Pentacene is a well-known conjugated organic molecule with high mobility and a sensitive photo response. It is widely used in electronic devices, such as in organic thin-film transistors (OTFTs), organic light-emitting diodes (OLEDs), photodetectors, and smart sensors. With the development of flexible and wearable electronics, the deposition of good-quality pentacene films in large-scale organic electronics at the industrial level has drawn more research attention. Several methods are used to deposit pentacene thin films. The thermal evaporation technique is the most frequently used method for depositing thin films, as it has low contamination rates and a well-controlled deposition rate. Solution-processable methods such as spin coating, dip coating, and inkjet printing have also been widely studied because they enable large-scale deposition and low-cost fabrication of devices. This manuscript summarizes the deposition principles, control parameters and electronics performance of each deposition method for pentacene and its derivatives.
- pentacene
- thin-film deposition
- solution-process
- thermal vacuum evaporation
- organic thin-film transistor
- inkjet printing
Introduction
Many pentacene precursors and substitutes have been made, and these pentacene derivatives had been synthesized and characterized for suitable usage in organic thin film transistors (OTFTs) [1,2], organic light emitting diodes (OLEDs) [3,4], and other organic electronic devices, such as thin-film sensors [5–12]. Pentacene is preferable due to its excellent semiconducting properties, being well understood and relatively cheap compared to the latest generation of organic semiconductors. With the newer improved pentacene derivatives, solution–process deposition has been made possible.
Pentacene itself contains five linearly fused aromatic rings and is also known as a polyaromatic hydrocarbon. Figure 1 shows an illustration of the flat molecules and carbon numbering of pentacene.
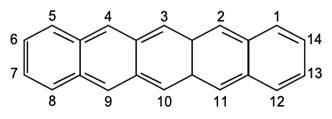
Figure 1. Carbon numbering of pentacene.
Unfortunately, pentacene is sensitive to ambient air (moisture and oxygen) and has low solubility, making it unstable when used with common fabrication techniques. Thus, pentacene should only be deposited using evaporation techniques. Initially, only ultra-high vacuum equipment was used in the deposition process for pentacene. Pentacene and its derivatives are not new materials. Nevertheless, they are still widely used in research and prototyping, either alone or in conjunction (stacked or blended) with other compounds.
The sensitivity of pentacene is due to its structure. This is because the diffusion of its five rings tempers the aromaticity and causes it to be more reactive [13]. This commonly occurs in the central ring, as the reaction of oxygen with the pentacene will produce an endo-peroxide on the central ring. Light and oxygen are the causes of this degradation and should be avoided during storage. These problems of solubility and stability were solved over the years through the development of two different methods [14].
The first method involves soluble pentacene precursors, as shown in Figure 2, which can be easily purified and deposited in solution form. This means that they are more soluble than the planar pentacene when deposited onto a substrate and can be converted into pentacene again using either heat or light. This process is called a retro-Diels–Alder reaction, which allows the precursors to be reconverted into semiconducting pentacene. This makes it competitive with other soluble semiconducting polymers, such as polypyrrole (PPy), polyaniline, and polydopamine (PDA).
Figure 2. Example of a pentacene precursor structure.
The second method involves introducing substituents onto the aromatic core (Figure 3), which increases the stability and solubility of the pentacene. It can also improve the electrical properties due to the self-assembly of pentacene moieties, which causes them to be packed closely together. This method has better purity control and allows solution processing. It also improves the stability of the original pentacene against oxygen [15,16]. However, these pentacene derivatives cannot be converted back into pentacene in the way pentacene precursors can.
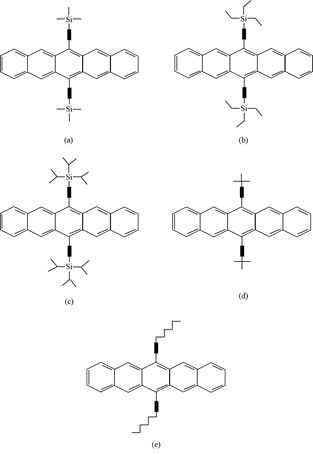
Figure 3. Structures of pentacene derivatives: (a) trimethylsilyl (TMS) pentacene; (b) triethylsilyl (TES) pentacene; (c) triisopropylsilyl (TIPS) pentacene; (d) t-butyl pentacene; (e) hexyl pentacene.
- Thermal Vacuum Evaporation Methods
Thermal vacuum evaporation techniques are employed when the organic semiconductor can be sublimed, which is common for most small-molecule semiconductors [17,18]. Some examples of these techniques are organic molecular beam deposition (OMBD) [19] and organic vapor-phase deposition (OVPD) [20].
For molecules such as pentacene, the OMBD technique is frequently used [21–23]. The technique uses a high to ultra-high vacuum (10−6–10−12 Torr) to evaporate the material (evaporant). The substrate is be placed parallel to the sublimed molecules inside a vacuum chamber and a semiconductor layer is formed on the substrate. The advantage of this technique is the efficient control of the growth conditions of the molecule film on the substrate. This means that the thin film produced on the substrate is in a well-ordered condition. The common thermal vacuum evaporation method is illustrated in Figure 4.
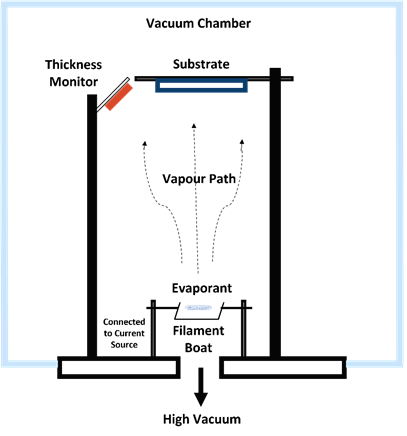
Figure 4. Thermal vacuum evaporation method.
The OVPD technique, however, uses a relatively low vacuum pressure combined with a carrier gas [24,25]. In this technique, the material is evaporated and the carrier gas transports the molecules out through an outlet and towards the substrate that has been placed underneath. This technique is illustrated in Figure 5.
Figure 5. Organic vapor-phase deposition.
The performance results for thermally evaporated OTFTs and others are listed in Table 1 [26,27–30]. Note that the table lists only the average typical values relevant to this manuscript. The typical mobility rate for pristine pentacene OTFT is around 1 cm2 V−1 s−1; much higher mobility values have been reported in previous studies, such as from our previous work, which required more complex treatments with mobility rates of up to 4.7 cm2 V−1 s−1 [27].
Table 1. Electrical performance comparison of OTFTs based on pentacene and its derivatives.
