Transcription factor 21 (TCF21) could promote chicken preadipocytes differentiation at least in part via activating MAPK/JNK pathway.
- broiler
- adipogenesis
- transcription factor 21
- MAPK/JNK signaling
1. Introduction
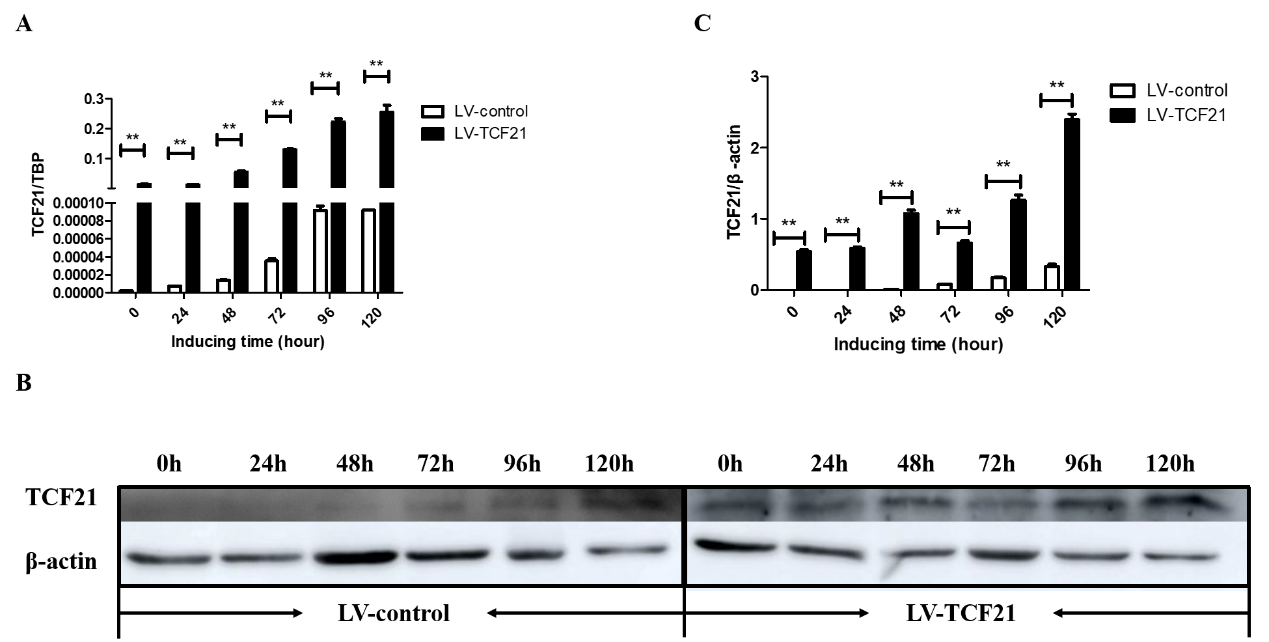
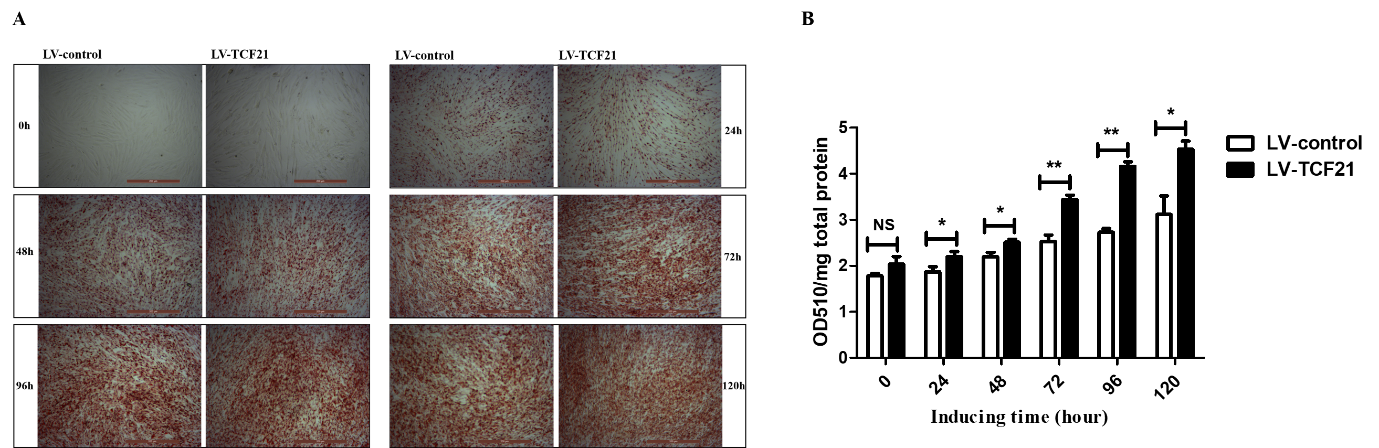
2. Overexpression of TCF21 Leads to Enhanced Lipid Droplets Accumulation
 3. MAPK/JNK Signaling Pathway Was Activated by TCF21 Overexpression
3. MAPK/JNK Signaling Pathway Was Activated by TCF21 Overexpression
| Pathway |
LV-control(mean±SE) |
LV-TCF21(mean±SE) |
P-value |
|
Amino acid deprivation |
0.41 ± 0.37 |
0.062 ± 0.043 |
0.44 |
|
Androgen |
