1. Water Filtration
Numerous technologies for water treatment have been developed, including physical, chemical, and biological treatments. Coagulation/flocculation, ion exchange, and chlorination/fluorination are all conventional methods that have a high operating cost and a low removal efficiency. These disadvantages can be overcome by pressure-driven membrane technologies that are capable of removing contaminants as small as 5 nm in size with a typical filtration efficiency of greater than 95% [
73].
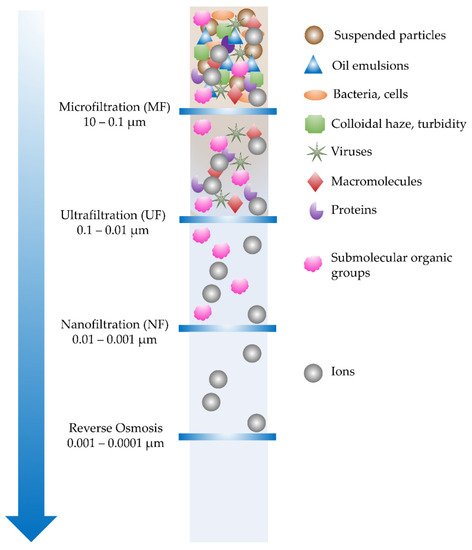
Figure 12 illustrates the four major types of pressure-driven membrane filtrations, including micro-, ultra-, and nanofiltration (MF, UF, NF), and reverse osmosis (RO) [
18].
Figure 12. Type of membrane filtration: microfiltration (MF), ultrafiltration (UF), nanofiltration (NF), and reverse osmosis (RO).
Recently, forward osmosis (FO) has been introduced to address the high energy consumption of pressure-driven membrane filtration systems. The FO membrane has a very small pore size of approximately 0.4–1.0 nm, which enables the removal of micro-pollutants and a high solute rejection rate. In FO, filtration occurs as a result of an osmotic pressure difference between the different sides of a semi-permeable membrane, which regulates the transport of fluid from a low concentration region to a high concentration region across the membrane. FO is particularly attractive because it does not require external pressure during the separation process and has a low fouling tendency [
77].
A filter membrane with good selective permeability is the core component in membrane separation technology to separate the mixture solutes using the driving force derived from external energy or chemical potential difference [
18]. Current material science breakthroughs provide a diverse range of raw materials for membrane development. Polymeric materials dominate the current membrane materials used in water and wastewater filtration. However, the primary disadvantages of most polymeric membranes are severe fouling caused by their hydrophobic nature, which has a detrimental effect on separation efficiency, flux, and membrane lifetime. Inorganic membranes, on the other hand, have a limited application due to their brittleness and high operating costs [
73]. In this regard, nanocellulose materials are particularly promising for water filtration technologies due to their abundance, renewability, and biodegradability, as well as their exquisite properties of high water permeability, increased surface area, and superior mechanical properties.
The filtration efficiency of nanocellulose-based membranes is primarily governed by water permeation flux and the removal capacity of the specific pollutant. Membrane rejection is accomplished via two distinct mechanisms either through size exclusion or the membrane affinity to the target pollutant. The size exclusion process, as the name suggests, involves selective rejection of pollutants based on their size relative to the membrane pore size. The size-exclusion process is predominant in nanocellulose membranes, and the pore size is determined by the construction of a randomly aligned CNF or CNC network, which forms the “micro/nano-pores”. Affinity membranes, on the other hand, reject pollutants based on membrane–pollutant electrostatic interactions [
63,
78].
Varanasi et al. (2015) [
79] fabricated biodegradable and recyclable ultrafiltration membranes using suspensions of CNFs, silica nanoparticles, and polyamide-amine-epichlorohydrin (PAE). The PAE adhered the negatively charged silica nanoparticles to the CNFs and improved the membrane’s wet strength. In addition, the nanocomposite membrane showed great potential for ultrafiltration with a water flux of 80 LMH and an MWCO of 200 kDa. Membranes fabricated via this process can be re-used in conventional paper recycling. Banana peel-derived bacterial nanocellulose has been used by Sijabat et al. (2019) [
80] to develop a membrane for water filtration.
