Your browser does not fully support modern features. Please upgrade for a smoother experience.
Please note this is an old version of this entry, which may differ significantly from the current revision.
Subjects:
Health Care Sciences & Services
Colistin (polymyxin E) has been used as a growth promoter in food-production animals, but it is also used in pets for the prevention and treatment of bacterial infections.
- colistin resistance
- mcr genes
- dogs
- cats
- pets
1. Introduction
As previously reported, the overuse of colistin has been shown to cause colistin resistance in bacteria colonizing the intestinal gut of animals [1]. However, only a few countries have prohibited the long-term use of colistin. Colistin reuse has resurfaced due to necessity and even more limited treatment options, particularly since the spread of multidrug-resistant bacteria (MDR) and carbapenem-resistant bacteria [2]. Colistin-resistant bacteria have become a significant public health issue. Indeed, colistin is a last-resort antibiotic; its failure to treat patients necessitates the development of new and more effective antibiotic therapies [3]. Since the discovery of the first plasmid harboring mcr-1 in China from pigs, the microbiota of animals appears as a source of colistin-resistant bacteria [4]. A variety of colistin-resistance gene variants have been discovered, ranging from mcr-2 to mcr-10 [5][6][7][8][9][10][11][12][13]. In 2016, mcr-1 was detected in several samples from humans and from food-producing animals on all seven continents and in more than 30 countries [14]. It is crucial to have a better understanding of the microorganisms found in pets, as well as the risk of pathogen transmission and antibiotic resistance genes such as mcr-1. mcr-1-producing bacteria have been reported in zoonotic transmission from pets to humans who adopt them for protection, entertainment, or companionship [15]. Close contact between cats, dogs, and their owners led to the occurrence and transmission of antibiotic-resistant microorganisms [16].
In Asia and the United States, mcr-1 in Enterobacteriaceae has already been reported in dogs and cats. The mcr genes in pets were also detected in Beijing, China, between 2012 and 2016 [17]. Further studies reported that dog feces in a city park in Quito (Ecuador) have a high prevalence of multidrug-resistant bacteria (MDR) [18]. Between 2017 and 2019, researchers in China reported an abundance of MDR such as Klebsiella pneumoniae harboring colistin resistance genes (mcr-1, mcr-8) and β-lactamases (blaOXA-181, blaNDM-5) in cats, which was significantly higher than in dogs [19]. In Europe, almost all colistin resistance studies have been focused in food-producing animals such as pigs and poultry [20][21][22]. Regarding Asia, for the first time, the mcr-1 gene was detected in dogs from South Korea, with an average nucleotide identity analysis similar to those found in Korean patients [23]. However, diseased dogs from Taiwan have Klebsiella spp and Enterobacter spp carrying mcr-1 gene [24].
Interestingly, colistin-resistant strains in pets have never been investigated in France, according to the literature. Colistin is currently not recommended for treating animal infections caused by Enterobacteriaceae in France unless it is necessary. Colistin is a critically important antibiotic that is used as a last resort treatment. Furthermore, because of unfavorable and toxic effects of colistin, its restriction has been requested by some agricultural or veterinary organizations and also by the European commission in 2016 [1]. Despite these precautions, the emergence of colistin resistance in animals is becoming more common, as evidenced by various studies on the subject. As reported, colistin resistance in animals is likely mediated by environmental factors and animal nutrition [25].
Colistin resistance in pets is rarely studied because the majority of reported studies were focused on food animals. However, pets are in daily close contact with humans. Determining the prevalence of colistin-resistant bacteria in pets is critical in order to identify any potential risk factors for colistin resistance transmission, particularly zoonotic transmission of bacteria [26].
