s) for Pulsatile Transdermal Drug Delivery: An Overview and Perspectives. J. Control. Release 2020, 319, 450–474.
- Zhu, Y.J.; Chen, F. PH-Responsive Drug-Delivery Systems. Chem. Asian J. 2015, 10, 284–305.
- Deirram, N.; Zhang, C.; Kermaniyan, S.S.; Johnston, A.P.R.; Such, G.K. PH-Responsive Polymer Nanoparticles for Drug Delivery. Macromol. Rapid Commun. 2019, 40, 1800917.
- Xiong, H.; Wu, Y.; Jiang, Z.; Zhou, J.; Yang, M.; Yao, J. PH-Activatable Polymeric Nanodrugs Enhanced Tumor Chemo/Antiangiogenic Combination Therapy through Improving Targeting Drug Release. J. Colloid Interface Sci. 2019, 536, 135–148.
- Wu, W.; Luo, L.; Wang, Y.; Wu, Q.; Dai, H.B.; Li, J.S.; Durkan, C.; Wang, N.; Wang, G.X. Endogenous PH-Responsive Nanoparticles with Programmable Size Changes for Targeted Tumor Therapy and Imaging Applications. Theranostics 2018, 8, 3038–3058.
- Goyal, A.K.; Rath, G.; Faujdar, C.; Malik, B. Application and Perspective of PH-Responsive Nano Drug Delivery Systems. In Applications of Targeted Nano Drugs and Delivery Systems; Elsevier: Amsterdam, The Netherlands, 2019; pp. 15–33.
- Hu, F.; Yue, H.; Lu, T.; Ma, G. Cytosolic Delivery of HBsAg and Enhanced Cellular Immunity by PH-Responsive Liposome. J. Control. Release 2020, 324, 460–470.
- Liao, Z.S.; Huang, S.Y.; Huang, J.J.; Chen, J.K.; Lee, A.W.; Lai, J.Y.; Lee, D.J.; Cheng, C.C. Self-Assembled PH-Responsive Polymeric Micelles for Highly Efficient, Noncytotoxic Delivery of Doxorubicin Chemotherapy to Inhibit Macrophage Activation: In Vitro Investigation. Biomacromolecules 2018, 19, 2772–2781.
- Zhou, X.X.; Jin, L.; Qi, R.Q.; Ma, T. Ph-Responsive Polymeric Micelles Self-Assembled from Amphiphilic Copolymer Modified with Lipid Used as Doxorubicin Delivery Carriers. R. Soc. Open Sci. 2018, 5, 171654.
- Seo, J.; Lee, J.; Lee, C.B.; Bae, S.K.; Na, K. Nonpolymeric PH-Sensitive Carbon Dots for Treatment of Tumor. Bioconjug. Chem. 2019, 30, 621–632.
- Cao, Y.; Huang, H.Y.; Chen, L.Q.; Du, H.H.; Cui, J.H.; Zhang, L.W.; Lee, B.J.; Cao, Q.R. Enhanced Lysosomal Escape of PH-Responsive Polyethylenimine-Betaine Functionalized Carbon Nanotube for the Codelivery of Survivin Small Interfering RNA and Doxorubicin. ACS Appl. Mater. Interfaces 2019, 11, 9763–9776.
- Prabhakar, A.; Banerjee, R. Nanobubble Liposome Complexes for Diagnostic Imaging and Ultrasound-Triggered Drug Delivery in Cancers: A Theranostic Approach. ACS Omega 2019, 4, 15567–15580.
- Canavese, G.; Ancona, A.; Racca, L.; Canta, M.; Dumontel, B.; Barbaresco, F.; Limongi, T.; Cauda, V. Nanoparticle-Assisted Ultrasound: A Special Focus on Sonodynamic Therapy against Cancer. Chem. Eng. J. 2018, 340, 155–172.
