2.1. The CRISPR-Cas Complex Role in the Immunity System
When investigating the
iap gene product in the opportunistic pathogen
Escherichia coli, Ishino et al. (1987) [
18] observed an atypical structure, specifically the repetition of several homologous sequences. Later, this type of structure was observed in various bacterial, as well as archaeal strains [
19,
20]. Subsequently, these repetitive sequences were linked with exogenous genetic material, and following several years, their assembling mechanism and function were elucidated [
21]. This type of sequence can be placed on the chromosomal DNA, but it can also be found on the plasmid DNA [
22].
The scientists demonstrated that CRISPR-Cas, which is present in one-third of bacteria and nearly in all archaea, has a key role in host’s adaptive immunity. It protects the organism against various intruders, such as viruses, but it also offers protection against other mobile genetic elements, such as transposons or plasmids [
23].
The CRISPR-Cas system structure includes three main components, i.e., the CRISPR arrays, the associated Cas proteins, and the leader nucleotide sequence. The first genetic component, the CRISPR locus, is characterized by identical repeats structures (21–37 bp) that are highly conserved and the spacer sequences that are acquired fragments of invader’s nucleic acid material. The CRISPR array is located downstream from
cas genes. The latter encodes for Cas proteins that are crucial to the immune reaction [
24].
Initially, only four distinct Cas proteins (1–4) were reported, due to the rapid evolution of biological sciences; currently, there numerous Cas proteins have been described [
25,
26], Cas1 being the most analyzed [
27].
CRISPR-Cas has a great adaptability, with host-related specificities; thus, it exhibits a significant diversity. The varying feature is defined by the CRISPR array and the
cas gene sequences. The classification of these types of systems is based on the signature Cas proteins. Currently, there are two major classes of CRISPR-Cas systems, each also di-vided in several groups [
28]. Regarding the leader nucleotide sequence, it has been shown that it has a key role by carrying the essential promoter sequences for the transcription of CRISPR loci. Besides the promoter, the leader contains specific signals that are crucial for the adaptation stage from the first phase of CRISPR-Cas activation [
29].
The adaptation is the first functional stage of the CRISPR-Cas mechanism, during which the foreign nucleic acid is recognized by several Cas proteins [
30] and consequently integrated next to a leader sequence. Through this mechanism, in evolution, the spacers are arranged chronologically, and this feature helps bacteria and archaea to enhance their protection against the genetic material of the latest foreign encounter [
31]. Each new acquired spacer is accompanied by a repeat sequence; therefore, the CRISPR array expands with every invasion [
32].
The CRISPR array is transcribed in the second step, specifically in the biogenesis phase [
33]. First of all, it is being transcribed into a precursor CRISPR RNA (crRNA). At the end of this phase, there are numerous mature crRNAs molecules, resulting from the action of RNase III that process the precursor crRNA. Each crRNA includes a spacer and a repeat sequence [
31,
34,
35].
The last step of the system’s mechanism is the interference phase. It involves the degradation of the foreign nucleic acid, by targeting and cleaving it [
36]. The products of the biogenesis phase, the crRNAs, act like guides for targeting the invader, which is then cleaved following a Cas proteins cascade that act like molecular scissors [
37].
2.2. The CRISPR-Cas System as a Genome Editing Tool
When it comes to leading tools in genetic engineering, the CRISPR-Cas system can be considered the foremost instrument. After elucidating its function in various organisms, scientists aimed to exploit its versatility, in order to overcome the disadvantages of other available genome editing tools [
38]. Even if scientific studies still report the use of ZFNs and TALENs techniques as editing tools, the CRISPR-Cas system is the most effective genome editing instrument, standing on top in regards to efficiency, cost-effectiveness, and the relative simplicity of use [
39] (
Table 1). Another considerable advantage of this system is represented by its capacity to simultaneously target multiple genes [
40].
Table 1. Comparison between TALEN, ZFN, and CRISPR-Cas gene editing technologies.