2. Screening of Colistin-Resistant Bacteria in Pets
Among 157 collected fecal samples from domestic animals (dogs and cats), a total of 218 bacterial isolates were obtained from selective medium agar LBJMR (Vancomycin and colistin). As already reported, the gastrointestinal tract of animals can harbor a highly complex mixture of organisms that help maintain intestinal flora and overall health [27]. Indeed, the study of fecal samples depends upon the natural composition of the microorganisms and is specific to each animal. According to the literature, most studies have been performed on the general microorganism composition and most data have been derived from the analysis of feces from healthy laboratory animals [28][29]. In their study, the composition of microorganisms in the feces samples was described in terms of naturally colistin-resistant bacteria and those with acquired colistin resistance via mcr genes. Identification of colistin-resistant bacteria in cats revealed 150 bacteria, as represented in Figure 1. Identification encompassed 23 different bacterial species. Eighty percent (n = 120) of all identified bacteria were Gram-positive and 20% (n = 30) were Gram-negative. The dominant strains were naturally colistin-resistant for all GPB: Bacillus cereus, B. vallismortis, Enterococcus durans, E. faecalis, E. faecium, E. hirae, Leuconostoc citreum, L. mesenteroides, and Pediococcus pentosacens. A high proportion of Lactobacillus species were identified: L. curvatus, L. brevis, L. fructivorans, L. murinus, L. paracasei, L. planetarium, L. reuteri, L. sakei, and L. salivarius. In contrast, the GNB that were isolated were believed to have acquired resistance to colistin via mcr genes or other mechanisms. Here, 13% (n = 4) of colistin-resistant GNB were screened as being mcr carriers: E. coli (n = 3) and Rahnella aquatilis (n = 1). In addition, 87% (n = 26) of GNB strains were naturally resistant to colistin: Sphingomonas paucimobilis, Providencia heimbachae, and Hafnia alvei (Figure 1).
