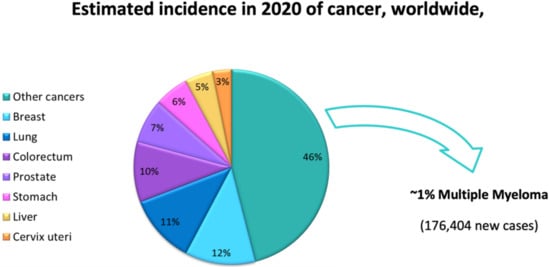
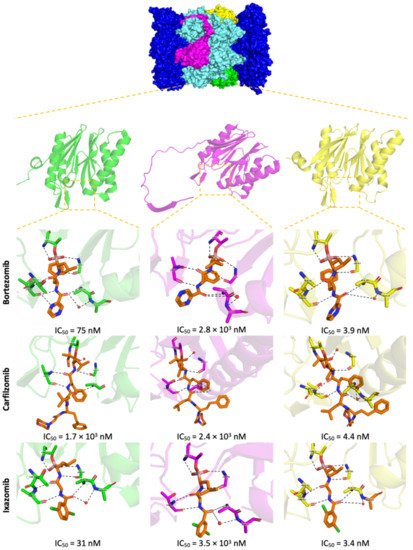
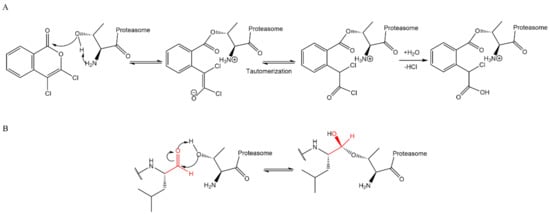
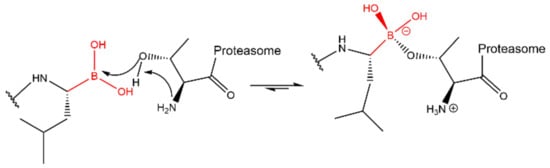
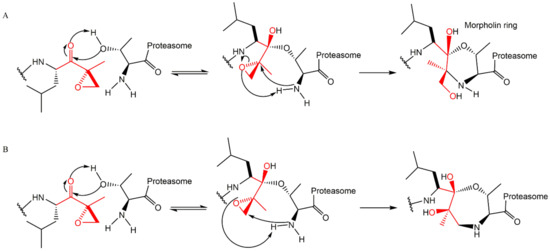
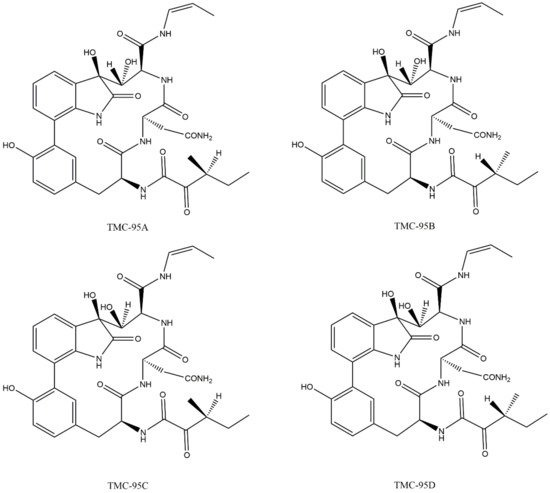
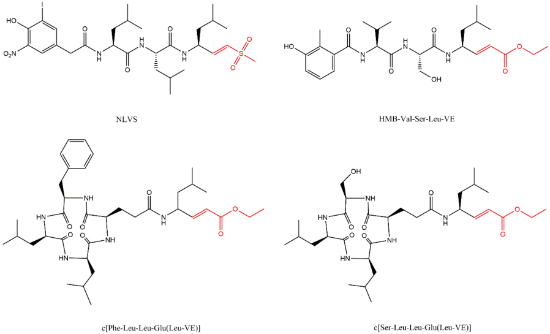
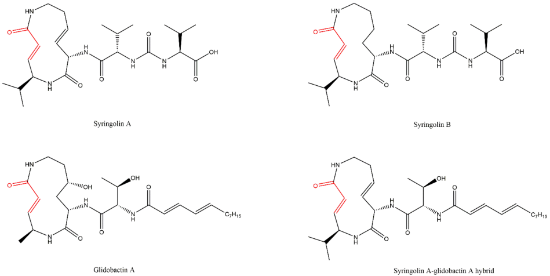
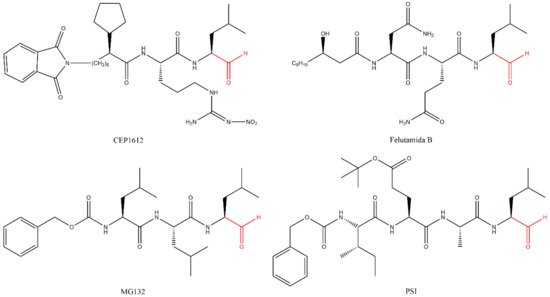
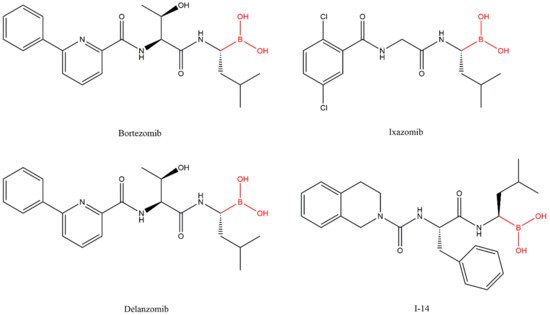
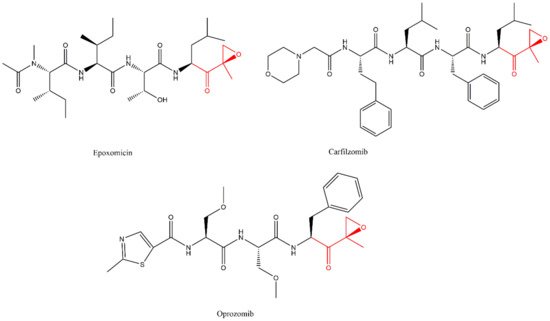
Lactacystin (a metabolite from Streptomyces gram-positive) was the first natural non-peptide-like proteasome inhibitor to be found in nature, and it bears a β-lactone moiety. In vivo, it acts as a prodrug which is hydrolyzed at neutral pH into clasto-lactacystin-β-lactone (also called omuralide) (
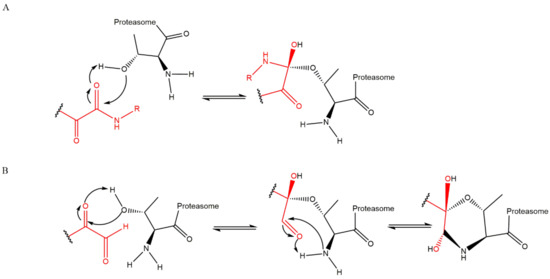
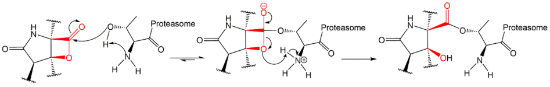
Figure 17), which can cross the plasma membranes of mammalian cells (whereas the lactacystin form cannot) and it is covalently and irreversibly bound to the β5 subunit’s Thr1, resulting in the opening of the β-lactone ring and acylation of the hydroxyl group in Thr1 (