Nanocellulose can be blended with metals, minerals, lignin, plasticizers, and even other cellulosic particles and work synergistically to improve the mechanical, rheological, and barrier properties of many polymeric systems [
81,
82]. Haafiz et al. [
83] used the solution casting method to create poly(lactic acid) (PLA) bionanocomposites by incorporating the PLA into cellulose nanowhiskers (CNW) derived from oil palm-empty fruit bunches. The CNW/PLA nanocomposites demonstrated the ability to improve the properties of a polymer matrix at low filler loading and to enhance the tensile and thermal properties of bionanocomposites. Cellulose nanowhiskers have also been employed to coat alginate membranes for water purification. Mokhena et al. (2017) [
83] fabricated a filtration membrane using two-layered nanoscale alginate nanofibres reinforced with maize cellulose nanowhiskers. The nanocellulose reinforcement produced a membrane with enhanced mechanical properties, and the membrane was able to completely remove water pollutants via size exclusion. The membrane was shown to reject 80% of chromium (Cr(VI)) at a pH of 11, indicating that it is suitable for short-term wastewater treatment and residential water purification.
To fabricate the support layer of thin-film composite membranes, a metalized nanocellulose composite was developed using silver and platinum nanoparticles as additive materials. The fabricated membranes demonstrated increased water flux and selectivity [
84]. Chitosan and nanocellulose, two naturally occurring biodegradable polysaccharides, were used to create composite membranes for the removal of heavy metals (chromium ion) [
85]. The nanocellulose was extracted from sugarcane bagasse via acid hydrolysis followed by a delignification procedure. The prepared membrane performed consistently well in removing chromium ions at concentrations ranging from 87 to 29 ppm over a four-cycle period. Nanocellulose/filter paper (NC/FP) composite filtration membranes were developed by Wang et al. (2019) [
62] using a layer deposition method. The NC/FP composite membranes with a higher nanocellulose composition and aspect ratio exhibit improved rejection rates with reduced water flux.
Apart from having efficient separation properties, nanocellulose also exhibits an antifouling behavior. The addition of suitable additives to cellulose changes its permeability and antifouling properties. Vetrivel et al. (2020) [
87] customized cellulose acetate ultrafiltration membranes with copper oxide nanoparticles for efficient separation with antifouling behavior. The CuO nanoparticles were synthesized from cupric nitrate using a wet precipitation method. The cellulose acetate nanocomposite membrane with 0.5 wt% of hydrophilic CuO exhibited an enhanced PWF of 118.6 Lm
−2 h
−1 due to the improvement in porosity and water uptake. Its superiority for antifouling was demonstrated by a BSA separation rate of 95.5% and flux recovery ratio of 94.7. Kong et al. [
94] discovered an ultrafiltration membrane prepared with improved antifouling properties. The ultrafiltration membranes were produced via the phase inversion technique using hydrophilic TEMPO-oxidized cellulose nanofibrils (TOCNFs) as modifying agents. The addition of TOCNFs improved the membrane’s permeability and mechanical properties. Aguilar-Sanchez et al. (2021) [
93] reported that the coating of TOCNFs on a commercial polyethersulfone (PES) microfiltration membrane via the layer-by-layer method could enhance the membrane’s antifouling and antibacterial properties.
2. Environmental Remediation
Nanocellulose is of great interest in environmental remediation owing to its cost-effectiveness and renewable adsorption [
95]. Extensive research has been conducted as evidenced by many reviews available related to environmental remediation by nanocellulose [
96]. For instance, Tshikovhi et al., (2020) [
95] reviewed the application of nanocellulose-based composites for the removal of various organic and inorganic contaminants from wastewater. The review discussed the building blocks, structure, properties, and isolation of nanocellulose-based composites with various types of reinforcements. Qiao et al., (2021) [
14] addressed the surface modifications of nanocellulose-based adsorbents with various effective adsorption groups and polymer grafting as well as the fabrication of a hybrid composite. Recent progress in the synthesis, characterization, and possible applications of nanocellulose produced from various types of lignocellulosic biomass was reviewed by Marakana et al., (2021) [
9]. Additionally, surface modification routes such as esterification, amidation, silylation, etherification, and carbamation to create advanced nanocellulose materials for desired applications were also discussed. Reshmy et al., (2022) [
97] recently published a review on the prospect of nanocellulose as biosorbent, scaffold, and membrane for the bioremediation of heavy metals. They focused on methods to design nanocellulose biosorbents with the goal of improving the adsorption efficiency according to specific contaminants. In this section, recent research on the application of nanocellulose as an adsorbent for organic and inorganic pollutants, as a photocatalyst, and for gas separation will be reviewed.