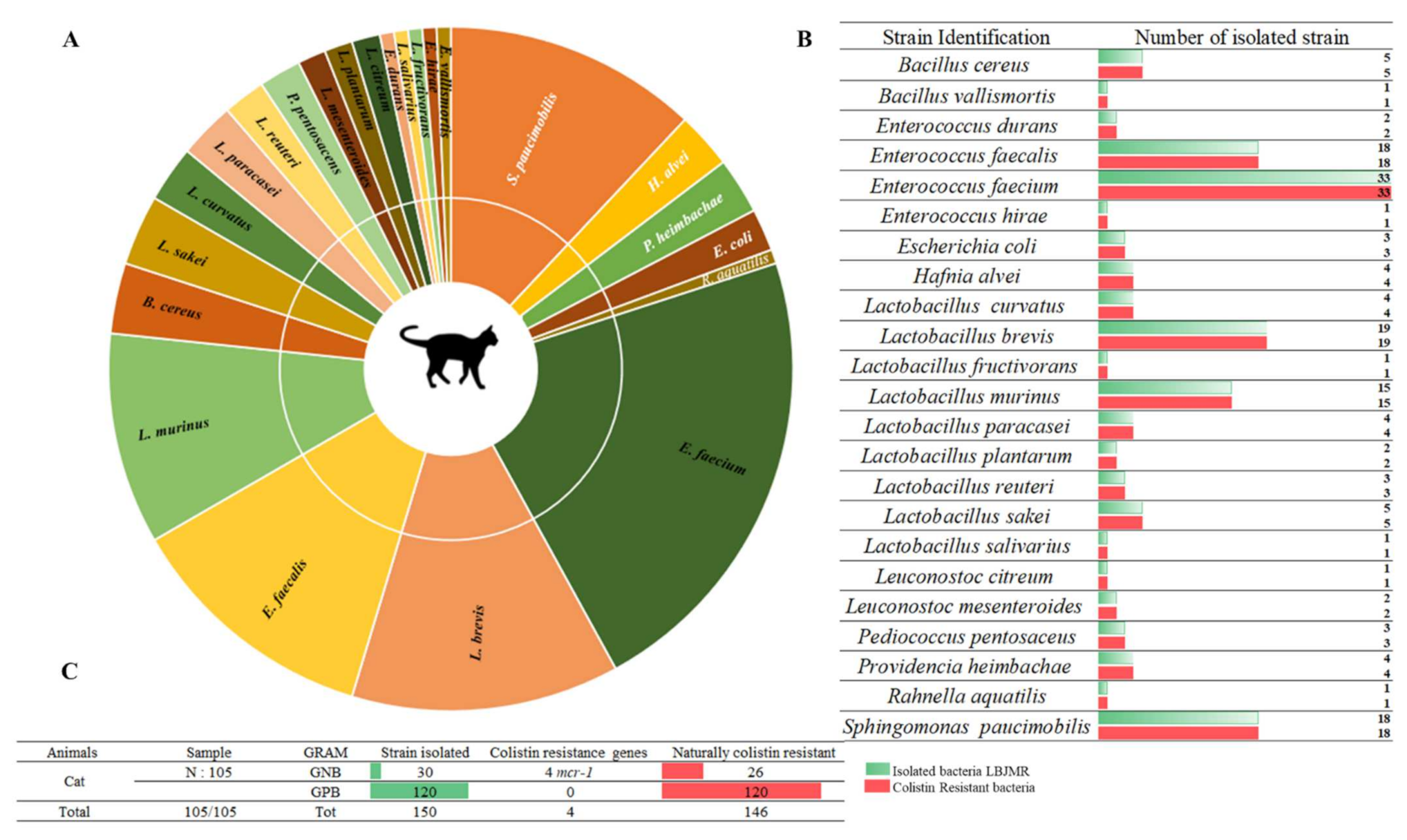
Figure 1. Proportion of colistin-resistant bacteria in feces of cats. (A). Pie chart representing the distribution of strains isolated in LBJMR. (B). Colistin-resistant bacteria verified by AST testing. (C). Statistical data on isolated bacteria according to Gram status and resistance mechanism.
The selective bacterial culture of dogs’ feces allowed us to isolate a total of 68 bacterial strains, divided into 14 different species. Fifty percent (n = 34) of the isolated strains were Gram-negative and 50% (n = 34) were Gram-positive. The isolated GPB in dogs’ feces were: L. brevis, L. coryniformis, L. murinus, L. paracasei, L. planetarium, L. reuteri, L. saerimmeri, L. sakei and P. pentosacens. The Gram-negative strains corresponded to E. coli, Moellerella wisconsensis and Brevundimonas diminuta. Sixty-five percent (n = 22) of the GNB were naturally resistant to colistin: H. alvei and Morganella morganii (Figure 2). Colistin-resistant bacteria differ widely from one animal to another. As mentioned previously, bacteria in fecal samples from dogs exhibited broad variability [30]. Generally, colistin is used orally, with low bioavailability, so the gastrointestinal microorganisms are directly exposed to colistin [31]. In 2016, the polymyxin family was the fifth best-selling antibiotic in Europe, and among polymyxins, colistin accounted for more than 99.9% of sales [32]. This uncontrollable use accelerated the dissemination of colistin resistance in animals and, subsequently, human beings [33]. Consequently, in 2016 the European Medicines Agency (EMA) updated its guidelines to minimize the use of colistin in animals to reduce its impact on human health [32][34]. The restricted colistin use in cats and dogs in France has not prevented the abundance of colistin-resistant bacteria in their microbiome composition. These findings highlight a topic that requires more research on the sources of colistin resistance.