|
Reference No. |
Deposition Method |
Material |
Carrier Mobilities |
ION/OFF |
Threshold Voltage (V) |
|
[31] |
Thermal vacuum evaporation |
TMS-pentacene, |
~10−5 |
NIL |
NIL |
|
[26] |
Thermal vacuum evaporation |
Pentacene |
0.62 |
102 |
−8.5 |
|
Spin coating |
Pentacene precursor of 13,6-N-Sulfinylacetamidopentacene (SAP), |
0.031 |
103 |
−12.5 |
|
|
[32] |
Organic molecular beam |
Pentacene |
0.435 |
1.83 × 106 |
4.77 |
|
[27] |
Thermal vacuum evaporation |
Pentacene |
2.5 |
107 |
−4 |
|
[28] |
Thermal vacuum evaporation |
Pentacene |
0.18 |
103 |
NIL |
|
[29] |
Thermal vacuum evaporation |
Pentacene |
0.1 |
3.3 × 105 |
1.5 |
|
[30] |
Thermal vacuum evaporation |
Pentacene |
0.025 |
NIL |
−1 |
|
[33] |
Spin coating |
TIPS-pentacene |
0.002 |
102 |
3.2 |
|
[34] |
Spin coating |
TIPS-pentacene |
0.005 |
NIL |
−1.3 |
|
[35] |
Spin coating |
TIPS-pentacene |
0.6 |
106 |
NIL |
|
[36] |
Spin coating |
TIPS-pentacene |
1.66 |
7 × 109 |
NIL |
|
[37] |
Spin coating |
TIPS-pentacene |
3.40 |
104 |
−10 |
|
[38] |
Spin coating |
TIPS-pentacene |
0.05 |
NIL |
NIL |
|
[39] |
Drop casting |
TIPS-pentacene |
0.12 |
104 |
−0.2 |
|
[40] |
Drop casting |
TIPS-pentacene |
0.005 |
NIL |
5 |
|
[41] |
Drop casting |
TIPS-pentacene |
0.00016 |
NIL |
−10 |
|
[42] |
Drop casting |
TIPS-pentacene |
0.57 |
NIL |
0.27 |
|
[43] |
Drop casting |
TIPS-pentacene |
2.22 |
1.3 × 104 |
−5.75 |
|
[44] |
Drop casting |
TIPS-pentacene |
0.013 |
NIL |
−0.14 |
|
[45] |
Drop casting |
TIPS-pentacene |
0.78 |
104 |
−0.56 |
|
[46] |
Drop casting |
TIPS-pentacene |
0.15 |
NIL |
NIL |
|
[47] |
Drop casting |
TIPS-pentacene |
0.44 |
105 |
−0.3 |
|
[48] |
Dip coating |
TIPS-pentacene |
3.0 |
109 |
10 |
|
[49] |
Dip coating |
TIPS-pentacene |
1.2 |
NIL |
NIL |
|
[50] |
Dip coating |
TIPS-pentacene |
0.047 |
NIL |
NIL |
|
[51] |
Inkjet printing |
TIPS-pentacene |
0.53 |
1.6 × 106 |
−0.7 |
|
[52] |
Inkjet printing |
TIPS-pentacene |
0.23 |
2.01 x 106 |
NIL |
|
[53] |
Inkjet printing |
TIPS-pentacene |
0.22 |
5.7 × 104 |
−0.4 |
|
[54] |
Inkjet printing |
TIPS-pentacene |
0.35 |
1.2 × 107 |
NIL |
Other than the common OFETs and OLEDs, pentacene was also fabricated onto solar cells. Table 2 lists optoelectronic device performance comparisons. The thermal vacuum evaporation technique, devices are individually fabricated due to the limitations on space in the vacuum chamber. This deposition method is very costly, time-consuming, and not suitable for mass production. Hence, other deposition methods have been explored in order to solve these issues in the production process. The following alternative deposition methods, namely spin coating, dip coating, drop casting, and inkjet printing, are discussed in this manuscript.
Table 2. Performance comparison of pentacene-based optoelectronic devices.
|
Reference No. |
Deposition Method |
Device |
Material |
Current |
Power |
Turn On Voltage |
Quantum Yield (%) |
Power |
|
[55] |
Thermal vacuum evaporation |
OLED |
Pentacene |
6.6 |
3.4 |
5.8 |
NIL |
NIL |
|
[56] |
Thermal vacuum evaporation |
Solar Cells |
Pentacene |
NIL |
NIL |
NIL |
NIL |
0.33 |
|
[57] |
Thermal vacuum evaporation |
Solar Cells |
Pentacene |
NIL |
NIL |
NIL |
NIL |
5.7 |
|
[58] |
Thermal vacuum evaporation |
OLED |
Pentacene |
NIL |
NIL |
5.0 |
32 |
NIL |
|
[59] |
Thermal vacuum evaporation |
OLED |
Pentacene |
3.4 |
NIL |
NIL |
NIL |
NIL |
- Spin Coating Method
Spin coating is one of the most used methods in academic studies, as well as in industry, due to its capability for mass production at cheaper costs [60–63]. It is also easier to perform as it only requires a few drops of solution. The solution is dropped on top of a substrate that is held onto a chuck within the spin coater, as illustrated in Figure 6. The spinning process starts with a certain acceleration rate and remains at a constant speed for a period of time. Upon completion, the desired thin film will form via a combination of evaporation processes and liquid flow. An additional step such as heat or UV treatment may be required depending on the solution’s characteristics [20].
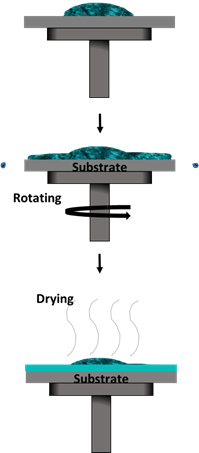
Figure 6. Spin coating method.
The surface coverage is important in this technique to ensure the uniformity of the thin film being deposited [64,65]. Bharti et al. [66] studied the deposition of TIPS-pentacene using this method with different dropping positions of the solution onto the substrate (Figure 7). They studied the effect it has on the alignment and crystallinity of the pentacene when it is deposited off-center to the substrate. Compared to the usual central dropping, the off-centered dropping approach was better for the crystallinity and pentacene alignment.
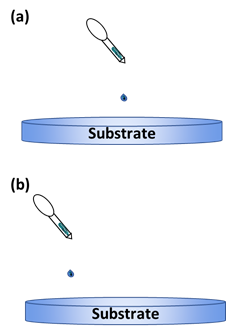
Figure 7. Illustration of the dropping positions: (a) central dropping; (b) off-centered dropping.
The thickness of the thin-film layer depends on the spin speed and duration of the spin. The solution’s properties, such as its density, viscosity, shear thinning, evaporation rates, and liquid flow, also effect the thickness. By altering the acceleration and spin speed, the liquid flow can be controlled. The evaporation rate on the other hand can majorly impact the morphology of the thin film. This method is suitably used to deposit pentacene precursors or pentacene derivatives, as they both are in solution form.
For TIPS-pentacene, the carrier mobility depends greatly on the crystal orientation [67–69], crystalline thickness, crystalline domain size, and presence of cracks. Past research had showed a mobility rate of 1.8 cm2 V−1 s−1 for TIPS-pentacene transistors [70,71]. This result depends on the solution concentration, solvent selection, temperature of the thermal treatment, method of deposition, and substrate material. In general, it has been shown that solvents with a high boiling point deliver slow crystal growth. This enables the thin film to be formed with a large lamellar structure, possibly giving higher mobility.
Although heat treatment is needed for complete solvent evaporation, temperatures above 60 C could damage the thin-film organic semiconductor. For this reason, it is not easy to obtain devices with uniform characteristics. Hence, polymer blends were developed to improve the morphology of the TIPS-pentacene. Doing so enabled control of the crystallinity of the film, grain size, and other parameters.
It is known that the electrical performance of OTFTs is greatly dependent on the surface energy of the gate dielectric. Considering the active channel is located at the interface between the dielectric and semiconductor layers, a hydrophobic treatment of the gate dielectric surface can effectively increase the carrier mobility by several orders of magnitude. Therefore, it is crucial that the gate dielectric’s surface energy be lowered to improve the mobility. This would require a trade-off between the surface energy and surface wettability, making it challenging to produce a uniform thin film of an organic semiconductor on a hydrophobic dielectric (low surface energy) using a simple spin coating method.
Hence, two major approaches were taken. The first was to apply doping to change the surface tension of the organic semiconductor solution, thereby improving the surface wettability [33]. However, this doping had significant effects on the purity and the crystallinity of the semiconductor, directly impacting the mobility of the OTFT. The second was to dissolve the organic semiconductor in a solvent, which allowed the surface tension to be modified and enabled a continuous thin film to form on a hydrophobic dielectric. This approach was reported previously by Liu et al. [72], whereby OTFTs were fabricated by employing a low surface tension hexane as the solvent. A small-molecule thin film was deposited on the octadecyltrimethoxysilane (OTMS)-treated dielectric surface; however, the mobility was low at 0.02 cm2 V−1 s−1, albeit this showed an improvement in wettability. This report showed the importance of controlling the wettability of the organic semiconductor solution on hydrophobic surfaces to form uniform and continuous thin films.
In another related study, Wang et al. [36] investigated a low-viscosity organic semiconductor solution by spin coating a hydrophobic surface. Different solvents were tested to find the most suitable for producing a smooth, uniform, and continuous thin film. TIPS-pentacene was dissolved in hexane, toluene, chlorobenzene, 1,2-dichlorobenzene, and 1,2,3,4-tetrahydronaphthalene individually. Each solution was spin-coated onto an OTS/SiO2 substrate. They found that hexane produced the best results in terms of device performance. It improved the solution’s wettability by completely wetting the hydrophobic OTS/SiO2 surface
The performance comparison of spin-coated OFETs are listed in Table 1.