- Tharkar, P.; Varanasi, R.; Wong, W.S.F.; Jin, C.T.; Chrzanowski, W. Nano-Enhanced Drug Delivery and Therapeutic Ultrasound for Cancer Treatment and Beyond. Front. Bioeng. Biotechnol. 2019, 7, 324.
- Wang, Y.; Deng, Y.; Luo, H.; Zhu, A.; Ke, H.; Yang, H.; Chen, H. Light-Responsive Nanoparticles for Highly Efficient Cytoplasmic Delivery of Anticancer Agents. ACS Nano 2017, 11, 12134–12144.
- Linsley, C.S.; Wu, B.M. Recent Advances in Light-Responsive on-Demand Drug-Delivery Systems. Ther. Deliv. 2017, 8, 89–107.
- Raza, A.; Hayat, U.; Rasheed, T.; Bilal, M.; Iqbal, H.M.N. “smart” Materials-Based near-Infrared Light-Responsive Drug Delivery Systems for Cancer Treatment: A Review. J. Mater. Res. Technol. 2019, 8, 1497–1509.
- Liguori, I.; Russo, G.; Curcio, F.; Bulli, G.; Aran, L.; Della-Morte, D.; Gargiulo, G.; Testa, G.; Cacciatore, F.; Bonaduce, D.; et al. Oxidative Stress, Aging, and Diseases. Clin. Interv. Aging 2018, 13, 757–772.
- Huo, M.; Yuan, J.; Tao, L.; Wei, Y. Redox-Responsive Polymers for Drug Delivery: From Molecular Design to Applications. Polym. Chem. 2014, 5, 1519–1528.
- Fang, T.; Zhang, J.; Zuo, T.; Wu, G.; Xu, Y.; Yang, Y.; Yang, J.; Shen, Q. Chemo-Photothermal Combination Cancer Therapy with ROS Scavenging, Extracellular Matrix Depletion and Tumor Immune Activation by Telmisartan and Diselenide-Paclitaxel Prodrug Loaded Nanoparticles. ACS Appl. Mater. Interfaces 2020, 12, 31292–31308.
- Guo, X.; Cheng, Y.; Zhao, X.; Luo, Y.; Chen, J.; Yuan, W.E. Advances in Redox-Responsive Drug Delivery Systems of Tumor Microenvironment. J. Nanobiotechnol. 2018, 16, 74.
- Kumari, R.; Sunil, D.; Ningthoujam, R.S. Hypoxia-Responsive Nanoparticle Based Drug Delivery Systems in Cancer Therapy: An up-to-Date Review. J. Control. Release 2020, 319, 135–156.
- Sada, K.; Nishikawa, T.; Kukidome, D.; Yoshinaga, T.; Kajihara, N.; Sonoda, K.; Senokuchi, T.; Motoshima, H.; Matsumura, T.; Araki, E. Hyperglycemia Induces Cellular Hypoxia through Production of Mitochondrial ROS Followed by Suppression of Aquaporin-1. PLoS ONE 2016, 11, e0158619.
- Yu, J.; Zhang, Y.; Ye, Y.; DiSanto, R.; Sun, W.; Ranson, D.; Ligler, F.S.; Buse, J.B.; Gu, Z.; Ho, D. Microneedle-Array Patches Loaded with Hypoxia-Sensitive Vesicles Provide Fast Glucose-Responsive Insulin Delivery. Proc. Natl. Acad. Sci. USA 2015, 112, 8260–8265.
- Hu, X.; Yu, J.; Qian, C.; Lu, Y.; Kahkoska, A.R.; Xie, Z.; Jing, X.; Buse, J.B.; Gu, Z. H2O2-Responsive Vesicles Integrated with Transcutaneous Patches for Glucose-Mediated Insulin Delivery. ACS Nano 2017, 11, 613–620.