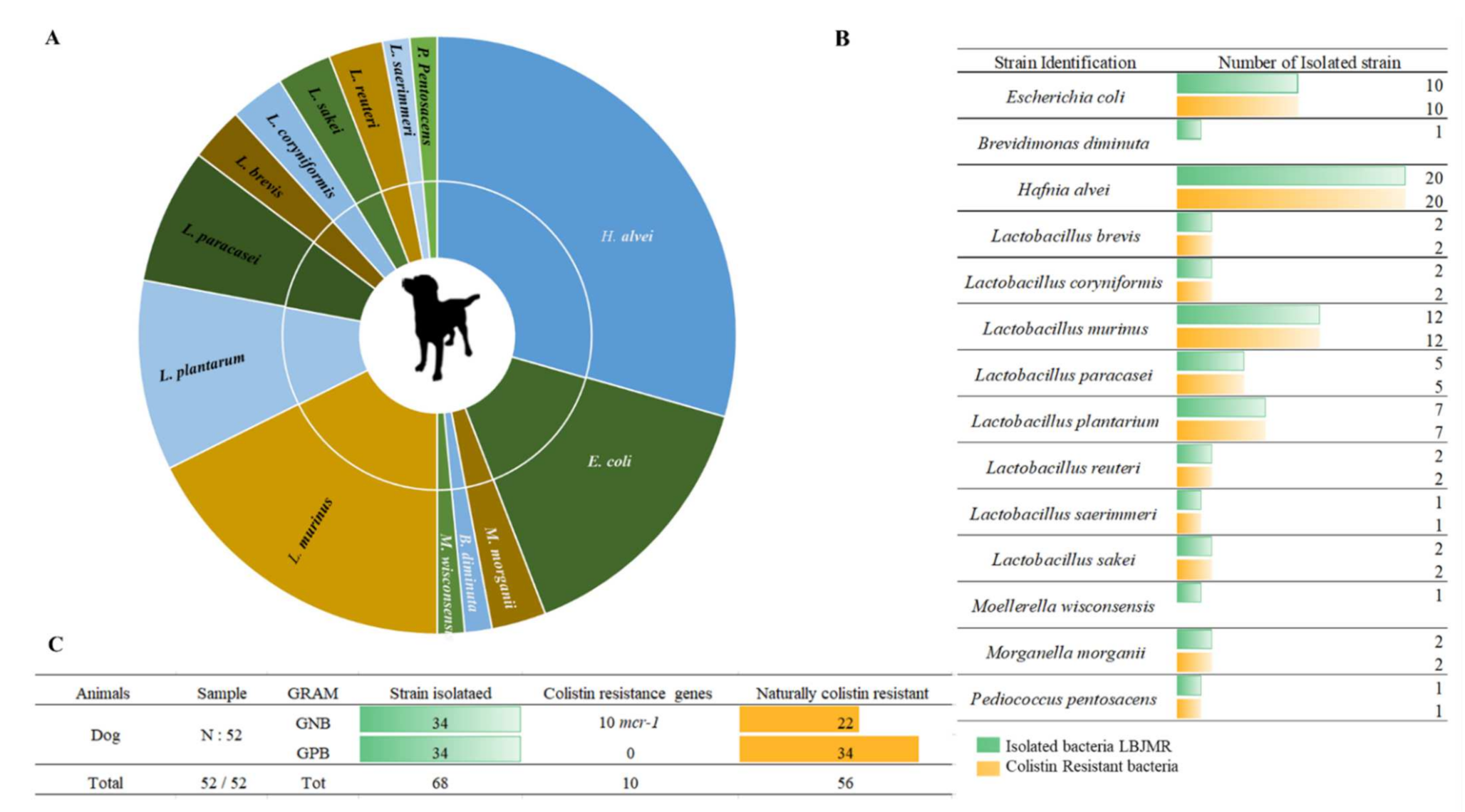
Figure 2. Illustration of colistin-resistant bacteria in the feces of dogs. (A). Distributed representation of colistin-resistant bacteria in a pie chart. (B). Distinguished colistin-resistant bacteria isolated in LBJMR. (C). Statistical analysis according to Gram status and colistin resistance.