- Drop Casting Method
The drop casting method is an alternative deposition method that is more suitable for small-area deposition [73–75]. This method is performed by dropping a specified amount of solution on a static substrate and letting the solution evaporate for a desired amount of time. The substrate can also be baked to enhance the evaporation process. A solid thin film will then be formed on the substrate once the solution has dried up. The advantage of this method is its simple process. However, this method’s main disadvantages are the difficulty in obtaining a uniform and continuous coating on the deposited layer. The thickness of the deposited layer is also uncontrollable. Figure 8 shows an illustration of the drop casting method.
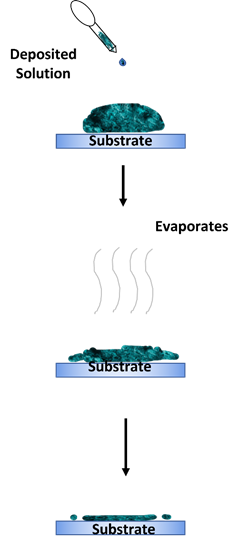
Figure 8. Drop casting method.
Park et al. [76] used this deposition method to study terahertz modulation using a TIPS-pentacene thin film deposited on a patterned silicon substrate. The experiment was performed by drop casting 50 µL of TIPS-pentacene solution with a concentration of 2 mg/mL onto the substrate. It was then covered with a glass lid and heated on a 50 °C hotplate for 5 min. Despite the non-uniformity in the deposition process, the concentration of carriers injected into the TIPS-pentacene thin film rapidly became consistent over the whole area of the organic layer near the organic/inorganic interface. This was probably due to the fast in-plane diffusion of the carriers in the thin film itself.
The drop casting approach soon became known for producing semiconductors with a good crystallite structure, increasing device performance. Raghuwanshi et al. [39] investigated the crystal growth of TIPS-pentacene and its electrical stability on a flexible OFET upon bending. The tensile strain effect on the field-effect mobility was studied. TIPS-pentacene was mixed with toluene (1 wt.%) by stirring at 70 °C for 2.5 h. The solution was then deposited onto the substrate, which was tilted at an angle of ~5° and later covered with a glass Petri dish to maintain the substrate in a solvent-rich environment throughout the drying process. The substrate then underwent heat treatment at 80 °C to remove the residual solvent. The device was operated at a low voltage of –15 V and showed excellent p-channel characteristics. It achieved a maximum carrier mobility of 0.12 cm2 V−1 s−1 and threshold voltages as low as −0.2 V. The device was conditioned to a tensile strain test with different bending radii for a period of 5 min, which resulted in a slight decrease in mobility and also an increase in the threshold voltage. The changes in the device performance were mainly caused by the changes in the dielectric morphology, as well as the surface roughness caused by the strain. Additionally, a disruption in uniformity at the semiconductor–dielectric interface also occurred, which was influenced by the magnitude of the strain rather than the duration. This indicated that the device had very good electrical stability under mechanical strain.
Through the drop casting deposition method, the study of the application of pentacene solutions for device fabrication has broadened from conventional OFETs to electrolyte-gated OFETs (EGOFETs). Lago et al. [44] reported the use of TIPS-pentacene as a high-performance biocompatible electronic device that can operate in water. Organic semiconductors such as pentacene have attracted interest and have been implemented in new applications due to their fascinating properties, including their flexibility, transparency, and low-cost processability. This deposition method, which is more affordable and simpler, makes device fabrication a lot easier and more cost-efficient. In their study, they aimed to develop a biocompatible sensor, focusing on the stability of the device operation when in prolonged contact with a strong saline solution.
The performance comparisons of drop-casted OFETs are listed in Table 1 [46,47].
Here, we highlight multiple flexible OTFTs and high-voltage OTFTs that were fabricated using the drop casting deposition method. This deposition method is mostly performed to study the operational capability of such devices. The method is usually performed on a small substrate, avoiding the use of large amounts of materials, as the first step is usually just a draft of the device. There is no other force that influences the deposited droplet; hence, the crystal growth orientation is only influenced by the substrate’s surface tension. Although it produces seemingly good crystal growth and is suitable for the fabrication of flexible and conformable devices, the thin film’s uniformity is not guaranteed. The non-uniformity of the thin film causes the carrier mobility of the device to be low and similar to that of spin-coated thin films. Furthermore, it is not suitable for mass production. Similar to spin coating, this technique is also very vulnerable to contamination from oxygen reactions with the material and humidity, which can damage the material unless the process is performed in an inert environment.
- Dip Coating Method
The dip coating method is also a commonly used technique, as it is fast and offers high uniformity when depositing thin-film layers [77–79]. The downside is that it requires a large amount of precursor solution in the reservoir. It is usually used to form thin films with complex and varying surface morphologies. It is conducted by suspending the substrate onto a clipper and then immersing it in the precursor solution below it. After a specified amount of time, the substrate will then be raised from the solution, enabling liquid film formation. It will then undergo an evaporation process that allows it to then form into a solid thin film. The thin film can then be processed further through thermal annealing or any other type of post-processing technique. Figure 9 shows an illustration of the dip coating
method.
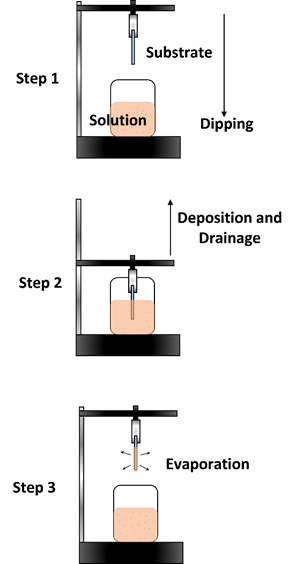
Figure 9. Dip coating method.
The increase or withdrawal speed of the substrate from the precursor solution determines the thickness of the thin-film layer. Other procedures can also be performed to alter the thickness of the thin film, such as varying the concentration of the precursor, the substrate temperature during the deposition, the acceleration rate, or even by performing the deposition at an angle [80–83]. However, it must be noted that several other forces influence the formation of the thin film, including the inertial forces, viscous drag, gravity, and the gradient of the surface tension [20].
The dip coating deposition allows researchers to study the possibility of depositing materials on rigid, flexible, and three-dimensional substrates, leading to a wide range of device development approaches for light-weight, flexible, and potentially low-cost devices. This has increased the interest in the deposition approach for large-area applications. Wang et al. [48] fabricated a single-crystalline organic nanoribbon array on large-area OFETs. Usually, OFETs are made in smaller areas measuring ~10 cm2 in research labs. Instead, they fabricated OFETs in a large area measuring 50 cm2 by using the dip coating method. The OFETs were constructed on silicon wafers with 300 nm of thermally grown SiO2 gate dielectric as the substrate. TIPS-pentacene was used as the active channel. A low boiling point solvent was preferred; thus, the solution was prepared using dichloromethane at a concentration of 4 mg mL−1. The withdrawal speeds in the dip coating process were 10, 30, 60, 80, and 120 m s−1, in order to observe and obtain the optimum results for achieving high carrier mobility. This dip coating process was performed at room temperature.
The nanoribbons were observed to be deposited along the withdrawal direction as the dichloromethane solvent gradually evaporated. The nanoribbons formed in a continuous and well-aligned manner. The withdrawal speed at 80 m s−1 was the most optimal for producing uniform and continuous crystal growth. The continuous crystal growth suggested the OFET device had high mobility. Nanoribbons measuring approximately 50 nm in thickness and 7–10 µm in width were obtained at this withdrawal speed, which had faceted edges with a ~1.2 nm smooth surfaces. Withdrawal speeds below 60 µm s−1 produced periodically aligned and short nanoribbons. At this rate, the TIPS-pentacene gradually accumulated at the contact line, which made the solution’s meniscus become too weighty, causing the depinning force to increase. This resulted in the formation of the short nanoribbon arrays. However, when the withdrawal speed was increased up to 120 µm s−1, non-continuous and defected nanoribbon arrays formed with lower crystallinity. It is known that the carrier mobility is highly affected by the crystallinity of the thin film. Other possible influences were further observed by varying the channel length from 10 to 200 µm. It was found that the carrier mobility gradually increased from 0.1 to 1.35 cm2 V−1 s−1 as the channel was lengthened [48].