- Yu, J.; Qian, C.; Zhang, Y.; Cui, Z.; Zhu, Y.; Shen, Q.; Ligler, F.S.; Buse, J.B.; Gu, Z. Hypoxia and H2O2 Dual-Sensitive Vesicles for Enhanced Glucose-Responsive Insulin Delivery. Nano Lett. 2017, 17, 733–739.
- Cadenas, S. ROS and Redox Signaling in Myocardial Ischemia-Reperfusion Injury and Cardioprotection. Free Radic. Biol. Med. 2018, 117, 76–89.
- Serracino-Inglott, F.; Habib, N.A.; Mathie, R.T. Hepatic Ischemia-Reperfusion Injury. Am. J. Surg. 2001, 181, 160–166.
- Minutoli, L.; Puzzolo, D.; Rinaldi, M.; Irrera, N.; Marini, H.; Arcoraci, V.; Bitto, A.; Crea, G.; Pisani, A.; Squadrito, F.; et al. ROS-Mediated NLRP3 Inflammasome Activation in Brain, Heart, Kidney, and Testis Ischemia/Reperfusion Injury. Oxid. Med. Cell. Longev. 2016, 2016, 2183026.
- Lee, D.; Bae, S.; Hong, D.; Lim, H.; Yoon, J.H.; Hwang, O.; Park, S.; Ke, Q.; Khang, G.; Kang, P.M. H2O2 -Responsive Molecularly Engineered Polymer Nanoparticles as Ischemia/Reperfusion-Targeted Nanotherapeutic Agents. Sci. Rep. 2013, 3, 2233.
- Bae, S.; Park, M.; Kang, C.; Dilmen, S.; Kang, T.H.; Kang, D.G.; Ke, Q.; Lee, S.U.; Lee, D.; Kang, P.M. Hydrogen Peroxide-Responsive Nanoparticle Reduces Myocardial Ischemia/Reperfusion Injury. J. Am. Heart Assoc. 2016, 5, e003697.
- Eshun, D.; Saraf, R.; Bae, S.; Jeganathan, J.; Mahmood, F.; Dilmen, S.; Ke, Q.; Lee, D.; Kang, P.M.; Matyal, R. Neuropeptide Y3-36 Incorporated into PVAX Nanoparticle Improves Functional Blood Flow in a Murine Model of Hind Limb Ischemia. J. Appl. Physiol. 2017, 122, 1388–1397.
- Mahmood, E.; Bae, S.; Chaudhary, O.; Feng, R.; Mahmood, F.; Robson, S.; Lee, D.; Kang, P.M.; Matyal, R. Neuropeptide Y3-36 Incorporated into PVAX Nanoparticle Improves Angiogenesis in a Murine Model of Myocardial Ischemia. Eur. J. Pharmacol. 2020, 882, 173261.
- Minami, K.; Bae, S.; Uehara, H.; Zhao, C.; Lee, D.; Iske, J.; Fanger, M.W.; Reder, J.; Morrison, I.; Azuma, H.; et al. Targeting of Intragraft Reactive Oxygen Species by APP-103, a Novel Polymer Product, Mitigates Ischemia/Reperfusion Injury and Promotes the Survival of Renal Transplants. Am. J. Transplant. 2020, 20, 1527–1537.
- Elkassih, S.A.; Kos, P.; Xiong, H.; Siegwart, D.J. Degradable Redox-Responsive Disulfide-Based Nanogel Drug Carriers: Via Dithiol Oxidation Polymerization. Biomater. Sci. 2019, 7, 607–617.
- McBain, S.C.; Yiu, H.H.P.; Dobson, J. Magnetic Nanoparticles for Gene and Drug Delivery. Int. J. Nanomed. 2008, 3, 169–180.
- Mahmoudi, M.; Sant, S.; Wang, B.; Laurent, S.; Sen, T. Superparamagnetic Iron Oxide Nanoparticles (SPIONs): Development, Surface Modification and Applications in Chemotherapy. Adv. Drug Deliv. Rev. 2011, 63, 24–46.