/ |
/ |
/ |
|
Antioxidant response |
/ |
/ |
/ |
|
ATF6 |
0.015 ± 0.0048 |
0.013 ± 0.0027 |
0.72 |
|
C/EBP |
/ |
/ |
/ |
|
cAMP/PKA |
/ |
/ |
/ |
|
Cell cycle |
/ |
/ |
/ |
|
DNA damage |
/ |
/ |
/ |
|
EGR1 |
/ |
/ |
/ |
|
ER stress |
0.95 ± 0.72 |
0.90 ± 0.60 |
0.96 |
|
Estrogen |
/ |
/ |
/ |
|
GATA |
/ |
/ |
/ |
|
Glucocorticoid |
/ |
/ |
/ |
|
Heat shock |
/ |
/ |
/ |
|
Heavy metal |
0.090 ± 0.065 |
0.11 ± 0.078 |
0.87 |
|
Hedgehog |
/ |
/ |
/ |
|
HNF4 |
/ |
/ |
/ |
|
Hypoxia |
/ |
/ |
/ |
|
Interferon regulation |
/ |
/ |
/ |
|
Type 1 interferon |
/ |
/ |
/ |
|
Interferon-r |
/ |
/ |
/ |
|
KLF4 |
/ |
/ |
/ |
|
Liver X |
/ |
/ |
/ |
|