Yang et al. [84] demonstrated a two-phase dip coating process in order to avoid the need for a large volume of solution simply by using a floating thin layer of solution on a reservoir of denser liquid, which did not form a homogenous mixture. This method requires strong wetting between the solvent and the substrate to promote the spreading of the solvent when it is withdrawn from the solution. This is also to ensure that the solution spreads continuously. TIPS-pentacene was used by mixing it with hexane at a concentration of 2 mg/mL. To start, a beaker was filled with 80% water and the substrate was partially submerged. As the substrate was withdrawn from the reservoir, the prepared solution was pipetted onto the water surface. The withdrawal speed was 1.2 cm h−1 and only 20 µL of solution was pipetted. The results for this technique showed that the thin film yielded an oriented crystalline morphology with efficient charge transport along the long axis of the crystallites, with the hole mobility reaching 0.83 cm2 V−1 s−1. This technique was shown to be robust and required a minimal amount of material. It can also be completed in a short time with simplified instrumentation. Hence, it could be a preferrable method for low-cost applications.
As mentioned above, it can be said that the dip coating deposition method does improve the continuity of the thin-film layer, which increases the uniformity of the thin film and results in less defects compared to the spin coating method. As discussed, this approach is preferable for various types of substrates that are flexible and irregular in shape, as this deposition method allows the solution to be spread over the substrate’s entire surface. The withdrawal speed is the main factor determining the thickness of the film. Other factors such as viscosity, liquid density, gravity, and surface tension can also influence the thickness. This is important, as a thin film’s thickness plays a role in enabling the carrier mobility flow. The withdrawal speed also influences the crystallinity growth of the thin film. Note that TIPS-pentacene was mainly used in the dip coating method. The crystalline growth of TIPS-pentacene thin films is important in producing a high carrier mobility rate. The optimum withdrawal speed and the type of solvent used when preparing the solution itself influence the formation of the thin film. This deposition method has shown much improvement as compared to the spin coating deposition method. However, this deposition method is not favorable due to the large volume of solutions used as the reservoir. This causes a lot of material waste, which conflicts with the aim of reducing the costs of the device fabrication process.
- Inkjet Printing Method
The inkjet printing method is a widely used approach for digital printing, allowing high precision and control. During inkjet printing, droplets of ink with adjusted viscosity are propelled through a nozzle under piezoelectric or thermal force, which are deposited onto a substrate [85–87]. This technique can be applied with a wide range of materials. However, it also relies on the solution having sufficiently high surface tension while having sufficiently low viscosity to allow the solution to flow through the nozzle head only when required to avoid leak. The quality of the solution is important in obtaining a uniform deposition.
For piezoelectric inkjet printing, the solution is deposited according to the voltage pulse applied to the piezo transducer by distorting the nozzle and creating pressure to force out a controlled amount of solution. To stop the flow, the polarity of the voltage is reversed, expanding the nozzle. This inkjet printing deposition method is suitable for complex morphologies and arrays of materials [88–93]. Figure 10 shows an illustration of the inkjet printing method.
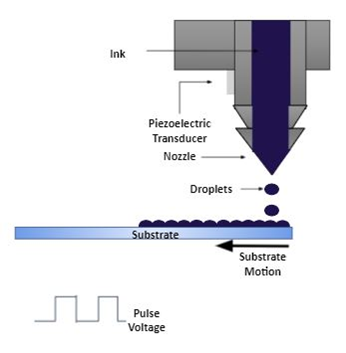
Figure 10. Inkjet printing method.
This method makes it possible for large-area deposition of various soluble materials. Since polymer blends have been known to overcome the difficulty of controlling crystal growth in soluble-processed TIPS-pentacene, as shown in previous studies [94], Cho et al. [51] investigated the behavior of TIPS-pentacene and amorphous polycarbonate (APC) in a polymer blend as the active layer using inkjet printing. Although the single-droplet inkjet printing of TIPS-pentacene with other polymer blends has been reported previously [95,96], the fabricated thin-film transistors showed issues in terms of film uniformity, especially when a geometrical parallel-type source drain was used, as the crystal orientation of the TIPS-pentacene in the single droplet was quite arbitrary.
The ratio of the blend was determined using the Flory–Huggins [97,98] and Cahn–Hilliard theories [99], whereby the Gibbs free energy when blending a polymer with TIPS-pentacene was evaluated. It is important to determine this ratio as it can affect the segregation strength between the TIPS-pentacene and the polymer phase. According to the theories, phase separation becomes easier with more positive Gibbs free energy values. In this study, the ratios were varied from 1:1 to 1:8 and were tested based on the OTFT performances. The highest mobility rate was obtained at 0.53 cm2 V−1 s−1 from the 1:4 ratio. This characteristic was observed to be closely related to the measurement of the phase separation strength between the TIPS-pentacene and the polymer phase. The crystal structures were then observed based on the various ratios. The crystals of inkjet-printed, non-blended TIPS-pentacene were highly anisotropic with various grain sizes, which led to a mobility rate of 0.22 cm2 V−1 s−1. The 1:1, 1:6, and 1:8 blended ratios produced plate-like wave crystalline structures with a random orientation, which resulted in carrier mobilities of 0.27 cm2 V−1 s−1, 0.35 cm2 V−1 s−1, and 0.17 cm2 V−1 s−1, respectively. Meanwhile, the 1:2 and 1:4 blended ratio produced stripe-shaped crystallite domains, resulting in carrier mobility rates of 0.46 cm2 V−1 s−1 and 0.53 cm2 V−1 s−1, respectively. This shows that the optimal TIPS-pentacene/APC mixing ratio induces strong phase separation and sequential crystal orientation, and it has a strong influence on the electrical properties of OTFTs.
The effect of the ink viscosity in wt.% on the device performance was examined [51]. The concentration was varied from 0.1 wt.% to 2.0 wt.% and it was found that the electrical performance of TIPS-pentacene/APC relied on the concentration as well as the viscosity of the inks. The average field-effect mobility seemed to increase from 0.04 cm2 V−1 s−1 to 0.53 cm2 V−1 s−1 when the concentrations were decreased from 2.0 wt.% to 1.0 wt.%. However, ink concentrations below 0.5 wt.% resulted in a lower mobility rate of 0.07 cm2 V−1 s−1 compared to the 1.0 wt.% and 1.5 wt.% concentrations at 0.53 cm2 V−1 s−1 (mentioned above) and 0.29 cm2 V−1 s−1, respectively. The inkjet-printed film with higher viscosity of 2.0 wt.% exhibited a broad and even distribution of TIPS-pentacene at all depth positions of the layer, with a slight occurrence of TIPS-pentacene at the top of the surface of the thin layer. The thin film with a 1.5 wt.% concentration showed a disconnected TIPS-pentacene layer in the top area, while at 1.0 wt.% the TIPS-pentacene was found to be discrete and showed a sharp phase separation in the upper area of the film. The OTFTs with the low-viscosity inks at 0.1 wt.% and 0.5 wt.% concentrations showed barely any distinguishable TIPS-pentacene layer at any position of the film, which meant that there were very small numbers of TIPS-pentacene crystals. Thus, ink viscosities lower than 0.5 wt.% does not produce a good OTFT performance.