- Liu, J.F.; Lan, Z.; Ferrari, C.; Stein, J.M.; Higbee-Dempsey, E.; Yan, L.; Amirshaghaghi, A.; Cheng, Z.; Issadore, D.; Tsourkas, A. Use of Oppositely Polarized External Magnets to Improve the Accumulation and Penetration of Magnetic Nanocarriers into Solid Tumors. ACS Nano 2020, 14, 142–152.
- Zhou, Z.; Shen, Z.; Chen, X. Tale of Two Magnets: An Advanced Magnetic Targeting System. ACS Nano 2020, 14, 7–11.
- Price, P.M.; Mahmoud, W.E.; Al-Ghamdi, A.A.; Bronstein, L.M. Magnetic Drug Delivery: Where the Field Is Going. Front. Chem. 2018, 6, 619.
- Shapiro, B.; Kulkarni, S.; Nacev, A.; Muro, S.; Stepanov, P.Y.; Weinberg, I.N. Open Challenges in Magnetic Drug Targeting. Wiley Interdiscip. Rev. Nanomed. Nanobiotechnol. 2015, 7, 446–457.
- Liu, Y.L.; Chen, D.; Shang, P.; Yin, D.C. A Review of Magnet Systems for Targeted Drug Delivery. J. Control. Release 2019, 302, 90–104.
- De la Rica, R.; Aili, D.; Stevens, M.M. Enzyme-Responsive Nanoparticles for Drug Release and Diagnostics. Adv. Drug Deliv. Rev. 2012, 64, 967–978.
- Mu, J.; Lin, J.; Huang, P.; Chen, X. Development of Endogenous Enzyme-Responsive Nanomaterials for Theranostics. Chem. Soc. Rev. 2018, 47, 5554–5573.
- Fouladi, F.; Steffen, K.J.; Mallik, S. Enzyme-Responsive Liposomes for the Delivery of Anticancer Drugs. Bioconjug. Chem. 2017, 28, 857–868.
- Shahriari, M.; Zahiri, M.; Abnous, K.; Taghdisi, S.M.; Ramezani, M.; Alibolandi, M. Enzyme Responsive Drug Delivery Systems in Cancer Treatment. J. Control. Release 2019, 308, 172–189.
- Zhang, C.; Pan, D.; Li, J.; Hu, J.; Bains, A.; Guys, N.; Zhu, H.; Li, X.; Luo, K.; Gong, Q.; et al. Enzyme-Responsive Peptide Dendrimer-Gemcitabine Conjugate as a Controlled-Release Drug Delivery Vehicle with Enhanced Antitumor Efficacy. Acta Biomater. 2017, 55, 153–162.
- Shay, G.; Lynch, C.C.; Fingleton, B. Moving Targets: Emerging Roles for MMPs in Cancer Progression and Metastasis. Matrix Biol. 2015, 44–46, 200–206.
- Eskandari, P.; Bigdeli, B.; Porgham Daryasari, M.; Baharifar, H.; Bazri, B.; Shourian, M.; Amani, A.; Sadighi, A.; Goliaei, B.; Khoobi, M.; et al. Gold-Capped Mesoporous Silica Nanoparticles as an Excellent Enzyme-Responsive Nanocarrier for Controlled Doxorubicin Delivery. J. Drug Target. 2019, 27, 1084–1093.
- Korkmaz, B.; Horwitz, M.S.; Jenne, D.E.; Gauthier, F. Neutrophil Elastase, Proteinase 3, and Cathepsin G as Therapeutic Targets in Human Diseases. Pharmacol. Rev. 2010, 62, 726–759.
- Aimetti, A.A.; Tibbitt, M.W.; Anseth, K.S. Human Neutrophil Elastase Responsive Delivery from Poly(Ethylene Glycol) Hydrogels. Biomacromolecules 2009, 10, 1484–1489.