MAPK/Erk |
0.12 ± 0.043 |
0.11 ± 0.044 |
0.88 |
|
MAPK/Jnk |
0.028 ± 0.0060 |
0.19 ± 0.014 |
0.000423 |
|
MEF2 |
/ |
/ |
/ |
|
Myc |
/ |
/ |
/ |
|
Nanog |
/ |
/ |
/ |
|
Notch |
/ |
/ |
/ |
|
NFκB |
0.081 ± 0.037 |
0.20 ± 0.11 |
0.36 |
|
Oct4 |
/ |
/ |
/ |
|
Pax6 |
/ |
/ |
/ |
|
PI3K/Akt |
/ |
/ |
/ |
|
PKC/Ca+2 |
/ |
/ |
/ |
|
PPAR |
/ |
/ |
/ |
|
Progesterone |
/ |
/ |
/ |
|
Retinoic acid |
/ |
/ |
/ |
|
Retinoid X |
/ |
/ |
/ |
|
Sox2 |
/ |
/ |
/ |
|
SP1 |
0.3 ± 0.28 |
0.097 ± 0.067 |
0.52 |
|
STAT3 |
/ |
/ |
/ |
|
TGF-β |
/ |
/ |
/ |
|
Vitamin D |
/ |
/ |
/ |
|
Wnt |
/ |
/ |
/ |
|
Xenobiotic |
/ |
/ |
/ |
|
Negative control |
0.00057 ± 0.000067 |