|
Traits
|
TALEN
|
ZFN
|
CRISPR-Cas
|
References
|
|
Origin
|
Prokaryotic
|
Eukaryotic
|
Prokaryotic
|
[41]
|
|
Efficiency (%)
|
76
|
12
|
81
|
[42]
|
|
Specificity
|
Moderate
|
Low
|
High
|
[43,44,45,46]
|
|
Target site recognition
|
Any site
|
Any site
|
Pam motif (NGG) required
|
[43]
|
|
Multiplex potential
|
Low
|
Low
|
High
|
[43,45]
|
|
Processing time
|
Time consuming
|
Time consuming
|
Short
|
[45]
|
|
Methylation sensitive
|
Sensitive
|
Sensitive
|
Not sensitive
|
[42]
|
|
Engineering feasibility
|
Moderate/High
|
Moderate
|
Moderate/High
|
[42,45]
|
|
Dimerization required
|
Yes
|
Yes
|
No
|
[44]
|
|
Cost effectiveness
|
Moderate
|
No
|
Yes
|
[43,44]
|
Of the numerous CRISPR-Cas systems, CRISPR-Cas9 is currently the most used instrument in laboratories across the world [
47]. The Cas9 nuclease is the signature protein of CRISPR-Cas II systems and it is responsible for double strand DNA breaks [
27]. Three different methods to deliver the Cas9 endonuclease have been described. It can be directly delivered by microinjection into the embryos, while the other two delivery methods involve a plasmid that expresses the Cas9 enzyme, or a messenger RNA (mRNA) sequence that encodes it. Of the three techniques, in terms of genome engineering, the earliest mentioned is the best option due to its certain advantages. By directly delivering the protein, low immunogen effects were observed. Furthermore, the off-target activity is minimized compared with the other two methods [
48]. The use of CRISPR-Cas9 is a simple but powerful genome editing tool, with various implementations, and their impact on new research trends has been reviewed elsewhere [
49].
The CRISPR-Cas9 mechanism relies on the Cas9 nuclease and a guide sequence (gRNA). As the name implies, the gRNA has the role to guide the Cas9 nuclease to a target site in order to cleave the DNA. The key feature of gRNA is the extensive complementarity with the target sequence [
50].The protospacer-adjacent motif (PAM) bordering the target complementary sequence has a key role, since in its absence, the CRISPR-Cas systems would degrade their own CRISPR loci. In order to perform a cleavage, the Cas9 protein scans for the PAM sequence. Even if the gRNA is complementary with the target sequence, the Cas9 endonuclease will not cleave it in the absence of PAM [
51].
The central factor that influences the success of the gene editing process is the repair path of the double-strand breaks produced by Cas9. There are two main repair pathways: the homology-directed repair (HDR) and the nonhomologous end joining (NHEJ), respectively [
52]. More often, NHEJ is exploited in order to acquire indels mutations, specially to obtain small deletions. These deletions are extremely useful for disclosing gene functions [
53]. However, the HDR machinery is used not just to obtain knock-out or knock-down mutations, such as the expected output following NHEJ, but to generate target knock-ins. Therefore, by using HDR, exogenous sequences can be successfully integrated into the host’s genome. Currently, major efforts are being made in order to enhance the sequence replacement by using the HDR mechanism [
54].
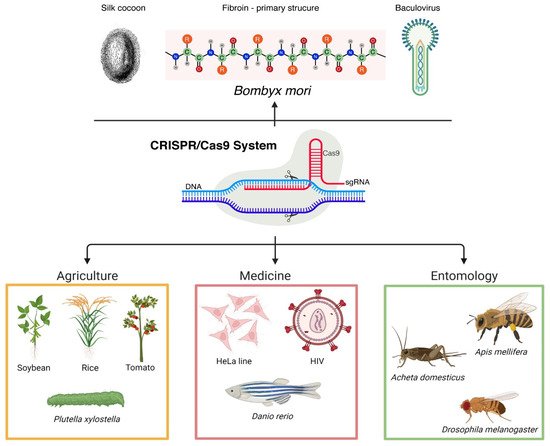
CRISPR-Cas9 is currently used in multiple research fields, such as agriculture (editing of various agricultural plant genomes or pest insect’s genome) [
55,
56,
57], biotechnology, food industry, and medicine (modeling diseases using HeLa cells, deciphering HIV infection mechanisms, using various experimental models, such as
Danio rerio to tackle cancer and neurological diseases, etc.) [
58,
59], just to mention a few (
Figure 1) [
60].
Figure 1. Schematic representation of the most important current applications of CRISPR-Cas9 in entomology, medicine, and agriculture. On top, a simplified description of CRISPR-Cas9 applicability in B. mori that is extensively described in the main text (created with BioRender.com, accessed on 2 December 2021).
2.3. CRISPR-Cas9 in Entomology
Being the most diverse and numerous category of organisms for decades [
61], insects have been intensively studied. Countless studies have been performed due to insects’ key roles in ecology, agriculture, and medicine [
62,
63]. Considering this, numerous research groups aimed to use the CRISPR-Cas9 system to manipulate the insects’ genome. The first application of CRISPR-Cas9 was performed in
Drosophila melanogaster [
64] due to its strategic importance as arguably the main experimental model organism for life sciences [
65]. Besides
D. melanogaster, the researchers also used the CRISPR-Cas9 applicability on
B. mori,
Apis mellifera,
Aedes aegypti, and
Tribolium castaneum [
66,
67].