2.1. Nanocellulose as Adsorbent
Water pollution is becoming a severe problem due to aggressive industrialization. There are two categories of water pollutants namely, organic and inorganic. Organic water pollutants include dyes, pesticides, drugs, organic solvents, among others. Inorganic water pollutants, on the other hand, include metals, fertilizers, acids, alkalis, and salts. Organic dyes and heavy metal ions such as Pb(II), Cd(II), Cu(II), Fe(III), Cr(VI), Hg(II), and Co(II) are hazardous pollutants that cannot be biodegraded [
98].
Mutar and Jasim (2021) [
105] reported the removal of disperse yellow (DY) dye from its aqueous solution by utilizing nanocellulose prepared from microcrytalline cellulose. According to the Giles classification, the adsorption isotherm of the (DY) dye was (S-type), which is well-fitted to the Freundlich equation. From the kinetic data, the adsorption process obeys the pseudo-second-order. In another application, Nematollahi et al. (2022) [
106] investigated the use of cinnamon nanocellulose synthesized via acid hydrolysis as a catalyst for the decolorization of methyl orange as an azo dye. A decolorization efficiency of 40.64% was achieved which is two times higher than that of an uncatalyzed system at room temperature. The nanocellulose also possessed structural and morphological stability. Very recently, an interesting finding on the excellent adsorption capacity (14.71 mg/g) of a nanocellulose-based membrane at extremely acidic pH (pH 1.22) was reported by Wu et al. (2022) [
110]. The membrane was prepared using a combination of chitosan and CNF via the gel-casting method. They postulated that the high stability of the membrane against a strong acidic environment is attributed to the ionic and hydrogen-bonding interactions between the chitosan and CNF.
2.2. Nanocellulose as Photocatalyst
Organic dyes are commonly used in the textile and paper industries. The process generates a large volume of toxic waste which is difficult to degrade, posing a threat to the environment and human health. Various methods have been proven to be effective for treating the organic dyes in wastewater such as non-covalent adsorption, membrane filtration, ion exchange, and chemical oxidation. Unfortunately, these strategies may lead to insufficient pollutant degradation, resulting in secondary pollution [
112]. Titanium dioxide (TiO
2) has been widely used as a semiconductor photocatalyst because of its reactivity, stability, and cost effectiveness. However, in practical photocatalytic applications, aggregation of TiO
2 nanoparticles is often inevitable, and they are also difficult to recycle. The use of nanocellulose as a supporting material has been suggested as a promising solution to overcome the aforementioned problem.
The construction of a TiO
2-based composite would be beneficial to achieve better photocatalytic performance [
18]. Voisin et al., (2021) [
113] reported the fabrication of CNC–TiO
2 hybrids with an improvement in photocatalytic properties in suspension compared to the as-prepared control anatase TiO
2 owing to a better dispersibility and colloidal stability. CNC’s interfacial properties were also preserved, enabling organic dye degradation in Pickering emulsions. Interestingly, the emulsions could be freeze-dried into versatile aerogels that degraded both aqueous and organic dyes efficiently. Liu et al. (2021) [
114] employed a similar strategy to synthesize an anatase TiO
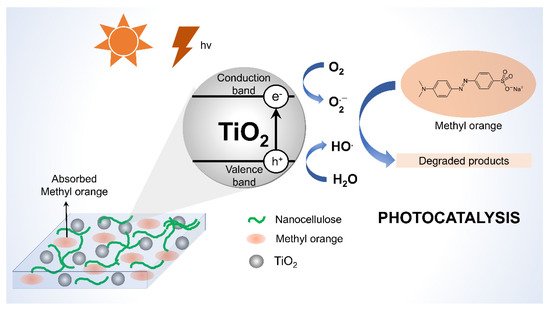
2/nanocellulose composite for photocatalytic degradation of methyl orange in an aqueous solution. The composite exhibited good morphological characteristics and an anatase crystal structure, with the specific surface area being proportional to the amount of CNFs used. Within 30 min, a 99.72% degradation rate of methyl orange was achieved with no noticeable activity loss observed after five cycles. The findings could aid in the development of effective photocatalysts for the treatment of organic dye effluent. The proposed mechanism of photocatalytic degradation of methylene blue by the action of TiO
2/nanocellulose is depicted in
Figure 13.
Figure 13. Mechanism of photocatalytic degradation of methylene blue by TiO2/GO/cellulose membrane.