0.000625 ± 0.000062 |
0.59 |

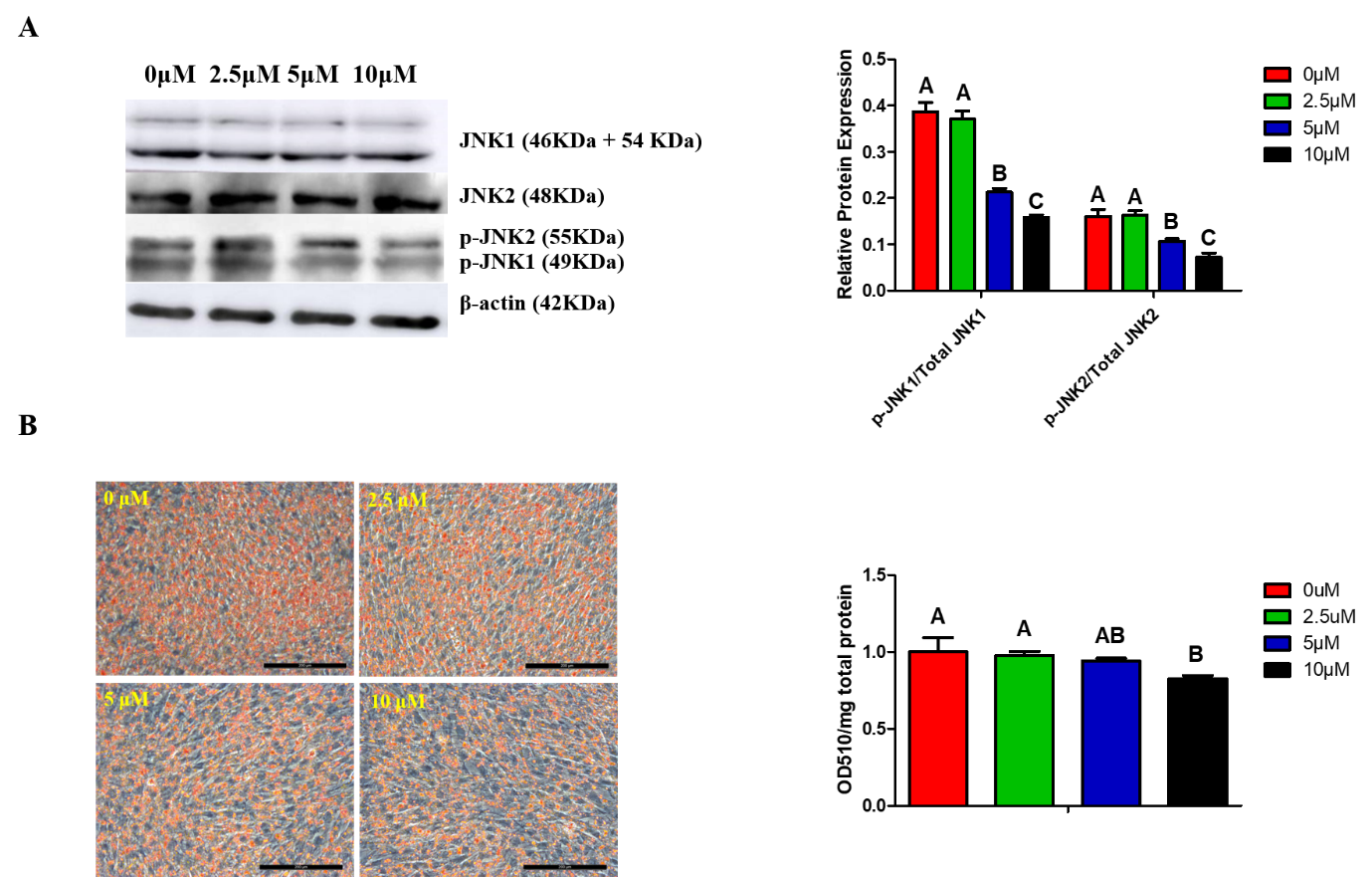
4. MAPK/JNK Signaling and Lipid Droplets Accumulation Are Inhibited by SP600125 in a Dose-Dependent Manner
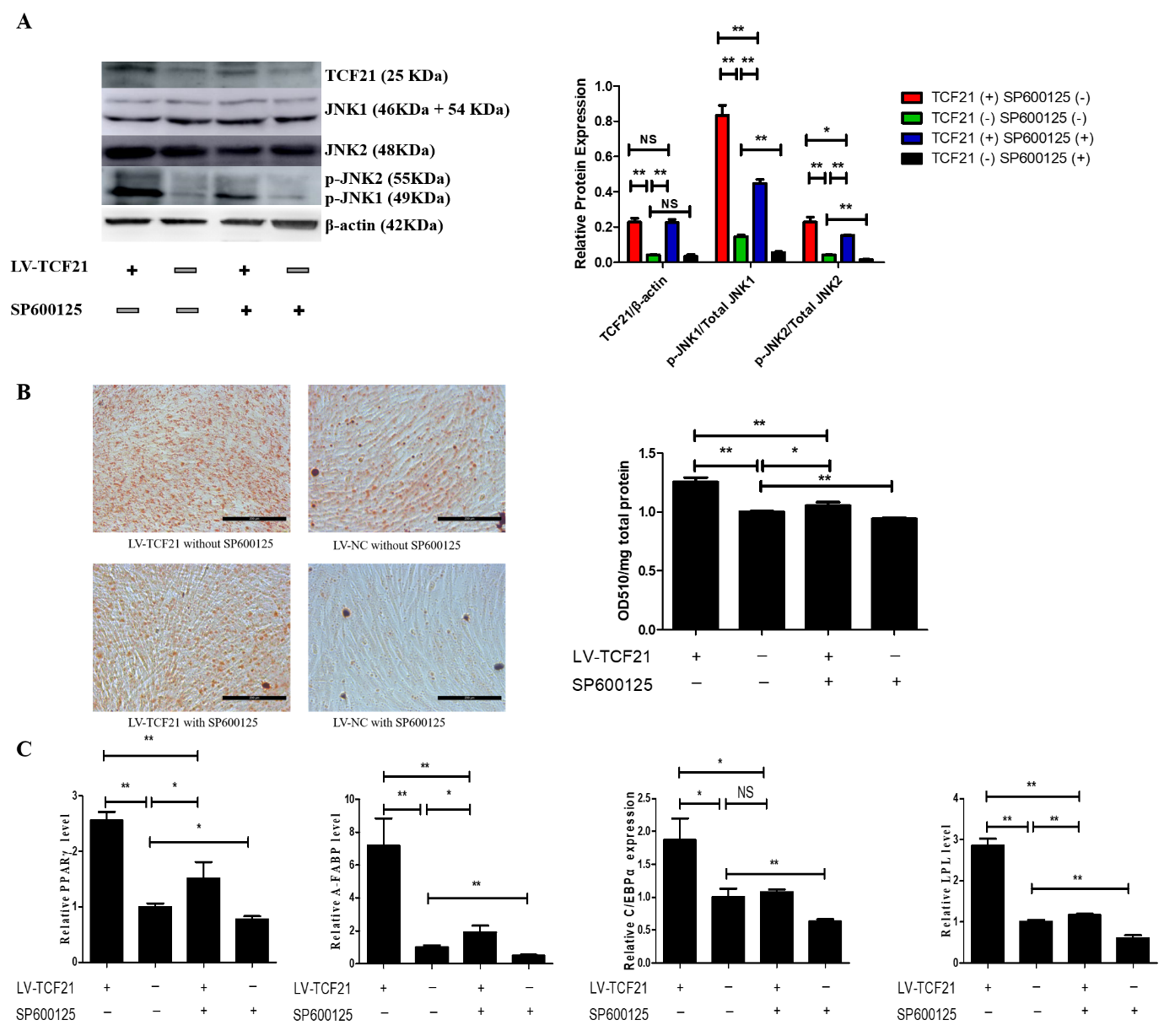
5. Inhibition of MAPK/JNK Signaling Attenuates TCF21-Mediated Promotion of Preadipocyte Differentiation
This entry is adapted from the peer-reviewed paper 10.3390/genes12121971
References
- Mallard, J.; Douaire, M. Strategies of selection for leanness in meat production. In Leanness in Domestic Birds: Genetic, Metabolic and Hormonal Aspects; Leclerq, B., Whitehead, C.C., Eds.; Elsevier: Amsterdam, The Netherlands, 1988; pp. 3–23.