Gratz et al. (2013) [
68] programmed CRISPR-Cas9 to edit
Drosophila’s genome. The authors targeted the
yellow gene, which is commonly used in various studies. First, they aimed to determine if this genome engineering tool could be efficient and could fulfill its role to induce breaks in the target sequence. By using the CRISPR-Cas9 system in
Drosophila, not only was the
yellow gene successfully knocked out, but the genome’s alterations were also germline transmitted. Subsequent to the deletion of the target gene, a donor sequence was designed. This sequence provided the template for the HDR repair pathway and its use was to test the accuracy of specific replacement of
yellow gene with an exogenous sequence. These sequence replacements were transmitted to descendants as well. Their data showed that there was no off-target activity and it highlighted the feasibility of using the CRISPR-Cas9 technology in eukaryotes [
68].
Aiming to further highlight the feasibility of choosing this system to perform genome alteration in
Drosophila, Yu et al. (2013) [
69] designed two gRNAs to induce mutations in two regions of the
yellow gene. In addition, they targeted other six sequences, both euchromatic and heterochromatic loci. Remarkably, a definite mutation in
ms(3)k81 was transmitted to descendants in a proportion of 100%. By successfully targeting heterochromatic loci, their result showed that the CRISPR-Cas9 system is efficient for altering the heterochromatin [
69].
Drosophila have been used in numerous studies in order to examine the insecticide resistance [
70,
71,
72]. In this direction, Douris et al. (2020) [
56] notably summarized the progress in using CRISPR-Cas9 to explore the genetic basis of this mechanism.
The CRISPR-Cas9 technique was used to perform functional analysis concomitantly on two genes belonging to the cricket (
Gryllus bimaculatus) [
73].
G. bimaculatus is an important insect for experimental studies; for example, it plays an important role for evolutionary developmental studies and comparative biology, but it is also a relevant model organism for neurobiology and behavioral sciences [
74]. The efficiency of inserting a donor sequence via a homology-independent technique was tested in two
hox genes, namely
Gb-Ubx and
Gb-abd-A. After inserting the donor fragment into essential exons of both genes, their function was lost. Thus, functional investigations of
hox genes could be carried out by using the knock-in/knock-out approaches [
73].
Being one of the most important social insects [
75] and as it plays a crucial role as a pollinator, the honeybee (
Apis mellifera) has been intensively studied. It also plays a pivotal role in various therapeutic areas due to honey production. This natural product has extraordinary benefits for human health, exhibiting antioxidant, antiviral, and antibacterial effects [
76]. Due to its special characteristics, the use of honey is not limited to humans, but this natural product is being used to improve certain features of other insects, such as silkworms [
77]. There are numerous studies that detail functional analysis of
A. mellifera genes by exploiting the CRISPR-Cas9 system. For instance, Hu et al. (2019) [
67] reported the successful utilization of this system for knocking out the
mrjp 1 gene from the honeybee genome. The CRISPR-Cas9 complex was delivered through microinjection and they tested two specific regions of embryos, for identifying the most convenient structure for delivering the gRNA and the Cas9 endonuclease. By microinjecting the construct at the dorsal posterior side, there was a low rate of successful manipulation (11.8%); however, when choosing the ventral cephalic side, the results showed a great rate of gene editing (93.3%). Trying to validate the previous results, the authors also targeted
pax6. Based on the previously obtained results, they microinjected the CRISPR-Cas9 construct at the ventral cephalic side. The results showed an editing rate of 100% [
67]. Targeting the same gene,
mrjp 1, similar results have been obtained in another study [
78]. Thus, functional analysis of
A. mellifera genes can be effectively performed by using the CRISPR-Cas9 system.
Considering the same topic of gene function research, Nie et al. (2021) [
79] used the CRISPR-Cas9 technology to determine if the
yellow-y gene plays a crucial role in the process of cuticular melanin synthesis in
A. mellifera. They targeted this gene due to its great potential for mutants screening, being a selectable marker. By disrupting it, the phenotype of worker cuticle has changed, mainly due to the black pigment decreasing, thus confirming the
yellow-y gene critical role in melanin pigmentation. However, as future prospects, this could be a great genetic marker for upcoming genomic research [
79].
Referring to
A. mellifera sex determination, it is controlled by the heterozygosity at a particular locus that harbors the key
complementary sex determiner (
csd) gene. The bees that are heterozygous at this specific locus are females, while the males are homozygous or hemizygous [
80]. In a recent study, Wang et al. (2021) [
81] used the CRISPR-Cas9 tool in order to knock out the
csd gene and, thus, eliminated the genetic difference between females and males. Subsequently, they aimed to observe the transcriptome difference between the two sexes in this particular genetic background. They also successfully induced target mutations in mutant haploid individuals. It was observed that the expression level of several male-biased genes was higher in the mutant males. On the other hand, the expression level of several specific female-biased genes was lower. Their data also confirmed that
csd interacts with certain genes, such as
fruitless,
troponin T, and
transformer-2 just to mention a few [
81].