Figure 18). Omuralide does not inhibit various serine and cysteine proteases, except for cathepsin A and cytosolic tripeptidyl peptidase II [
27,
28,
61].
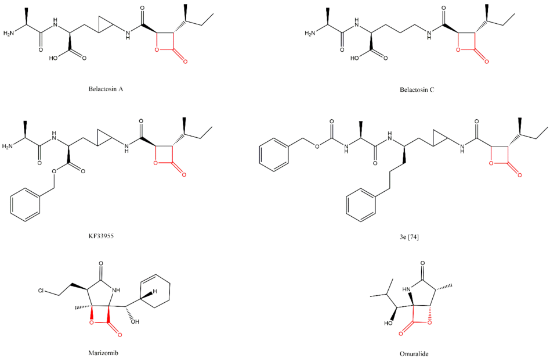
In 2000, belactosin A and C were isolated from
Streptomyces sp. by Asai et al. [
72] (
Figure 17). These compounds exhibited antitumor activity attributed to the inhibition of proteasome activity. Additionally, with the goal of increasing the potency of belactosin A, a benzyl group was introduced (KF33955,
Figure 17) [
73]. Other derivatives were synthesized in 2013 by Kawamura et al. [
74], who identified the 3e derivative (
Figure 17) as an inhibitor comparable to bortezomib (IC
50 value of 5.7 nM for the β5 subunit). Belactosin C analogs of the boronate inhibitors class were synthesized with the purpose of developing reversible inhibitors [
75]. However, the most potent boronate inhibitor developed exhibited a value of IC
50 for the β5 subunit of 20S CP, 10-fold than bortezomib’s value (IC
50 = 280 nM).