Cats and dogs, on the other hand, share certain bacterial species; however, as shown in Figure 3, colistin-resistant bacteria in cat and dog feces are variable. In cats, researchers isolated a variety of species of the genera: Lactobacillus, Bacillus, and Enterococcus, whereas in dogs Lactobacillus was the most abundant genus, with seven different species. Moreover, GNB isolated in cats and dogs are different and do not belong to the same bacterial genera. In addition, cats and dogs also share certain bacterial species. The bacterial cross-link between cats and dogs corresponds to the following bacteria: GNB (E. coli, H. alvei, P. heimbachae and S. paracaseimobilis); and GPB (L. murinus, L. plantarum, L. sakei, L. reuteri, L. paracasei, L. brevis and P. pentosaceus). In colistin-resistant bacteria, natural colistin resistance is stronger than acquired resistance. It is interesting to note that both humans and animals can have a cross-link of identical bacterial species in their stools, which could lead to bacterial exchanges between the two species [35].
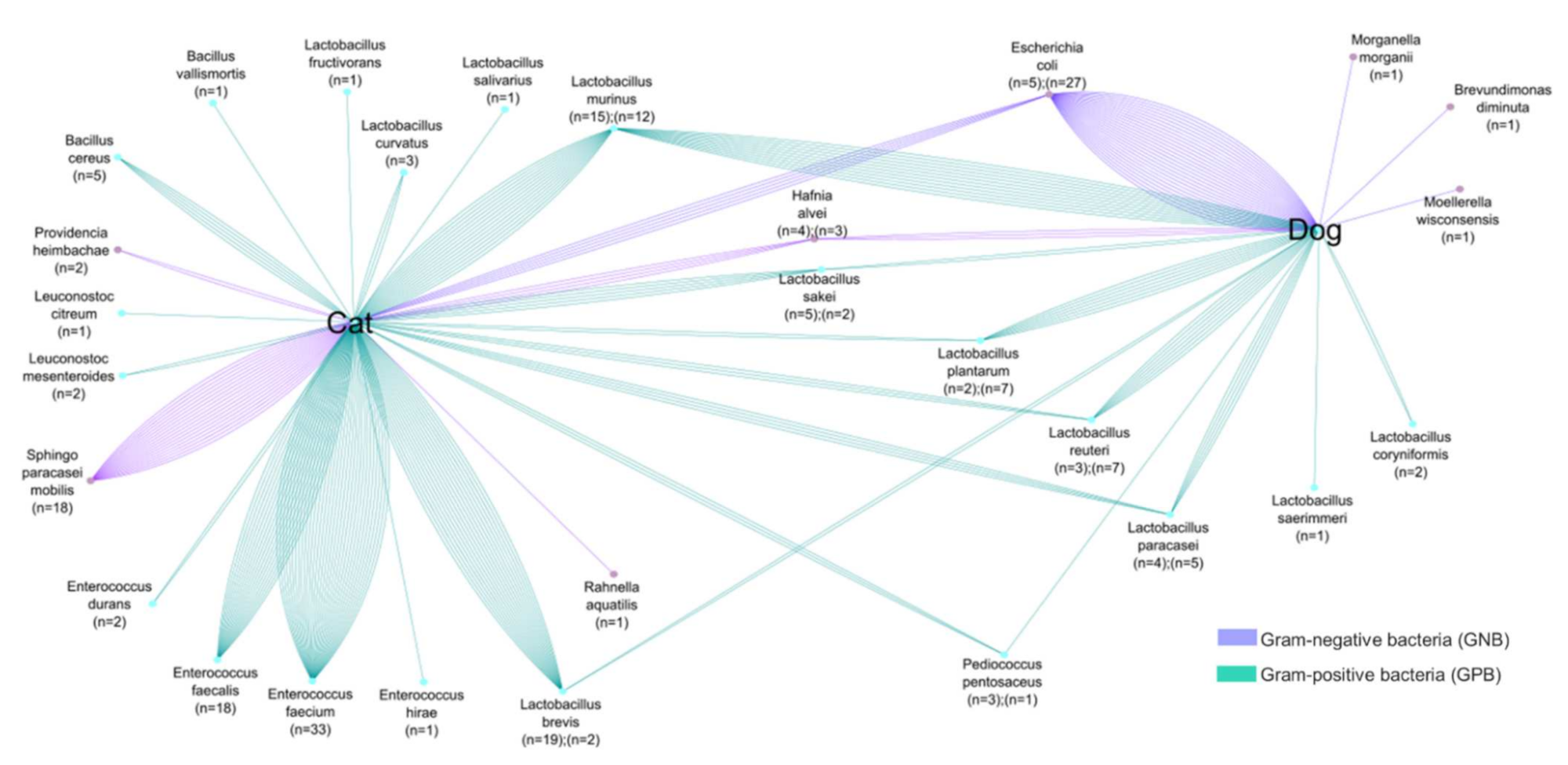
Figure 3. Comparative analyses of common colistin-resistant bacterial species found in the fecal samples of dogs and cats. The number of strains is proportional to the number of lines.
