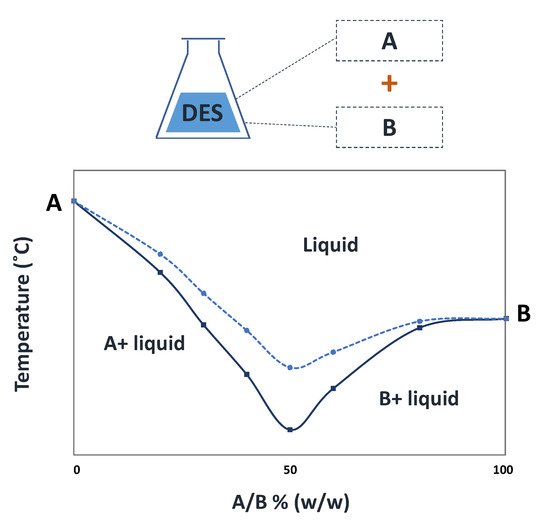
Deep eutectic solvents (DES) are eutectic mixtures that present a deviation from the ideal thermodynamic solid–liquid phase behavior, where a significant depression in the melting temperature occurs. If properly designed and chosen, DES may be liquid at room and the human body’s temperatures and display a biocompatible character, thus representing relevant options in the pharmaceutical field. Accordingly, DES have been studied as alternative solvents or in formulations of pharmaceuticals to improve their solubility and stability. Depending on the DES components, these mixtures might exhibit interesting biological activities compatible with several applications. The use of DES as functional agents or as novel liquid forms of active pharmaceutical ingredients (API-DES) with the goal of improving bioavailability, permeability and therapeutic efficacy of a given API stands as alternative strategies in the pharmaceutical field for drug delivery purposes.
- deep eutectic solvents
- active pharmaceutical ingredients
- solubility
- bioavailability
- therapeutic efficacy
| DES | Molar Ratio | API Solubilized | Ref |
|---|---|---|---|
| [Ch]Cl–urea | 1:2 | Piroxicam | [13] |
| Betaine–urea | 1:2 | Clavulanic acid | [14] |
| [Ch]Cl–lactic acid–water | 1:0.9:0.6 | Aprepitant | [13] |
| [Ch]Cl–1,2-propanediol | 1:2 | Aspirin | [15] |
| [Ch]Cl–ethyleneglycol | 1:2 | Naproxen | [16] |
| Betaine–glycerol–water | 1:2:1 | Indomethacin | [13] |
| [Ch]Cl–malic acid | 1:1 | Celecoxib | [16] |
| Camphor–menthol | 1:1 | Ibuprofen | [17] |
| [Ch]Cl–levulinic acid | 1:2 | Ketoprofen | [15] |
| Parent API | API–DES | Molar ratio | Ref |
| Aspirin | [Ch]Cl–aspirin | 1:2 | [18] |
| Ibuprofen | Ibuprofen–menthol | 1:3 | [19] |
| Ibuprofen | Ibuprofen–benzoic acid | 1:3 | [19] |
| Ibuprofen | Ibuprofen–phenylacetic acid | 1:2 | [19] |
| Ibuprofen | Ibuprofen–limonene | 1:8 | [20] |
| Ethambutol | Citric Acid–Ethambutol–water | 2:1:10 | [21] |
References
- Shekunov, B.Y.; York, P. Crystallization process in pharmaceutical technology and drug delivery design. J. Cryst. Growth 2000, 211, 122–136.
- Singhal, D.; Curatolo, W. Drug polymorphism and dosage form design: A practical perspective. Adv. Drug Deliv. Rev. 2004, 56, 335–347.
- Savjani, K.T.; Gajjar, A.K.; Savjani, J.K. Drug Solubility: Importance and Enhancement Techniques. ISRN Pharm. 2012, 2012, 195727.
- Sekiguchi, K.; Obi, N. Studies on Absorption of Eutectic Mixture. I. A Comparison of the Behavior of Eutectic Mixture of Sulfathiazole and that of Ordinary Sulfathiazole in Man. Chem. Pharm. Bull. 1961, 9, 866–872.