Using the optimal ratio of 1:4, various solvents were tested for the TIPS-pentacene/APC solution, including toluene, toluene/chloroform, toluene/p-xylene, and toluene/tetralin. This was to investigate the effects of solvents on the field-effect mobility. The boiling point was the focus, as only 5 wt.% of the minor solvent was mixed with 95 wt.% of the major solvent (toluene), which was fixed. Toluene/p-xylene was shown to produce the highest mobility of 0.53 cm2 V−1 s−1 compared to toluene/chloroform, toluene, and toluene/tetralin, which exhibited carrier mobility rates up to 0.30, 0.31, and 0.40 cm2 V−1 s−1, respectively. This led to the observation of the thin films’ morphologies. It was shown that the Marangoni and convective flows of the drying ink are crucial for controlling the morphology of the inkjet droplets [100]. The high boiling point solvent, which was used as the major solvent, was mixed with a lower boiling point solvent, which was used as the minor solvent to control the evaporation rate of the droplets. As the evaporation rate increased at the edges of the droplets, this caused a buildup of convective flow from the center toward the drying edge of the droplets. This drove the surface tension gradient between the center and edge of the droplets. However, the opposing Marangoni flow arose in order to recirculate the solvents in the droplet. When the lower minor solvent boiling point was used, it produced a greater convective flow toward the drying edge, resulting in a higher edge wall of the droplet, which was known as the coffee ring effect. Thus, in this case, where a higher minor solvent boiling point was used, a weaker convective flow was created, with a smaller coffee ring effect. This behavior of the inkjet droplets influenced the morphology of the final printed thin film. Better-oriented TIPS-pentacene crystals were produced from the toluene/p-xylene solvents compared to the others. This also resulted in non-oriented large grain boundaries.
A low-voltage OTFT on a plastic substrate was fabricated by Lai et al. [53]. Again, TIPS-pentacene was used as the active channel, which was inkjet-printed using a 16 nozzle cartridge with a volume of 10 pL in a single drop. The solution was prepared at a concentration of 1.5 wt.% using anhydrous anisole as the solvent. Anisole was chosen because of its high boiling point of 153.8 C, which avoided evaporation before the printing process was completed. The printed organic semiconductor was dried in ambient air and completely dried after 1 min. The fabricated device was able to operate at a low voltage of less than 5 V, with a carrier mobility of 0.22 cm2 V−1 s−1. This study showed that the possibility of using multiple-nozzle inkjet printing on a plastic substrate to fabricate OTFTs and to obtain good carrier mobility. The performance levels of other inkjet-printed OFETs are listed in Table 1 [54].
It can be recognized that the inkjet printing deposition method is a modernized method that provides uniform thin films and potentially well-oriented crystal growth. This is due to the efficient use of the materials and almost complete lack of defects such as pinholes that can be formed on the thin films, as previously reported. Previous studies have revolved more around the concentrations of materials used for the deposition process and the inkjet printer configurations. The inkjet process is costly in terms of its machinery; however, it does allow high-quality thin-film deposition at a faster rate and allows mass production. It is very suitable for use in industrial production sites. There is no material waste when it comes to this deposition method, as it uses the materials efficiently (additive versus subtractive fabrication). There is no limitation on the types of materials used, as one is also able to use polymer blends with this printing method. Flexible substrates have also been studied with this deposition method, indicating its suitability for roll-to-roll fabrication, which could be considerably cheaper.
An important thing to mention is that the rest of the techniques discussed in this manuscript are all blanket deposition approaches, whereas the inkjet method is a “pattern” deposition approach, meaning that the deposition pattern can be produced in specific area with a specific shape, without using a mask or photoresist as the template. This offers a great advantage compared to other deposition methods and potentially reduces the fabrication steps and costs.
Conclusion
This manuscript compares various commonly used deposition methods that are associated with pentacene and its derivatives, including the classic thermal vacuum evaporation method, which is the initial method used for thin-film deposition. This approach uses pure pentacene in solid form to produce a uniform thin film, which results in good device performance. However, the disadvantage of this deposition method is the cost of the equipment and materials, as the specified amount of material can only be used in a single deposition process as compared to the soluble-process deposition method, where the same amount can be used for multiple depositions. This limits its production rate and output, as it is performed in a small vacuum chamber. For these reasons, when aiming to achieve low-cost fabrication at mass production rates, soluble process deposition methods are used. The commonly used soluble process deposition method in lab environment is the spin coating process. This is favorable as the cost of the equipment is not as expensive as the equipment used for thermal vacuum evaporation. It also does not require a large volume of source material to be used in its deposition process, which minimizes material waste. Hence, it is suitable for multiple device fabrication on a single substrate by using a small amount of source material. It forms a uniform thin-film layer, which can be enhanced with the use of surface treatments. The crystalline growth of the material in the thin film is also excellent, although the crystal orientation is highly influenced by the centrifugal force during the spinning process. This method has several disadvantages, as the outcome of the deposited layer is uncertain and is vulnerable to chemical reactions with the ambient environment. This issue is overcome by utilizing a glove box in an inert environment. The drop casting method is usually used for deposition on small substrates. It is preferable for the drafting of newly proposed devices and is suitable for flexible and conformable substrates. It has also been shown to produce good crystalline growth due to the small concentrated area involved. Nevertheless, the uniformity of the thin film itself is not guaranteed, as it is influenced by the gravitational force and the surface tension of the solution itself. This is a low-cost deposition method, as it uses only a small volume of material and is not normally used for mass production, which makes it a preferable method when it comes to new device structures or proposals. The dip coating method produces very uniform thin-film layers with good crystalline growth. This method is suitable for deposition on flexible and irregularly shaped substrates. The only downside of this deposition method is that it is not suitable for mass production, as it requires a large reservoir. This is costly and results in a large amount of material waste. Lastly, the inkjet printing method does not focus on the thin-film layer; instead, it forms a uniform and crystalline thin-film layer. This eliminates material waste as it uses the material in an efficient manner. It is suitable to use this method when a change of the device structure is the main variable in the study. Although the equipment is costly, it is well refined for the soluble process method and can also be used for mass production with high production speeds. The inkjet printing technology is mature, and thanks to this it can be integrated within organic electronics field. Every deposition method has its specified purposes, and they can be used according to their suitability for the desired device. All these deposition methods are still being used in ongoing research studies to find ways to improve and enhance their usage.
References:
- Lin, Y.-Y.; Gundlach, D.J.; Nelson, S.F.; Jackson, T.N. Stacked pentacene layer organic thin-film transistors with improved characteristics. IEEE Electron Device Lett. 1997, 18, 606–608.
- Park, S.K.; Jackson, T.N.; Anthony, J.E.; Mourey, D.A. High mobility solution processed 6, 13-bis (triisopropyl-silylethynyl) pentacene organic thin film transistors. Phys. Lett. 2007, 91, 63514.
- Kitamura, M.; Imada, T.; Arakawa, Y. Organic light-emitting diodes driven by pentacene-based thin-film transistors. Phys. Lett. 2003, 83, 3410–3412.
- Wolak, M.A.; Delcamp, J.; Landis, C.A.; Lane, P.A.; Anthony, J.; Kafafi, Z. High‐Performance Organic Light‐Emitting Diodes Based on Dioxolane‐Substituted Pentacene Derivatives. Funct. Mater. 2006, 16, 1943–1949.
- Zhuo, M.; Chen, Y.; Sun, J.; Zhang, H.; Guo, D.; Zhang, H.; Li, Q.; Wang, T.; Wan, Q. Humidity sensing properties of a single Sb doped SnO2 nanowire field effect transistor. Actuators B Chem. 2013, 186, 78–83.
- Noh, Y.-Y.; Kim, D.-Y. Organic phototransistor based on pentacene as an efficient red light sensor. Solid-State Electron. 2007, 51, 1052–1055.
- Intaniwet, A.; Keddie, J.L.; Shkunov, M.; Sellin, P.J. High charge-carrier mobilities in blends of poly (triarylamine) and TIPS-pentacene leading to better performing X-ray sensors. Electron. 2011, 12, 1903–1908.
- Ji, T.; Jung, S.; Varadan, V.K. Field-controllable flexible strain sensors using pentacene semiconductors. IEEE Electron Device Lett. 2007, 28, 1105–1107.
- Kim, J.-M.; Jha, S.K.; Chand, R.; Lee, D.-H.; Kim, Y.-S. DNA hybridization sensor based on pentacene thin film transistor. Bioelectron. 2011, 26, 2264–2269.
- Azeman, N.H.; Ahmad Khusaini, M.A.; Daik, R.; Ismail, A.G.; Yeop Majlis, B.; Mat Salleh, M.; T. H. Tg Abdul Aziz, T.H.; A Bakar, A.A.; Md Zain, A.R.; Teh, C.-H. Synthesis of a novel 1, 4‐bis [2‐(5‐thiophen‐2‐yl)‐1‐benzothiophene]‐2, 5‐dioctyloxybenzene pentamer for creatinine detection. Asian J. Org. Chem. 2021, 10, 2406. doi.org/10.1002/ajoc.202100374.