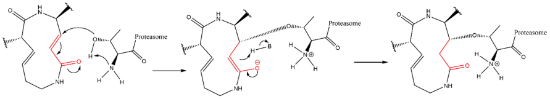
Marizomib (also named salinosporamide A or NPI-0052) (
Figure 17) is a secondary metabolite of the marine actinomycete
Salinispora tropica. This is the only non-peptidic proteasome inhibitor for which the Committee for Orphan Medicinal Products has issued a positive opinion regarding the orphan drug designation for the treatment of MM (2014) and for the treatment of glioma (2018), because marizomib crosses the blood-brain barrier [
76,
77]. According to the Triphase Accelerator Corporation, the orphan drug designation was also granted by the FDA, for the treatment of glioblastoma and MM [
78,
79,
80]. It is an irreversible proteasome inhibitor whose carbonyl group interacts with Thr1′s hydroxyl group (it inhibits the three catalytic subunits of the 20S CP quickly and for a long period of time). Although it is orally bioavailable [
36], all related (completed and ongoing) clinical studies have reported the drug administration as intravenous [
81,
82,
83,
84,
85,
86,
87,
88,
89,
90,
91]. Marizomib was first tested in a phase I clinical trial conducted in patients with advanced solid tumor malignancies or refractory lymphoma whose disease had progressed after standard treatment [
91]. Afterwards, two studies of phases I and II were conducted to evaluate the safety, pharmacokinetics and pharmacodynamics of escalating maximum tolerated and recommended doses of marizomib (and low dose dexamethasone) in patients with advanced malignancies including solid tumors, lymphomas, leukemias and MM (one of the studies only analyzed MM). These studies demonstrated that marizomib does not induce severe peripheral neuropathy or hematologic toxicity associated with bortezomib and carfilzomib, and it was verified that it is well-tolerated in heavily pretreated relapsed and/or refractory MM patients [
82,
84,
92]. A phase I clinical trial has been completed to assess marizomib in combination with the histone deacetylase inhibitor vorinostat, in patients with melanoma, non-small cell lung cancer, pancreatic cancer or lymphoma [
85]. This study demonstrated that this combination therapy is feasible and well-tolerated. Albeit confirmed responses were not reported, 61% of the evaluable patients reported a stable disease and 39% had decreases in tumor measurements (up to 25%) [
93]. In a phase I clinical trial in patients with relapsed/refractory MM, a combination of marizomib, pomalidomide and low-dose dexamethasone demonstrated that this combination is well tolerated and promising in heavily pre-treated patients, including those who were refractory to prior treatment with carfilzomib, bortezomib and/or lenalidomide, and patients with high-risk cytogenetics (17p deletion and/or 4:14 chromosome translocation) [
86,
94,
95]. The safety and preliminary efficacy of marizomib, alone or in combination with bevacizumab, were evaluated in patients with recurrent glioblastoma in a phase I/II clinical trial. This study demonstrated that marizomib is safe, as monotherapy or in combination with bevacizumab, for patients with recurrent glioblastoma. However, it did not show a benefit to patients from the addition of marizomib to bevacizumab. Marizomib was also shown to inhibit the proteolytic activity of all three subunits, with repeated dosing, at all doses assessed [
87,
96]. The detailed results from a completed phase I clinical trial to evaluate the combination of marizomib with Optune
TM, temozolomide and radiotherapy in patients with newly diagnosed WHO Grade IV malignant glioma are expected [
88,
97]. Three studies are ongoing, but not yet recruiting participants: (1) a phase III trial to evaluate marizomib in combination with standard temozolomide-based radiochemotherapy versus standard temozolomide-based radiochemotherapy alone in patients with newly diagnosed glioblastoma; (2) a phase II clinical trial to evaluate nanoparticle albumin-bound rapamycin as a single agent or combined with standard therapies (including marizomib) in bevacizumab-naïve subjects with progressive high grade glioma following prior therapy and subjects with newly diagnosed glioblastoma; and (3) a phase I study to evaluate the safety, tolerability, pharmacokinetic parameters and preliminary efficacy of the drugs marizomib and panobinostat in pediatric patients with diffuse intrinsic pontine glioma [
81,
83,
89]. A phase II study to evaluate the efficacy of treatment with marizomib for recurrent low-grade and anaplastic supratentorial, infratentorial was terminated because the pharmaceutical company leading the study closed their program evaluating marizomib [
90]. A phase II study will be carried out to evaluate the combination of marizomib, more pomalidomide and dexamethasone in patients with relapsed/refractory MM patients and patients with central nervous system involvement [
98].