- Julibert, A.; Bibiloni, M.D.M.; Mateos, D.; Angullo, E.; Tur, J.A. Dietary fat intake and metabolic syndrome in older adults. Nutrients 2019, 11, 1901.
- Vincent, M.J.; Allen, B.; Palacios, O.M.; Haber, L.T.; Maki, K.C. Meta-regression analysis of the effects of dietary cholesterol intake on LDL and HDL cholesterol. Am. J. Clin. Nutr. 2019, 109, 7–16.
- Wu, G.Q.; Deng, X.M.; Li, J.Y.; Li, N.; Yang, N. A potential molecular marker for selection against abdominal fatness in chickens. Poult. Sci. 2006, 85, 1896–1899.
- Sahraei, M. Feed restriction in broiler chickens production: A review. Glob. Vet. 2012, 8, 449–458.
- Julian, R.J. Production and growth related disorders and other metabolic diseases of poultry—A review. Vet. J. 2015, 169, 350–369.
- Chen, C.Y.; Huang, Y.F.; Ko, Y.J.; Liu, Y.J.; Chen, Y.H.; Walzem, R.L.; Chen, S.E. Obesity-associated cardiac pathogenesis in broiler breeder hens: Development of metabolic cardiomyopathy. Poult. Sci. 2017, 96, 2438–2446.
- Chen, C.Y.; Lin, H.Y.; Chen, Y.W.; Ko, Y.J.; Liu, Y.J.; Chen, Y.H.; Walzem, R.L.; Chen, S.E. Obesity-associated cardiac pathogenesis in broiler breeder hens: Pathological adaption of cardiac hypertrophy. Poult. Sci. 2017, 96, 2428–2437.
- Zhang, X.Y.; Wu, M.Q.; Wang, S.Z.; Zhang, H.; Du, Z.Q.; Li, Y.M.; Cao, Z.P.; Luan, P.; Leng, L.; Li, H. Genetic selection on abdominal fat content alters the reproductive performance of broilers. Animal 2018, 12, 1232–1241.
- Tang, Q.Q.; Lane, M.D. Adipogenesis: From stem cell to adipocyte. Annu. Rev. Biochem. 2012, 81, 715–736.
- Schwalie, P.C.; Dong, H.; Zachara, M.; Russeil, J.; Alpern, D.; Akchiche, N.; Caprara, C.; Sun, W.; Schlaudraff, K.; Soldati, G.; et al. A stromal cell population that inhibits adipogenesis in mammalian fat depots. Nature 2018, 559, 103–108.
- Rosen, E.D.; Macdougald, O.A. Adipocyte differentiation from inside out. Nat. Rev. Mol. Cell Biol. 2006, 7, 885–896.
- Cristancho, A.G.; Lazar, M.A. Forming functional fat: A growing understanding of adipocyte differentiation. Nat. Rev. Mol. Cell Biol. 2011, 12, 722–734.
- Ali, A.T.; Hochfeld, W.E.; Myburgh, R.; Pepper, M.S. Adipocyte and adipogenesis. Eur. J. Cell Biol. 2013, 92, 229–236.
- Zhang, X.; Cheng, B.; Liu, C.; Du, Z.; Zhang, H.; Wang, N.; Wu, M.; Li, Y.; Cao, Z.; Li, H. A novel regulator of preadipocyte differentiation, transcription factor TCF21, functions partially through promoting LPL expression. Front. Physiol. 2019, 10, 458.