- Nashruddin, S.N.A.; Abdullah, J.; Mohammad Haniff, M.A.S.; Mat Zaid, M.H.; Choon, O.P.; Mohd Razip Wee, M.F. Label Free Glucose Electrochemical Biosensor Based on Poly(3,4-ethylenedioxy thiophene):Polystyrene Sulfonate/Titanium Carbide/Graphene Quantum Dots. Biosensors. 2021, 11, 267. doi.org/10.3390/bios11080267.
- Wan Khalid, W.E.F.; Heng, L.Y. HENG; Mat Arip, M.N. Surface Modification of Cellulose Nanomaterial for Urea Biosensor Application. Sains Malaysiana. 2018, 47(5), 941-949. doi.org/10.17576/jsm-2018-4705-09.
- Tang, Q.; Zhang, D.; Wang, S.; Ke, N.; Xu, J.; Yu, J.C.; Miao, Q. A meaningful analogue of pentacene: Charge transport, polymorphs, and electronic structures of dihydrodiazapentacene. Mater. 2009, 21, 1400–1405.
- Bhatia, R.; Wadhawa, D.; Gurtu, G.; Gaur, J.; Gupta, D. Methodologies for the synthesis of pentacene and its derivatives. Saudi Chem. Soc. 2019, 23, 925–937.
- Kim, H.G.; Choi, H.H.; Song, E.; Cho, K.; Choi, E.-J. Synthesis, stability and electrical properties of new soluble pentacenes with unsaturated side groups. RSC Adv. 2015, 5, 8070–8076.
- Benor, A.; Hoppe, A.; Wagner, V.; Knipp, D. Electrical stability of pentacene thin film transistors. Electron. 2007, 8, 749–758.
- Sun, Y.; Liu, Y.; Zhu, D. Advances in organic field-effect transistors. Mater. Chem. 2005, 15, 53–65.
- Dey, A.; Singh, A.; Das, D.; Iyer, P.K. Organic Semiconductors: A new future of nanodevices and applications. In Thin Film Structures in Energy Applications; Springer: Berlin, Germany, 2015; pp. 97–128.
- Kowarik, S.; Gerlach, A.; Schreiber, F. Organic molecular beam deposition: Fundamentals, growth dynamics, and in situ studies. Phys. Condens. Matter 2008, 20, 184005.
- Glynn, C.; O’Dwyer, C. Solution processable metal oxide thin film deposition and material growth for electronic and photonic devices. Mater. Interfaces 2017, 4, 1600610.
- Wit, B. Defects in Pentacene Thin Films Grown by Supersonic Molecular Beam Deposition; University of Groningen: Groningen, The Netherlands.
- Gong, X.; Xin, M.; Li, M.; Yuan, H.; Xie, J. Compatibilizer improving properties of tea dust/polylactic acid biomass composites. Nongye Gongcheng Xuebao Trans. Chin. Soc. Agric. Eng. 2017, 33, 308–314, doi:10.11975/j.issn.1002-6819.2017.02.043.
- Xu, J.; Zhu, X.; Tan, S.; Zhang, Y.; Li, B.; Tian, Y.; Shan, H.; Cui, X.; Zhao, A.; Dong, Z. Determining structural and chemical heterogeneities of surface species at the single-bond limit. Science 2021, 371, 818–822.
- Navarro, F.F.; Djurovich, P.I.; Thompson, M.E. Metal deposition for optoelectronic devices using a low vacuum vapor phase deposition (VPD) system. Electron. 2014, 15, 3052–3060.
- Burrows, P.E.; Forrest, S.R.; Sapochak, L.S.; Schwartz, J.; Fenter, P.; Buma, T.; Ban, V.S.; Forrest, J.L. Organic vapor phase deposition: A new method for the growth of organic thin films with large optical non-linearities. Cryst. Growth 1995, 156, 91–98.
- Ochiai, S.; Palanisamy, K.; Kannappan, S.; Shin, P.-K. Pentacene active channel layers prepared by spin-coating and vacuum evaporation using soluble precursors for OFET applications. Sch. Res. Netw. Condens. Matter Phys. 2012, 2012, 313285.
- Liao, K.; Ismail, A.G.; Kreplak, L.; Schwartz, J.; Hill, I.G. Designed Organophosphonate Self‐Assembled Monolayers Enhance Device Performance of Pentacene‐Based Organic Thin‐Film Transistors. Mater. 2010, 22, 3081–3085.
- Yang, C.-Y.; Cheng, S.-S.; Ou, T.-M.; Wu, M.-C.; Wu, C.-H.; Chao, C.-H.; Lin, S.-Y.; Chan, Y.-J. Pentacene-based planar-and vertical-type organic thin-film transistor. IEEE Trans. Electron Devices 2007, 54, 1633–1636.
- Guo, E.; Xing, S.; Dollinger, F.; Wu, Z.; Tahn, A.; Löffler, M.; Leo, K.; Kleemann, H. High‐Performance Static Induction Transistors Based on Small‐Molecule Organic Semiconductors. Mater. Technol. 2020, 5, 2000361.
- Hiroki, M.; Maeda, Y.; Ohmi, S. Top-gate pentacene-based organic field-effect transistor with amorphous rubrene gate insulator. J. Appl. Phys. 2018, 57, 02CA08.
- Sheraw, C.D.; Jackson, T.N.; Eaton, D.L.; Anthony, J.E. Functionalized pentacene active layer organic thin‐film transistors. Mater. 2003, 15, 2009–2011.
- Lee, T.; Lim, B.; Yong, K.; Kwon, W.; Park, M. Effects of oxygen plasma generated in magnetron sputtering of ruthenium oxide on pentacene thin film transistors. Korean J. Chem. Eng. 2017, 34, 2502–2506.
- Da Silva Ozório, M.; Nogueira, G.L.; Morais, R.M.; da Silva Martin, C.; Constantino, C.J.L.; Alves, N. Poly (3-hexylthiophene): TIPS-pentacene blends aiming transistor applications. Thin Solid Films 2016, 608, 97–101.
- Ozório, M.S.; Camacho, S.A.; Cordeiro, N.J.A.; Duarte, J.L.; Alves, N. Solvent Effect on Morphology and Optical Properties of Poly (3-hexylthiophene): TIPS-Pentacene Blends. Electron. Mater. 2018, 47, 1353–1361.
- Birnie, D.P., III A model for drying control cosolvent selection for spin-coating uniformity: The thin film limit. Langmuir 2013, 29, 9072–9078.
- Wang, S.; Zhao, X.; Tong, Y.; Tang, Q.; Liu, Y. Directly Spin Coating a Low‐Viscosity Organic Semiconductor Solution onto Hydrophobic Surfaces: Toward High‐Performance Solution‐Processable Organic Transistors. Mater. Interfaces 2020, 7, 1901950.
- Yoo, H.; Choi, H.H.; Shin, T.J.; Rim, T.; Cho, K.; Jung, S.; Kim, J. Self‐Assembled, Millimeter‐Sized TIPS‐Pentacene Spherulites Grown on Partially Crosslinked Polymer Gate Dielectric. Funct. Mater. 2015, 25, 3658–3665.
- Kim, H.S.; Park, J.H.; Lee, W.H.; Kim, H.H.; Park, Y.D. Tailoring the crystallinity of solution-processed 6, 13-bis (triisopropylsilylethynyl) pentacene via controlled solidification. Soft Matter 2019, 15, 7369–7373.
- Raghuwanshi, V.; Bharti, D.; Tiwari, S.P. Flexible organic field-effect transistors with TIPS-Pentacene crystals exhibiting high electrical stability upon bending. Electron. 2016, 31, 177–182.
- Shih, A.; Akinwande, A.I. Solution-processed high-voltage organic thin film transistor. MRS Adv. 2017, 2, 2961–2966.
- Shih, A.; Akinwande, A.I. Solution-Processed and Self-Assembled Monolayer-Treated High-Voltage Organic Thin Film Transistors for Flexible MEMS Integration. MRS Adv. 2018, 3, 1877–1882.