- El-Banna, H.M. Solid dispersion of pharmaceutical ternary systems I: Phase diagram of aspirin-acetaminophen-urea system. J. Pharm. Sci. 1978, 67, 1109–1111.
- Goud, N.R.; Suresh, K.; Sanphui, P.; Nangia, A. Fast dissolving eutectic compositions of curcumin. Int. J. Pharm. 2012, 439, 63–72.
- Abbott, A.P.; Capper, G.; Davies, D.L.; Rasheed, R.K.; Tambyrajah, V. Novel solvent properties of choline chloride/urea mixtures. Chem. Commun. 2003, 9, 70–71.
- Francisco, M.; Van Den Bruinhorst, A.; Kroon, M.C. Low-transition-temperature mixtures (LTTMs): A new generation of designer solvents. Angew. Chemie-Int. Ed. 2013, 52, 3074–3085.
- Mbous, Y.P.; Hayyan, M.; Hayyan, A.; Wong, W.F.; Hashim, M.A.; Looi, C.Y. Applications of deep eutectic solvents in biotechnology and bioengineering—Promises and challenges. Biotechnol. Adv. 2017, 35, 105–134.
- Martins, M.A.R.; Pinho, S.P.; Coutinho, J.A.P. Insights into the Nature of Eutectic and Deep Eutectic Mixtures. J. Solution Chem. 2019, 48, 962–982.
- Perna, F.M.; Vitale, P.; Capriati, V. Deep eutectic solvents and their applications as green solvents. Curr. Opin. Green Sustain. Chem. 2020, 21, 27–33.
- Smith, E.L.; Abbott, A.P.; Ryder, K.S. Deep Eutectic Solvents (DESs) and Their Applications. Chem. Rev. 2014, 114, 11060–11082.
- Palmelund, H.; Andersson, M.P.; Asgreen, C.J.; Boyd, B.J.; Rantanen, J. Tailor-made solvents for pharmaceutical use? Experimental and computational approach for determining solubility in deep eutectic solvents. Int. J. Pharm. X 2019, 1, 100034.
- Olivares, B.; Martínez, F.; Rivas, L.; Calde, C.; Munita, J.M.; Campodonico, P.R. A Natural Deep Eutectic Solvent Formulated to Stabilize β -Lactam Antibiotics. Sci. Rep. 2018, 8, 14900.
- Lu, C.; Cao, J.; Wang, N.; Su, E. Significantly improving the solubility of non-steroidal anti-inflammatory drugs in deep eutectic solvents for potential non-aqueous liquid administration. R. Soc. Chem. 2013, 7, 955–959.
- Mokhtarpour, M.; Shekaari, H.; Zafarani-moattar, M.T.; Golgoun, S. Solubility and solvation behavior of some drugs in choline based deep eutectic solvents at different temperatures. J. Mol. Liq. 2019, 297, 111799.
- Phaechamud, T.; Tuntarawongsa, S.; Charoensuksai, P. Evaporation Behavior and Characterization of Eutectic Solvent and Ibuprofen Eutectic Solution. AAPS PharmSciTech 2016, 17, 1213–1220.
- Abbott, A.P.; Ahmed, E.I.; Prasad, K.; Qader, I.B.; Ryder, K.S. Liquid pharmaceuticals formulation by eutectic formation. Fluid Phase Equilib. 2017, 448, 2–8.
- Duarte, A.R.C.; Ferreira, A.S.D.; Barreiros, S.; Cabrita, E.; Reis, R.L.; Paiva, A. A comparison between pure active pharmaceutical ingredients and therapeutic deep eutectic solvents: Solubility and permeability studies. Eur. J. Pharm. Biopharm. 2017, 114, 296–304.
- Pereira, C.V.; Silva, J.M.; Rodrigues, L.; Reis, R.L.; Paiva, A.; Duarte, A.R.C.; Matias, A. Unveil the Anticancer Potential of Limomene Based Therapeutic Deep Eutectic Solvents. Sci. Rep. 2019, 9, 14926.