This entry is adapted from the peer-reviewed paper 10.3390/ani12050633
References
- Kempf, I.; Jouy, E.; Chauvin, C. Colistin use and colistin resistance in bacteria from animals. Int. J. Antimicrob. Agents 2016, 48, 598–606.
- Andrasevic, A.T.; Antunovic, I.A. The world-wide spread of carbapenem-resistant Enterobacterales. One Health Risk Manag. 2020, 2, 4–12.
- Kontopidou, F.; Plachouras, D.; Papadomichelakis, E.; Koukos, G.; Galani, I.; Poulakou, G.; Dimopoulos, G.; Antoniadou, A.; Armaganidis, A.; Giamarellou, H. Colonization and infection by colistin-resistant Gram-negative bacteria in a cohort of critically ill patients. Clin. Microbiol. Infect. 2011, 17, E9–E11.
- Liu, Y.-Y.; Wang, Y.; Walsh, T.R.; Yi, L.-X.; Zhang, R.; Spencer, J.; Doi, Y.; Tian, G.; Dong, B.; Huang, X.; et al. Emergence of plasmid-mediated colistin resistance mechanism MCR-1 in animals and human beings in China: A microbiological and molecular biological study. Lancet Infect. Dis. 2016, 16, 161–168.
- Xavier, B.B.; Lammens, C.; Ruhal, R.; Kumar-Singh, S.; Butaye, P.; Goossens, H.; Malhotra-Kumar, S. Identification of a novel plasmid-mediated colistin-resistance gene, mcr-2, in Escherichia coli, Belgium, June 2016. Euro Surveill. Bull. Eur. Mal. Transm. Eurosurveillance 2016, 21, 30280.
- Yin, W.; Li, H.; Shen, Y.; Liu, Z.; Wang, S.; Shen, Z.; Zhang, R.; Walsh, T.R.; Shen, J.; Wang, Y. Novel Plasmid-Mediated Colistin Resistance Gene mcr-3 in Escherichia coli. mBio 2017, 8, e00543–e00617.
- Carattoli, A.; Villa, L.; Feudi, C.; Curcio, L.; Orsini, S.; Luppi, A.; Pezzotti, G.; Magistrali, C.F. Novel plasmid-mediated colistin resistance mcr-4 gene in Salmonella and Escherichia coli, Italy 2013, Spain and Belgium, 2015 to 2016. Eurosurveillance 2017, 22, 30589.
- Borowiak, M.; Fischer, J.; Hammerl, J.A.; Hendriksen, R.S.; Szabo, I.; Malorny, B. Identification of a novel transposon-associated phosphoethanolamine transferase gene, mcr-5, conferring colistin resistance in d-tartrate fermenting Salmonella enterica subsp. enterica serovar Paratyphi B. J. Antimicrob. Chemother. 2017, 72, 3317–3324.