Other researchers used a hydrolysis–precipitation approach to synthesize CeO
2/TiO
2 templates using nanocellulose. The photocatalytic performance of CeO
2/TiO
2-NCC was evaluated for the removal of Rhodamine B (RhB), methyl orange (MO), and the reduction of Cr (VI). Complete removal of a 250 mL and 50 ppm MO and RhB solution, as well as the reduction of a Cr(VI) solution, were accomplished in 70, 50, and 60 min, respectively [
98]. Despite the widespread usage of TiO
2 and other traditional photocatalysts such as zinc oxide (ZnO), they have been found to be ineffective under solar irradiation. This is due to the large bandgaps (3.2 eV) that allow only 4% of the potential 47% UV–Vis light radiation to be absorbed. Alternatively, the photocatalytic activities of silver phosphate (Ag
3PO
4) have been extensively studied for the removal of organic contaminants in the environment. Lebogang et al. (2019) [
112] synthesized an Ag
3PO
4/nanocellulose composite using an in situ facile method for the photocatalytic degradation of methylene blue and methyl orange under solar irradiation. The photocatalytic degradation rate of MO was 90% in DI and 70% in wastewater over the same period. Owing to the good compatibility between the particles, Ag
3PO
4 and nanocellulose, the nanocomposite will be a good candidate for the development of photocatalytic membranes for water and wastewater treatments.
On other occasions, Nahi et al. (2020) [
115] reported the green synthesis of a visible-light-driven photocatalyst made from a zinc oxide/nanocellulose composite (ZnO/NC) for the photodegradation of Enrofloxacin (EF), an antibiotic commonly used in veterinary medicine to treat animals with bacterial infections. The incorporation of nanocellulose enhanced the bandgap to the visible region, allowing degradation of the EF under visible light. Photo degradation was due to electron–hole interactions. The optimum pH was found to be 5, and the ZnO/NC dose was optimized to be 0.20 g/L. Equilibrium was attained at 120 min with a maximum degradation efficiency of 97%.
2.3. Nanocellulose for Gas Separation
Another important application of nanocellulose-based membranes is gas separation. CO
2 is the most prevalent anthropogenic greenhouse gas [
121]. Membranes selectively allow CO
2 to pass through from the feed side to the permeate side. A polyvinyl alcohol (PVA)/crystalline nanocellulose (CNC) nanocomposite membrane CO
2/CH
4 separation was fabricated by Jahan et al. (2018). The addition of 1.5% CNC to the PVA remarkably improved the membrane’s selectivity and permeance. In the study conducted by Torstensen et al. (2019) [
122], nanocellulose was employed as an additive to the PVA membrane for CO
2/N
2 mixed-gas separation. They discovered that the performance of the PVA/CNC membrane is comparable to that of PVA/carbon nanotubes, implying that CNCs can be used as an alternative to CNTs.
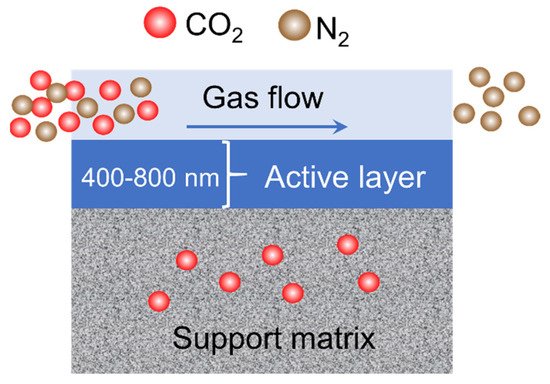
Figure 14 depicts the mechanism of CO
2/N
2 separation employing PVA/CNC as the active layer. Helberg et al. (2021) [
47] synthesized high-charge nanocellulose (H-P-CNF) and high-charge nanocellulose with screened size (H-P-CNF-S) and integrated them into a PVA matrix for CO
2/N
2 separation. The CO
2 permeability of the H-P-CNF-S/PVA membrane was 160% higher than that of the neat PVA. Their findings suggest that the high charge and small size of nanocellulose are useful properties for CO
2 separation under humid conditions.
Figure 14. Mechanism of CO2/N2 separation using PVA/CNC as active layer.