- Santos, F.; Leitão, M.I.P.S.; Duarte, A.R.C. Properties of therapeutic deep eutectic solvents of L-arginine and ethambutol for tuberculosis treatment. Molecules 2019, 24, 55.
- Aroso, I.M.; Craveiro, R.; Rocha, Â.; Dionísio, M.; Barreiros, S.; Reis, R.L.; Paiva, A.; Duarte, A.R.C. Design of controlled release systems for THEDES—Therapeutic deep eutectic solvents, using supercritical fluid technology. Int. J. Pharm. 2015, 492, 73–79.
- Byrne, F.P.; Jin, S.; Paggiola, G.; Petchey, T.H.M.; Clark, J.H.; Farmer, T.J.; Hunt, A.J.; Robert McElroy, C.; Sherwood, J. Tools and techniques for solvent selection: Green solvent selection guides. Sustain. Chem. Process. 2016, 4, 7.
- Adawiyah, N.; Moniruzzaman, M.; Hawatulaila, S.; Goto, M. Ionic liquids as a potential tool for drug delivery systems. Med. Chem. Commun. 2016, 7, 1881–1897.
- Gad, S.C. Pharmaceutical Manufacturing Handbook: Production and Processes; John Wiley Sons, Inc.: Hoboken, NJ, USA, 2008; pp. 314–316.
- Mota-Morales, J.D.; Gutiérrez, M.C.; Ferrer, M.L.; Sanchez, I.C.; Elizalde-Peña, E.A.; Pojman, J.A.; Monte, F.D.; Luna-Bárcenas, G. Deep eutectic solvents as both active fillers and monomers for frontal polymerization. J. Polym. Sci. Part A Polym. Chem. 2013, 51, 1767–1773.
- Lynam, J.G.; Kumar, N.; Wong, M.J. Deep eutectic solvents’ ability to solubilize lignin, cellulose, and hemicellulose; thermal stability; and density. Bioresour. Technol. 2017, 238, 684–689.
- Almeida, C.M.R.; Magalhães, J.M.C.S.; Souza, H.K.S.; Gonçalves, M.P. The role of choline chloride-based deep eutectic solvent and curcumin on chitosan films properties. Food Hydrocoll. 2018, 81, 456–466.
- Pedro, S.N.; Freire, M.G.; Freire, C.S.R.; Silvestre, A.J.D.; Pedro, S.N.; Freire, M.G.; Freire, C.S.R.; Silvestre, A.J.D. Deep eutectic solvents comprising active pharmaceutical ingredients in the development of drug delivery systems development of drug delivery systems. Expert Opin. Drug Deliv. 2019, 16, 497–506.
- Emami, S.; Shayanfar, A. Deep eutectic solvents for pharmaceutical formulation and drug delivery applications. Pharm. Dev. Technol. 2020, 25, 779–796.
- Zainal-Abidin, M.H.; Hayyan, M.; Ngoh, G.C.; Wong, W.F.; Looi, C.Y. Emerging frontiers of deep eutectic solvents in drug discovery and drug delivery systems. J. Control. Release 2019, 316, 168–195.
- Nguyen, C.H.; Augis, L.; Fourmentin, S.; Barratt, G.; Legrand, F.X. Deep Eutectic Solvents for Innovative Pharmaceutical Formulations. In Deep Eutectic Solvents for Medicine, Gas Solubilization and Extraction of Natural Substances; Fourmentin, S., Costa Gomes, M., Lichtfouse, E., Eds.; Springer: Cham, Switzerland, 2021.
- Gutiérrez, A.; Atilhan, M.; Aparicio, S. A theoretical study on lidocaine solubility in deep eutectic solvents. Phys. Chem. Chem. Phys. 2018, 20, 27464–27473.
- Abranches, D.O.; Larriba, M.; Silva, L.P.; Melle-Franco, M.; Palomar, J.F.; Pinho, S.P.; Coutinho, J.A.P. Using COSMO-RS to design choline chloride pharmaceutical eutectic solvents. Fluid Phase Equilib. 2019, 497, 71–78.