- Rosales, C. Neutrophil: A Cell with Many Roles in Inflammation or Several Cell Types? Front. Physiol. 2018, 9, 113.
- Bhat, R.; Ribes, À.; Mas, N.; Aznar, E.; Sancenón, F.; Marcos, M.D.; Murguía, J.R.; Venkataraman, A.; Martínez-Máñez, R. Thrombin-Responsive Gated Silica Mesoporous Nanoparticles As Coagulation Regulators. Langmuir 2016, 32, 1195–1200.
- Greineder, C.F.; Howard, M.D.; Carnemolla, R.; Cines, D.B.; Muzykantov, V.R. Advanced Drug Delivery Systems for Antithrombotic Agents. Blood 2013, 122, 1565–1575.
- Kemp, J.A.; Shim, M.S.; Heo, C.Y.; Kwon, Y.J. “Combo” Nanomedicine: Co-Delivery of Multi-Modal Therapeutics for Efficient, Targeted, and Safe Cancer Therapy. Adv. Drug Deliv. Rev. 2016, 98, 3–18.
- Moreira, A.F.; Dias, D.R.; Costa, E.C.; Correia, I.J. Thermo- and PH-Responsive Nano-in-Micro Particles for Combinatorial Drug Delivery to Cancer Cells. Eur. J. Pharm. Sci. 2017, 104, 42–51.
- Hiruta, Y.; Kanda, Y.; Katsuyama, N.; Kanazawa, H. Dual Temperature-and PH-Responsive Polymeric Micelle for Selective and Efficient Two-Step Doxorubicin Delivery. RSC Adv. 2017, 7, 29540–29549.
- Zhang, P.; Gao, D.; An, K.; Shen, Q.; Wang, C.; Zhang, Y.; Pan, X.; Chen, X.; Lyv, Y.; Cui, C.; et al. A Programmable Polymer Library That Enables the Construction of Stimuli-Responsive Nanocarriers Containing Logic Gates. Nat. Chem. 2020, 12, 381–390.
- Badeau, B.A.; Comerford, M.P.; Arakawa, C.K.; Shadish, J.A.; Deforest, C.A. Engineered Modular Biomaterial Logic Gates for Environmentally Triggered Therapeutic Delivery. Nat. Chem. 2018, 10, 251–258.
- Jia, H.R.; Zhu, Y.X.; Liu, X.; Pan, G.Y.; Gao, G.; Sun, W.; Zhang, X.; Jiang, Y.W.; Wu, F.G. Construction of Dually Responsive Nanotransformers with Nanosphere-Nanofiber-Nanosphere Transition for Overcoming the Size Paradox of Anticancer Nanodrugs. ACS Nano 2019, 13, 11781–11792.
- Kim, J.; Kim, J.; Jeong, C.; Kim, W.J. Synergistic Nanomedicine by Combined Gene and Photothermal Therapy. Adv. Drug Deliv. Rev. 2016, 98, 99–112.
- Deng, W.; Chen, W.; Clement, S.; Guller, A.; Zhao, Z.; Engel, A.; Goldys, E.M. Controlled Gene and Drug Release from a Liposomal Delivery Platform Triggered by X-Ray Radiation. Nat. Commun. 2018, 9, 2713.
- Ansari, C.; Tikhomirov, G.A.; Hong, S.H.; Falconer, R.A.; Loadman, P.M.; Gill, J.H.; Castaneda, R.; Hazard, F.K.; Tong, L.; Lenkov, O.D.; et al. Development of Novel Tumor-Targeted Theranostic Nanoparticles Activated by Membrane-Type Matrix Metalloproteinases for Combined Cancer Magnetic Resonance Imaging and Therapy. Small 2014, 10, 566–575.
- Sarkar, S.; Levi-Polyachenko, N. Conjugated Polymer Nano-Systems for Hyperthermia, Imaging and Drug Delivery. Adv. Drug Deliv. Rev. 2020. S0169-409X(20)30004-1.