- Raghuwanshi, V.; Bharti, D.; Mahato, A.K.; Varun, I.; Tiwari, S.P. Semiconductor: Polymer blend ratio dependent performance and stability in low voltage flexible organic field-effect transistors. Met. 2018, 236, 54–60.
- Zhou, Y.; Wang, H.; Tang, Q.; Tong, Y.; Zhao, X.; Liu, Y. Solution-Processed Single-Crystal Array for High-Performance Conformable Transistors. IEEE Electron Device Lett. 2018, 39, 595–598.
- Lago, N.; Buonomo, M.; Imran, S.; Bertani, R.; Wrachien, N.; Bortolozzi, M.; Pedersen, M.G.; Cester, A. TIPS-pentacene as biocompatible material for solution processed high-performance electronics operating in water. IEEE Electron Device Lett. 2018, 39, 1401–1404.
- Raghuwanshi, V.; Bharti, D.; Mahato, A.K.; Varun, I.; Tiwari, S.P. TIPS-Pentacene: PS Blend Organic Field-Effect Transistors with Hybrid Gate Dielectric on Paper Substrate. In Proceedings of the 2019 International Symposium on VLSI Technology, Systems and Application (VLSI-TSA), Hsinchu,Taiwan, 22–25 April 2019; IEEE: Piscataway, NJ, USA, 2019; pp. 1–5. doi.org/10.1109/VLSI-TSA.2019.8804675.
- He, Z.; Zhang, Z.; Bi, S.; Asare-Yeboah, K.; Chen, J.; Li, D. A facile and novel route to improve TIPS pentacene based organic thin film transistor performance with elastomer. Met. 2020, 262, 116337.
- Raghuwanshi, V.; Bharti, D.; Varun, I.; Mahato, A.K.; Tiwari, S.P. Performance enhancement in mechanically stable flexible organic-field effect transistors with TIPS-pentacene: Polymer blend. Electron. 2016, 34, 284–288.
- Wang, W.; Wang, L.; Dai, G.; Deng, W.; Zhang, X.; Jie, J.; Zhang, X. Controlled growth of large-area aligned single-crystalline organic nanoribbon arrays for transistors and light-emitting diodes driving. Nano-Micro Lett. 2017, 9org/10.1007/s40820-017-0153-5.
- Manaka, T.; Iwamoto, M. Spectroscopic Imaging for Studying Carrier Behaviour in Organic Semiconductor Thin Films. In Proceedings of the 2018 12th International Conference on Advanced Semiconductor Devices and Microsystems (ASDAM), Smolenice, Slovakia, 21–24 October 2018; IEEE: Piscataway, NJ, USA, 2018; pp. 1–4. doi.org/10.1109/ASDAM.2018.8544578.
- Yoshimoto, S.; Takahashi, K.; Suzuki, M.; Yamada, H.; Miyahara, R.; Mukai, K.; Yoshinobu, J. Highly anisotropic mobility in solution processed TIPS-pentacene film studied by independently driven four GaIn probes. Phys. Lett. 2017, 111, 73301.
- Cho, S.Y.; Ko, J.M.; Lim, J.; Lee, J.Y.; Lee, C. Inkjet-printed organic thin film transistors based on TIPS pentacene with insulating polymers. Mater. Chem. C 2013, 1, 914–923.
- Kim, J.S.; Song, C.K. Patterning process of ink-jet printed 6, 13-bis (triisopropylsilylethynyl) pentacene layer using bank structures for organic thin film transistors. Thin Solid Films 2015, 589, 620–626
- Lai, S.; Cosseddu, P.; Zucca, A.; Loi, A.; Bonfiglio, A. Combining inkjet printing and chemical vapor deposition for fabricating low voltage, organic field-effect transistors on flexible substrates. Thin Solid Films 2017, 631, 124–131.
- Ge, F.; Wang, X.; Zhang, Y.; Song, E.; Zhang, G.; Lu, H.; Cho, K.; Qiu, L. Modulating the Surface via Polymer Brush for High‐Performance Inkjet‐Printed Organic Thin‐Film Transistors. Electron. Mater. 2017, 3, 1600402.
- Saikia, D.; Sarma, R. Fabrication and Characterization of Organic Light Emitting Diode Using FTO/Pentacene as Bilayer Anode. Sci. Ind. J. 2016, 14, 103.
- Biber, M.; Aydoğan, Ş.; Çaldıran, Z.; Çakmak, B.; Karacalı, T.; Türüt, A. The influence of annealing temperature and time on the efficiency of pentacene: PTCDI organic solar cells. Results Phys. 2017, 7, 3444–3448.
- Zhang, X.; Li, M.; Dall’Agnese, C.; Chen, G.; Wang, X.-F.; Miyasaka, T. Thermo-evaporated pentacene and perylene as hole transport materials for perovskite solar cells. Dyes Pigments 2019, 160, 285–291.
- Wolak, M.A.; Jang, B.-B.; Palilis, L.C.; Kafafi, Z.H. Functionalized pentacene derivatives for use as red emitters in organic light-emitting diodes. Phys. Chem. B 2004, 108, 5492–5499.
- Rao, M.V.M.; Huang, T.-S.; Su, Y.-K.; Huang, Y.-T. Fullerene and pentacene as a pure organic connecting layer in tandem organic light emitting devices. Electrochem. Soc. 2009, 157, H69.
- Ismail, A.G. Photolithographically patterned N-channel organic thin film transistors using sensitized polyvinyl alcohol. Electron. 2018, 56, 111–115, doi:10.1016/j.orgel.2018.02.006.
- Van Franeker, J.J.; Hermida‐Merino, D.; Gommes, C.; Arapov, K.; Michels, J.J.; Janssen, R.A.J.; Portale, G. Sub‐Micrometer Structure Formation during Spin Coating Revealed by Time‐Resolved In Situ Laser and X‐Ray Scattering. Funct. Mater. 2017, 27, 1702516. doi.org/10.1002/adfm.201702516.
- Han, D.H.; Kim, D.; Yun, H.W.; Lee, J.; Lee, U.G.; Chung, H.S.; Kim, W.-B. Effects of aging on the thickness of a homogeneous film fabricated using a spin coating process. Coat. Technol. Res. 2021, 18, 641–647. doi.org/10.1007/s11998-020-00429-x.
- Lee, U.G.; Kim, W.-B.; Han, D.H.; Chung, H.S. A Modified Equation for Thickness of the Film Fabricated by Spin Coating. Symmetry 2019, 11, 1183.
- Rueda-Delgado, D.; Hossain, I.M.; Jakoby, M.; Schwenzer, J.A.; Abzieher, T.; Howard, I.A.; Richards, B.S.; Lemmer, U.; Paetzold, U.W. Solution-processed and evaporated C60 interlayers for improved charge transport in perovskite photovoltaics. Electron. 2020, 77, 105526.
- Dissanayake, N.; Abeysundara, S.; Wanasekara, N.D. Investigating the Feasibility of Applying Spin Coating Method for Textiles. In Proceedings of the 2020 Moratuwa Engineering Research Conference (MERCon), Moratuwa, Sri Lanka, 27–29 July 2020; IEEE: Piscataway, NJ, USA, 2020; pp. 448–452. doi.org/10.1109/MERCon50084.2020.9185265.
- Bharti, D.; Tiwari, S.P. Improved alignment and crystallinity of TIPS-Pentacene thin films by off-center spin coating. In Proceedings of the 2015 IEEE 15th International Conference on Nanotechnology (IEEE-NANO), Rome, Italy, 27–30 July 2015; IEEE: Piscataway, NJ, USA, 2015; pp. 432–435. doi.org/10.1109/NANO.2015.7388629.
- Akkerman, H.B.; Li, H.; Bao, Z. TIPS-pentacene crystalline thin film growth. Electron. 2012, 13, 2056–2062.
- He, Z. Tips Pentacene Crystal Alignment for Improving Performance of Solution Processed Organic Thin Film Transistors. Ph.D. Thesis University of Alabama, Tuscaloosa, AL, USA, 2014.
- Xiao, C.; Kan, X.; Liu, C.; Jiang, W.; Zhao, G.; Zhao, Q.; Zhang, L.; Hu, W.; Wang, Z.; Jiang, L. Controlled formation of large-area single-crystalline TIPS-pentacene arrays through superhydrophobic micropillar flow-coating. Mater. Chem. C 2017, 5, 2702–2707.