3. Pollutant Sensors
These days, sensor technologies are gaining a lot of attention, thanks to the rapid expansion of the Internet of Things (IoT) and the Industrial Revolution 4.0. Sensors detect and transform physical, chemical, and biological changes in their environment and convert them into analytical signals. Nanocellulose, being a natural nanomaterial, is crucial in the development of new sensors, especially in the context of developing a multidimensional architecture [
123,
124,
125,
126].
Nanocellulose has a number of hydroxyl groups that can be exploited to accommodate binding sites for selective analyte species adsorption, which increases electrochemical sensor selectivity, sensitivity, and durability. Taheri et al. [
124] discovered that an electrochemical sensor based on a D-penicillamine-anchored nano-cellulose (DPA-NC)-modified pencil graphite electrode exhibited good selectivity and sensitivity for copper ion in tap and river water samples, with a detection limit in the picomolar range (0.048 pM). The porous structure of the modifier and the formation of a compound between copper ions and nitrogen or oxygen-containing groups in DPA-NC may have contributed to the improved electrochemical responsiveness of the modified electrode. Shahi et al. [
125] found that the as-prepared CNF and (TEMPO)-oxidized cellulose nanofibers/glycerol (TOCNF/G) sensors demonstrated good gas sensing capability against acetone, ammonia, methane, and hydrogen sulphide after surface modification with TEMPO-mediated oxidation and glycerol. The presence of numerous carboxyl and hydroxyl groups was responsible for the change in ionic conductivity of the sensors in response to gas exposure.
TOCNF has also been used for the development of colorimetric sensors for the detection of fluoride ions. Fluoride in the soil, water, or even the environment can harm plants and has major effects on crop development, growth, and maturity, thus calling for effective monitoring and detection. Cross-linking of the TOCNF with chemically modified branched polyethyleneimine (bPEI) yielded a heterogeneous colorimetric sensor that enabled naked-eye detection of fluoride ions with concentrations of up to 0.05 M in DMSO. Moreover, it is highly selective towards fluoride ions over chloride, phosphate, and acetate ions [
101].
Pouzesh et al. developed an optical plasmonic chemosensor for detecting cyanide in environmental and industrial water samples. Cyanide ion (CN-) is one of the most hazardous ions for humans and microorganisms, even at low concentrations. The sensor was prepared by embedding stable copper nanoparticles in the nanocellulose framework (ECNPs-NC) by a direct chemical reduction of copper ions onto the nanocellulose using small amounts of sodium borohydride (NaBH4). Changes in the surface plasmon resonance absorption intensity of the ECNPs-NC film were monitored by a UV–Vis spectrophotometer. The linear range of absorption intensity was 0.25–0.40 μg mL−1 with a detection limit of 0.015 μg mL−1. The significant advantages of this method are its simplicity, no organic synthesis and solvent required, and sensing in pure aqueous media.
Besides chemical, electrochemical, and optical sensors, biosensors also have a vital role in detecting pollutants. CySense, a biosensor based on a red-fluorescent-sensing protein, known as cyanobacterial C-phycocyanin (CPC), and TOCNF has been fabricated by Weishaupt et al. [
127] for detection of copper ions. The covalent binding of CPC to TONFC stabilized the sensing molecule without the need for preservatives. The film production method used is reproducible in which the morphological and optical properties can be analyzed using techniques such as fluorescence spectrometry or microarray laser scanning. The ability to laser cut such CNF-based composite films could further aid their use in a variety of sensing applications.
The discovery of nanopaper was another significant step forward in the development of pollutant sensors based on nanocellulose. Nanopaper is a type of thin membrane constructed of cellulose nanofiber that opens up new avenues for producing simple, quick, low-cost, and single-use analytical instruments for a variety of applications, including environmental monitoring [
128]. The detection of iodide in seawater using nanopaper made from bacterial CNF is a recent example. Photoluminescence properties are obtained by embedding carbon-quantum dots in nanopaper. The nanopaper can be placed in a microcuvette for spectrofluorometer iodide sensing. Another application of nanopaper based on bacterial CNF is for the enantioselective detection of chiral compounds. In situ-synthesized silver nanoparticles were embedded in nanopaper, which displayed a significant color change from yellow to purple-brown in both the aqueous phase and the nanopaper upon aggregation of the silver nanoparticles induced by the chiral compounds [
133,
134]. Nanopapers can also be used to host nanoparticles activated by surface-enhanced Raman spectroscopy (SERS) such as silver and gold, for analyzing organic pollutants and aquatic pathogens [
135,
136,
137].