- AbuOun, M.; Stubberfield, E.J.; Duggett, N.A.; Kirchner, M.; Dormer, L.; Nunez-Garcia, J.; Randall, L.P.; Lemma, F.; Crook, D.W.; Teale, C.; et al. mcr-1 and mcr-2 (mcr-6.1) variant genes identified in Moraxella species isolated from pigs in Great Britain from 2014 to 2015. J. Antimicrob. Chemother. 2017, 72, 2745–2749.
- Yang, Y.-Q.; Li, Y.-X.; Lei, C.-W.; Zhang, A.; Wang, H.-N. Novel plasmid-mediated colistin resistance gene mcr-7.1 in Klebsiella pneumoniae. J. Antimicrob. Chemother. 2018, 73, 1791–1795.
- Wang, X.; Wang, Y.; Zhou, Y.; Wang, Z.; Wang, Y.; Zhang, S.; Shen, Z. Emergence of Colistin Resistance Gene mcr-8 and Its Variant in Raoultella ornithinolytica. Front. Microbiol. 2019, 10, 228.
- Carroll, L.M.; Gaballa, A.; Guldimann, C.; Sullivan, G.; Henderson, L.O.; Wiedmann, M. Identification of Novel Mobilized Colistin Resistance Gene mcr-9 in a Multidrug-Resistant, Colistin-Susceptible Salmonella enterica Serotype Typhimurium Isolate. mBio 2019, 10, e00853–e00919.
- Wang, C.; Feng, Y.; Liu, L.; Wei, L.; Kang, M.; Zong, Z. Identification of novel mobile colistin resistance gene mcr-10. Emerg. Microbes Infect. 2020, 9, 508–516.
- Schwarz, S.; Johnson, A.P. Transferable resistance to colistin: A new but old threat. J. Antimicrob. Chemother. 2016, 71, 2066–2070.
- Zhang, X.-F.; Doi, Y.; Huang, X.; Li, H.-Y.; Zhong, L.-L.; Zeng, K.-J.; Zhang, Y.-F.; Patil, S.; Tian, G.-B. Possible Transmission ofmcr-1–HarboringEscherichia colibetween Companion Animals and Human. Emerg. Infect. Dis. 2016, 22, 1679–1681.
- Dierikx, C.M.; Van Duijkeren, E.; Schoormans, A.H.W.; Van Essen-Zandbergen, A.; Veldman, K.; Kant, A.; Huijsdens, X.W.; Van Der Zwaluw, K.; Wagenaar, J.A.; Mevius, D.J. Occurrence and characteristics of extended-spectrum-β-lactamase- and AmpC-producing clinical isolates derived from companion animals and horses. J. Antimicrob. Chemother. 2012, 67, 1368–1374.
- Lei, L.; Wang, Y.; Schwarz, S.; Walsh, T.R.; Ou, Y.; Wu, Y.; Li, M.; Shen, Z. mcr-1 in Enterobacteriaceae from Companion Animals, Beijing, China, 2012–2016. Emerg. Infect. Dis. 2017, 23, 710–711.
- Ortega-Paredes, D.; Haro, M.; Leoro-Garzón, P.; Barba, P.; Loaiza, K.; Mora, F.; Fors, M.; Vinueza-Burgos, C.; Fernández-Moreira, E. Multidrug-resistant Escherichia coli isolated from canine faeces in a public park in Quito, Ecuador. J. Glob. Antimicrob. Resist. 2019, 18, 263–268.
- Zhang, Z.; Lei, L.; Zhang, H.; Dai, H.; Song, Y.; Li, L.; Wang, Y.; Xia, Z. Molecular Investigation of Klebsiella pneumoniae from Clinical Companion Animals in Beijing, China, 2017–2019. Pathogens 2021, 10, 271.
- De Jong, A.; Thomas, V.; Simjee, S.; Godinho, K.; Schiessl, B.; Klein, U.; Butty, P.; Vallé, M.; Marion, H.; Shryock, T.R. Pan-European monitoring of susceptibility to human-use antimicrobial agents in enteric bacteria isolated from healthy food-producing animals. J. Antimicrob. Chemother. 2011, 67, 638–651.