- Li, S.; Su, W.; Wu, H.; Yuan, T.; Yuan, C.; Liu, J.; Deng, G.; Gao, X.; Chen, Z.; Bao, Y.; et al. Targeted Tumour Theranostics in Mice via Carbon Quantum Dots Structurally Mimicking Large Amino Acids. Nat. Biomed. Eng. 2020, 4, 704–716.
- Riccardi, C.; Musumeci, D.; Trifuoggi, M.; Irace, C.; Paduano, L.; Montesarchio, D. Anticancer Ruthenium(III) Complexes and Ru(III)-Containing Nanoformulations: An Update on the Mechanism of Action and Biological Activity. Pharmaceuticals 2019, 12, 146.
- Xia, Q.; Zhang, Y.; Li, Z.; Hou, X.; Feng, N. Red Blood Cell Membrane-Camouflaged Nanoparticles: A Novel Drug Delivery System for Antitumor Application. Acta Pharm. Sin. B 2019, 9, 675–689.
- Liu, J.M.; Zhang, D.D.; Fang, G.Z.; Wang, S. Erythrocyte Membrane Bioinspired Near-Infrared Persistent Luminescence Nanocarriers for in Vivo Long-Circulating Bioimaging and Drug Delivery. Biomaterials 2018, 165, 39–47.
- Balasubramanian, V.; Poillucci, A.; Correia, A.; Zhang, H.; Celia, C.; Santos, H.A. Cell Membrane-Based Nanoreactor to Mimic the Bio-Compartmentalization Strategy of a Cell. ACS Biomater. Sci. Eng. 2018, 4, 1471–1478.
- Jin, J.; Bhujwalla, Z.M. Biomimetic Nanoparticles Camouflaged in Cancer Cell Membranes and Their Applications in Cancer Theranostics. Front. Oncol. 2020, 9, 1560.
- Xu, C.; Liu, W.; Hu, Y.; Li, W.; Di, W. Bioinspired Tumor-Homing Nanoplatform for Co-Delivery of Paclitaxel and SiRNA-E7 to HPV-Related Cervical Malignancies for Synergistic Therapy. Theranostics 2020, 10, 3325–3339.
- Balasubramanian, V.; Correia, A.; Zhang, H.; Fontana, F.; Mäkilä, E.; Salonen, J.; Hirvonen, J.; Santos, H.A. Biomimetic Engineering Using Cancer Cell Membranes for Designing Compartmentalized Nanoreactors with Organelle-Like Functions. Adv. Mater. 2017, 29, 1605375.
- Zhang, Z.; Guan, J.; Jiang, Z.; Yang, Y.; Liu, J.; Hua, W.; Mao, Y.; Li, C.; Lu, W.; Qian, J.; et al. Brain-Targeted Drug Delivery by Manipulating Protein Corona Functions. Nat. Commun. 2019, 10, 3561.
- Ernst, A.U.; Bowers, D.T.; Wang, L.H.; Shariati, K.; Plesser, M.D.; Brown, N.K.; Mehrabyan, T.; Ma, M. Nanotechnology in Cell Replacement Therapies for Type 1 Diabetes. Adv. Drug Deliv. Rev. 2019, 139, 116–138.
- Veiseh, O.; Tang, B.C.; Whitehead, K.A.; Anderson, D.G.; Langer, R. Managing Diabetes with Nanomedicine: Challenges and Opportunities. Nat. Rev. Drug Discov. 2014, 14, 45–57.
- Warshauer, J.T.; Bluestone, J.A.; Anderson, M.S. New Frontiers in the Treatment of Type 1 Diabetes. Cell Metab. 2020, 31, 46–61.
