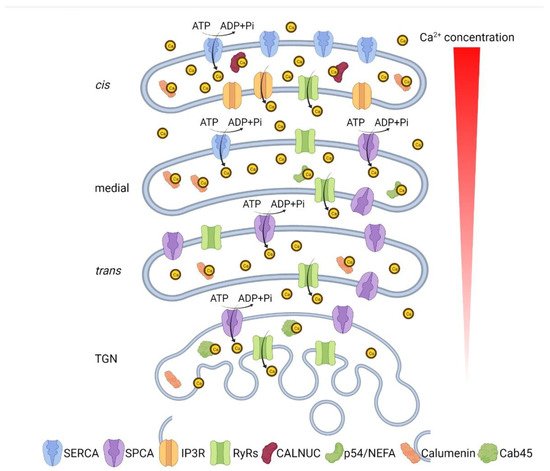
Inhibition of Ca
2+ pumps was reported to trigger a Golgi morphology change. Knockdown of SPCA1 leads to Golgi ribbon fragmentation, shortened and tubulated cisternae, and complete absence of
cis- and
trans-Golgi compartments [
41]. In a separate study, transient inhibition of SERCA by thapsigargin-induced Golgi fragmentation before the ER unfolded protein response (UPR) is triggered, indicating an ER-stress-independent Golgi morphology disruption [
65]. Further experiments demonstrated that thapsigargin-induced elevation of cytosolic Ca
2+ activates protein kinase C (PKCα), which subsequently phosphorylates and inactivates GRASP55, a key membrane tether in Golgi stack formation [
66,
67]. Golgi structural defects also impact membrane trafficking and secretion. Activation of PKCα with phorbol 12-myristate 13-acetate (PMA) and histamine modulates Golgi structure in a similar fashion, indicating a link between Ca
2+ signaling, Golgi structure and function, and human physiology [
37].
Many Golgi-resident glycosylation enzymes such as Golgi α-mannosidases, and proteases such as furin, show Ca
2+-binding activity [
68] or undergoes Ca
2+-dependent cleavage, which is required for their activation [
69]. Taking the Golgi mannosidases as examples, these
cis-Golgi resident enzymes are required in the early steps of N-glycosylation process, removing mannose residues from a high-mannose intermediate product. Ca
2+ binds to mannosidases prior to the substrate and remains associated during the enzymatic reaction. Divalent cations, including Mn
2+, compete with Ca
2+ and therefore inhibit the mannosidase activity [
70].
Mn
2+ is also a common ion associated with many Golgi-residing glycosylation enzymes, including N-acetylglucosaminyltransferases [
71], N-acetylgalactosaminyltransferases [
72], mannosyltransferases [
73], and some members in the fucosyltransferase family [
74,
75]. While Mn
2+ homeostasis in the Golgi lumen is required for proper glycosylation, high cytosolic Mn
2+ is toxic and related to many human diseases (
Table 1) [
76,
77]. By uptaking Mn
2+ from cytoplasm into the Golgi lumen, the SPCA pumps avoid cytotoxic accumulation of Mn
2+, therefore maintain Golgi Mn
2+ homeostasis and accurate glycosylation [
78,
79,
80]. On the other hand, exposure of Mn
2+ protects cells from some bacteria-originated toxins (e.g., Shiga toxin, STx) through down-regulation of Golgi phosphoprotein of 130 kDa (GPP130) and protects against STx-induced death [
81]. As a type 2 ribosome inactivating protein, STx binds to the cellular toxin receptor to be internalized, and is then transported via the retrograde trafficking route through endosomes and Golgi to the ER, where the toxin is translocated to the cytosol to practice its toxin activity [
82,
83]. GPP130 serves as a host-cell trafficking receptor that facilitates the intake of the toxin into the TGN via the interaction with Syntaxin 5 [
84]. Exposure to Mn
2+ blocks the retrograde trafficking of STx from endosome to Golgi and leads to its lysosomal degradation [
81]. Mn
2+ targets GPP130 and induces its oligomerization, causing GPP130 redistributed to lysosomes via a clathrin and Rab7-dependent mechanism, where it is subsequently degraded [
85,
86,
87]. Because of its sensitivity to Mn
2+ exposure, GPP130 can be used as an intra-Golgi Mn
2+ sensor in Golgi Mn
2+ homeostasis studies [
80].