- Delannoy, S.; Le Devendec, L.; Jouy, E.; Fach, P.; Drider, D.; Kempf, I. Characterization of Colistin-Resistant Escherichia coli Isolated from Diseased Pigs in France. Front. Microbiol. 2017, 8, 2278.
- Apostolakos, I.; Piccirillo, A. A review on the current situation and challenges of colistin resistance in poultry production. Avian Pathol. 2018, 47, 546–558.
- Moon, D.C.; Mechesso, A.F.; Kang, H.Y.; Kim, S.-J.; Choi, J.-H.; Kim, M.H.; Song, H.-J.; Yoon, S.-S.; Lim, S.-K. First Report of an Escherichia coli Strain Carrying the Colistin Resistance Determinant mcr-1 from a Dog in South Korea. Antibiotics 2020, 9, 768.
- Chang, M.-H.; Chen, G.-J.; Lo, D.-Y. Chromosomal Locations of mcr-1 IN Klebsiella pneumoniae AND Enterobacter cloacae from dogs. Taiwan Veter. J. 2019, 45, 79–84.
- Hussein, N.H.; Al-Kadmy, I.M.S.; Taha, B.M.; Hussein, J.D. Mobilized colistin resistance (mcr) genes from 1 to 10: A comprehensive review. Mol. Biol. Rep. 2021, 48, 2897–2907.
- Bhat, A.H. Bacterial zoonoses transmitted by household pets and as reservoirs of antimicrobial resistant bacteria. Microb. Pathog. 2021, 155, 104891.
- Lee, J.E.; Lee, S.; Sung, J.; Ko, G. Analysis of human and animal fecal microbiota for microbial source tracking. ISME J. 2010, 5, 362–365.
- Deng, P.; Swanson, K. Gut microbiota of humans, dogs and cats: Current knowledge and future opportunities and challenges. Br. J. Nutr. 2014, 113, S6–S17.
- Suchodolski, J.S. Intestinal Microbiota of Dogs and Cats: A Bigger World than We Thought. Veter. Clin. N. Am. Small Anim. Pract. 2011, 41, 261–272.
- Garcia-Mazcorro, J.F.; Dowd, S.E.; Poulsen, J.; Steiner, J.M.; Suchodolski, J.S. Abundance and short-term temporal variability of fecal microbiota in healthy dogs. MicrobiologyOpen 2012, 1, 340–347.
- Viel, A. Usages de la Colistine en Médecine Humaine et Vétérinaire: Exploration Pharmacocinétique et Problématique D’Antibiorésistance. Ph.D. Thesis, Université de Poitiers, Poitiers, France, 2017.
- European Medicines Agency. Updated advice on the use of colistin products in animals within the European Union: Development of resistance and possible impact on human and animal health. Eur. Med. Agency 2016, 44, 56.
- Walsh, T.R.; Wu, Y. China bans colistin as a feed additive for animals. Lancet Infect. Dis. 2016, 16, 1102.
- Catry, B.; Cavaleri, M.; Baptiste, K.; Grave, K.; Grein, K.; Holm, A.; Jukes, H.; Liebana, E.; Navas, A.L.; Mackay, D.; et al. Use of colistin-containing products within the European Union and European Economic Area (EU/EEA): Development of resistance in animals and possible impact on human and animal health. Int. J. Antimicrob. Agents 2015, 46, 297–306.
- Furet, J.-P.; Firmesse, O.; Gourmelon, M.; Bridonneau, C.; Tap, J.; Mondot, S.; Dorã, J.; Corthier, G. Comparative assessment of human and farm animal faecal microbiota using real-time quantitative PCR. FEMS Microbiol. Ecol. 2009, 68, 351–362.
This entry is offline, you can click here to edit this entry!