- Krueger, T.E.G.; Thorek, D.L.J.; Denmeade, S.R.; Isaacs, J.T.; Brennen, W.N. Concise Review: Mesenchymal Stem Cell-Based Drug Delivery: The Good, the Bad, the Ugly, and the Promise. Stem Cells Transl. Med. 2018, 7, 651–663.
- Dong, Y.; Siegwart, D.J.; Anderson, D.G. Strategies, Design, and Chemistry in SiRNA Delivery Systems. Adv. Drug Deliv. Rev. 2019, 144, 133–147.
- Bumcrot, D.; Manoharan, M.; Koteliansky, V.; Sah, D.W.Y. RNAi Therapeutics: A Potential New Class of Pharmaceutical Drugs. Nat. Chem. Biol. 2006, 2, 711–719.
- Fernandes, F.; Kotharkar, P.; Chakravorty, A.; Kowshik, M.; Talukdar, I. Nanocarrier Mediated SiRNA Delivery Targeting Stem Cell Differentiation. Curr. Stem Cell Res. Ther. 2019, 15, 155–172.
- Ramamoorth, M.; Narvekar, A. Non Viral Vectors in Gene Therapy—An Overview. J. Clin. Diagn. Res. 2015, 9, GE01–GE06.
- Wu, Y.F.; Wu, H.C.; Kuan, C.H.; Lin, C.J.; Wang, L.W.; Chang, C.W.; Wang, T.W. Multi-Functionalized Carbon Dots as Theranostic Nanoagent for Gene Delivery in Lung Cancer Therapy. Sci. Rep. 2016, 6, 21170.
- Bates, K.; Kostarelos, K. Carbon Nanotubes as Vectors for Gene Therapy: Past Achievements, Present Challenges and Future Goals. Adv. Drug Deliv. Rev. 2013, 65, 2023–2033.
- Nishiyama, N.; Bae, Y.; Miyata, K.; Fukushima, S.; Kataoka, K. Smart Polymeric Micelles for Gene and Drug Delivery. Drug Discov. Today Technol. 2005, 2, 21–26. [Google Scholar] [CrossRef] [PubMed]
- Kulkarni, J.A.; Witzigmann, D.; Chen, S.; Cullis, P.R.; Van Der Meel, R. Lipid Nanoparticle Technology for Clinical Translation of SiRNA Therapeutics. Acc. Chem. Res. 2019, 52, 2435–2444.
- Coelho, T.; Adams, D.; Silva, A.; Lozeron, P.; Hawkins, P.N.; Mant, T.; Perez, J.; Chiesa, J.; Warrington, S.; Tranter, E.; et al. Safety and Efficacy of RNAi Therapy for Transthyretin Amyloidosis. N. Engl. J. Med. 2013, 369, 819–829.
- Davis, M.E. The First Targeted Delivery of SiRNA in Humans via a Self-Assembling, Cyclodextrin Polymer-Based Nanoparticle: From Concept to Clinic. Mol. Pharm. 2009, 6, 659–668.
- Cheng, Q.; Wei, T.; Farbiak, L.; Johnson, L.T.; Dilliard, S.A.; Siegwart, D.J. Selective Organ Targeting (SORT) Nanoparticles for Tissue-Specific MRNA Delivery and CRISPR–Cas Gene Editing. Nat. Nanotechnol. 2020, 15, 313–320.
- Samaridou, E.; Heyes, J.; Lutwyche, P. Lipid Nanoparticles for Nucleic Acid Delivery: Current Perspectives. [published online ahead of print, 2020 Jun 8]. Adv. Drug Deliv. Rev. 2020. Jun 8;S0169-409.
- Riccardi, C.; Fàbrega, C.; Grijalvo, S.; Vitiello, G.; D’Errico, G.; Eritja, R.; Montesarchio, D. AS1411-Decorated Niosomes as Effective Nanocarriers for Ru(III)-Based Drugs in Anticancer Strategies. J. Mater. Chem. B 2018, 6, 5368–5384.