- Madec, M.-B.; Smith, P.J.; Malandraki, A.; Wang, N.; Korvink, J.G.; Yeates, S.G. Enhanced reproducibility of inkjet printed organic thin film transistors based on solution processable polymer-small molecule blends. Mater. Chem. 2010, 20, 9155–9160.
- Lee, M.W.; Ryu, G.S.; Lee, Y.U.; Pearson, C.; Petty, M.C.; Song, C.K. Control of droplet morphology for inkjet-printed TIPS-pentacene transistors. Eng. 2012, 95, 1–4.
- Liu, D.; Xu, X.; Su, Y.; He, Z.; Xu, J.; Miao, Q. Self‐Assembled Monolayers of Phosphonic Acids with Enhanced Surface Energy for High‐Performance Solution‐Processed N‐Channel Organic Thin‐Film Transistors. Chem. 2013, 125, 6342–6347.
- Asiri, A.M.; Chani, M.T.S.; Khan, S.B. Method of Making Thin Film Humidity Sensors. U.S. Patent 9976975B2, 2018.
- Eslamian, M. Inorganic and organic solution-processed thin film devices. Nano-Micro Lett. 2017, 9org/10.1007/s40820-016-0106-4.
- Lee, T.D.; Ebong, A.U. A review of thin film solar cell technologies and challenges. Sustain. Energy Rev. 2017, 70, 1286–1297.
- Park, J.-M.; Sohn, I.-B.; Kang, C.; Kee, C.-S.; Hwang, I.-W.; Yoo, H.K.; Lee, J.W. Terahertz modulation using TIPS-pentacene thin films deposited on patterned silicon substrates. Commun. 2016, 359, 349–352.
- Olcer, Y.A.; Tascon, M.; Eroglu, A.E.; Boyacı, E. Thin film microextraction: Towards faster and more sensitive microextraction. TrAC Trends Anal. Chem. 2019, 113, 93–101.
- Hilliard, S.; Baldinozzi, G.; Friedrich, D.; Kressman, S.; Strub, H.; Artero, V.; Laberty-Robert, C. Mesoporous thin film WO3 photoanode for photoelectrochemical water splitting: A sol–gel dip coating approach. Energy Fuels 2017, 1, 145–153.
- Miao, S.S.; Wu, M.S.; Ma, L.Y.; He, X.J.; Yang, H. Electrochemiluminescence biosensor for determination of organophosphorous pesticides based on bimetallic Pt-Au/multi-walled carbon nanotubes modified electrode. Talanta 2016, 158, 142–151, doi:10.1016/j.talanta.2016.05.030.
- Wu, K.; Li, H.; Li, L.; Zhang, S.; Chen, X.; Xu, Z.; Zhang, X.; Hu, W.; Chi, L.; Gao, X. Controlled growth of ultrathin film of organic semiconductors by balancing the competitive processes in dip-coating for organic transistors. Langmuir 2016, 32, 6246–6254.
- Liu, X.; Zhang, Y.; Zhang, X.; Li, R.; Hu, W. Continuous and highly ordered organic semiconductor thin films via dip-coating: The critical role of meniscus angle. China Mater. 2020, 63, 1257–1264.
- Ravariu, C.; Mihaiescu, D.; Morosan, A.; Vasile, B.S.; Purcareanu, B. Sulpho-Salicylic Acid Grafted to Ferrite Nanoparticles for n-Type Organic Semiconductors. Nanomaterials 2020, 10, 1787.
- Morosan, A.; Mihaiescu, D.E.; Istrati, D.; Voicu, G.; Fudulu, A.; Stan, R. Polar shell magnetic nanostructured systems for heterogeneous nanophase reactions. UPB Sci. Bull. Ser. B 2018, 80, 53–64.
- Yang, T.; Mehta, J.S.; Haruk, A.M.; Mativetsky, J.M. Targeted deposition of organic semiconductor stripes onto rigid, flexible, and three-dimensional substrates. Mater. Chem. C 2018, 6, 2970–2977.
- Zhou, Z.; Cantu, L.R.; Chen, X.; Alexander, M.R.; Roberts, C.J.; Hague, R.; Tuck, C.; Irvine, D.; Wildman, R. High-throughput characterization of fluid properties to predict droplet ejection for three-dimensional inkjet printing formulations. Manuf. 2019, 29, 100792.
- Winoto, A. Methods and Compositions for Ink Jet Deposition of Conductive Features. U.S. Patent 8632700B2,
- Mościcki, A.; Fałat, T.; Smolarek, A.; Kinart, A.; Felba, J.; Borecki, J. Interconnection process by ink jet printing method. In Proceedings of the 2012 12th IEEE International Conference on Nanotechnology (IEEE-NANO), Birmingham, UK, 20–23 August; IEEE: Piscataway, NJ, USA, 2012; pp. 1–5. doi.org/10.1109/NANO.2012.6322108.
- Mikami, Y.; Yoshioka, H.; Oki, Y. Fully room temperature and label free biosensing based on an ink-jet printed polymer microdisk laser. Mater. Express 2021, 11, 592–602.
- Muniz, N.O.; Vechietti, F.A.; dos Santos, L.A.L. Influence of several binders on the mechanical properties of alumina parts manufactured by 3D inkjet printing. Res. Express 2019, 6, 115341.
- Jeong, S.; Kim, D.; Moon, J. Ink-Jet-printed organic—Inorganic hybrid dielectrics for organic thin-film transistors. Phys. Chem. C 2008, 112, 5245–5249.
- Wen, Y.X.; Liu, S.G.; Tao, B.X.; Luo, H.Q.; Li, N.B. A signal-off photocathode biosensor based on a novel metal-organic polymer for the detection of glucose. Actuators B Chem. 2020, 304, 127279.
- Xiang, C.; Wu, L.; Lu, Z.; Li, M.; Wen, Y.; Yang, Y.; Liu, W.; Zhang, T.; Cao, W.; Tsang, S.-W. High efficiency and stability of ink-jet printed quantum dot light emitting diodes. Commun. 2020, 11 doi.org/10.1038/s41467-020-15481-9.
- Zhang, G.; Zhang, P.; Chen, H.; Guo, T. Modification of polymer gate dielectrics for organic thin-film transistor from inkjet printing. Phys. A 2018, 124, 481. doi.org/10.1007/s00339-018-1899-4.
- Kwon, Y.-J.; Park, Y.D.; Lee, W.H. Inkjet-printed organic transistors based on organic semiconductor/insulating polymer blends. Materials 2016, 9, 650.
- Li, X.; Smaal, W.T.T.; Kjellander, C.; van der Putten, B.; Gualandris, K.; Smits, E.C.P.; Anthony, J.; Broer, D.J.; Blom, P.W.M.; Genoe, J. Charge transport in high-performance ink-jet printed single-droplet organic transistors based on a silylethynyl substituted pentacene/insulating polymer blend. Electron. 2011, 12, 1319–1327.
- James, D.T.; Kjellander, B.K.C.; Smaal, W.T.T.; Gelinck, G.H.; Combe, C.; McCulloch, I.; Wilson, R.; Burroughes, J.H.; Bradley, D.D.C.; Kim, J.-S. Thin-film morphology of inkjet-printed single-droplet organic transistors using polarized Raman spectroscopy: Effect of blending TIPS-pentacene with insulating polymer. Chem. Soc. Nano 2011, 5, 9824–9835.
- Flory, P.J. Thermodynamics of high polymer solutions. Chem. Phys. 1942, 10, 51–61.
- Huggins, M.L. The viscosity of dilute solutions of long-chain molecules. IV. Dependence on concentration. Am. Chem. Soc. 1942, 64, 2716–2718.
- Michels, J.J. Surface‐Directed Spinodal Decomposition of Solvent‐Quenched Organic Transistor Blends. ChemPhysChem 2011, 12, 342–348.
- Lim, J.A.; Lee, H.S.; Lee, W.H.; Cho, K. Control of the morphology and structural development of solution‐processed functionalized acenes for high‐performance organic transistors. Funct. Mater. 2009, 19, 1515–1525.
This entry is adapted from the peer-reviewed paper 10.3390/polym14061112
