A point-of-care (POC) can be defined as an in vitro diagnostic test that can provide results within minutes. It has gained enormous attention as a promising tool for biomarkers detection and diagnosis, as well as for screening of chronic noncommunicable diseases such as diabetes mellitus. Diabetes mellitus type 2 is one of the metabolic disorders that has grown exponentially in recent years, becoming one of the greatest challenges to health systems. Early detection and accurate diagnosis of this disorder are essential to provide adequate treatments. Diabetes-monitoring tools must be accessible and affordable; thus, POC platforms are attractive, especially paper-based ones. Paper-based POCs are simple and portable, can use different matrixes, do not require highly trained staff, and are less expensive than other platforms. These advantages enhance the viability of its application in low-income countries and hard-to-reach zones.
- point-of-care testing
- paper-based analytical device
- colorimetry
- glucose
- type 2 diabetes mellitus
1. Introduction
2. Paper-Based Point-of-Care Platforms for Screening and Monitoring of Type 2 Diabetes Mellitus
Colorimetry is the most reported technique for determining glucose in clinical samples, mainly through the bienzymatic system consisting of glucose oxidase (GOD) and horseradish peroxidase (HRP) coupled to chromogens. The reaction catalyzed by glucose oxidase results in gluconic acid and hydrogen peroxide production. Peroxidase catalyzes the reaction of the hydrogen peroxide with the chromogen(s) to generate the color change. The two most commonly used HRP chromogenic substrates are 4-amino antipyrine (4-AAP) and 3,3’,5,5’-tetramethylbenzidine (TMB) [20][22][33][34]. Examples of the widespread application of the bienzymatic system GOD/HRP are shown in Table 1. It is vital to choose the right chromogen during the design of the paper-based assay platform to achieve suitable selectivity and specificity values for clinical application. There are several reports of using 4-AAP as a chromogen with detection limits relevant not only for the glucose determination in blood [4][35][36][37][38][39] but also in other biological fluids with low concentrations such as tears [40], urine [39], and saliva [41][42]. Other systems, such as GOD-HRP-TMB [43] and GOD-HRP-o-dianisidine [44], have shown promising results for detecting glucose in sweat.
| Substrate | System | Sample | Detection | LOD (mg/dL) | References |
|---|---|---|---|---|---|
| Wax printing on Whatman chromatography paper 595 | GOD/HRP/ 4-AAP/HBA |
Tears | Smartphone camera | NM | [40] |
| Whatman filter paper No. 1 with lamination film | GOD/BP | Saliva | Smartphone camera | 24.6 | [41] |
| Wax printing on qualitative filter paper and Schirmer strips | GOD/Au(I) complex (AuC2C6H4OMe)2 (Ph2P(C6H4)3PPh2) | Simulated tear fluid and blood | Bifurcated optical fiber system | 16.2 (plasma) 1.4 (tear) |
[45] |
| Whatman cellulose filter paper No. 1 treated with CH | GOD/HRP/ EDC/o-PD |
Urine | Smartphone camera | 18.0 | [46] |
| Whatman filter paper No. 40 stamped with paraffin and treated with CH | GOD/HRP/ TBHBA/4-AAP |
Artificial and human saliva | Naked eye | 0.8 | [42] |
| High-purity cellulose membranes | GOD/HRP/TMB | Urine | Digital camera | 8.1 | [34] |
| Whatman filter paper No. 1 with lamination film | GOD/BP | Saliva | Handheld optical biosensor | 32.0 | [47] |
| Wax printing on Whatman filter paper No. 1 | GOD/HRP/ KI or TMB |
Plasma | Smartphone camera | 27.0 (KI) 0.9 (TMB) |
[48] |
| Wax printing on Whatman filter paper No. 1 treated with CH | GOD/HRP/ 4-AAP/HBA |
Blood | Scanner | NM | [36] |
| Whatman qualitative paper No.1 treated with PB | GOD | Serum | Distance-based measurements | 19.8 | [49] |
| Nitrocellulose membranes | GOD/HRP/4-AAP/COL/MADB | Serum | Chemidoc imaging system | 0.2 | [37] |
| Wax printing on Whatman No. 1 cellulose chromatography paper treated with BSA | GOD/HRP/ 4-AAP/DHBS |
Serum | Scanner | 5.4 | [4] |
| Whatman qualitative filter paper No. 1 coated with a UV-curable resin | GOD/HRP/ MAOS/4-AAP |
Serum | Smartphone camera | 5.4 | [38] |
| Wax printing in Whatman No. 1 chromatography filter paper treated with CH | GOD/HRP/TMB | Blood | Smartphone-based optical platform | 5.0 | [50] |
| Whatman filter paper No. 3 treated with OTS and MTS | GOD/HRP/ phenol/4-AAP |
Plasma | Portable scanner | 15.1 | [35] |
| Wax printing in Whatman No. 1 qualitative filter paper loaded with ZnNR | GOD/ 4-AAP/ DHBS |
Serum and urine | Smartphone camera | 0.05 | [39] |
| Whatman filter paper No. 41 treated with BSA-Tween | GOD/HRP/TMB | Sweat | Scanner and Smartphone camera | 0.18 | [43] |
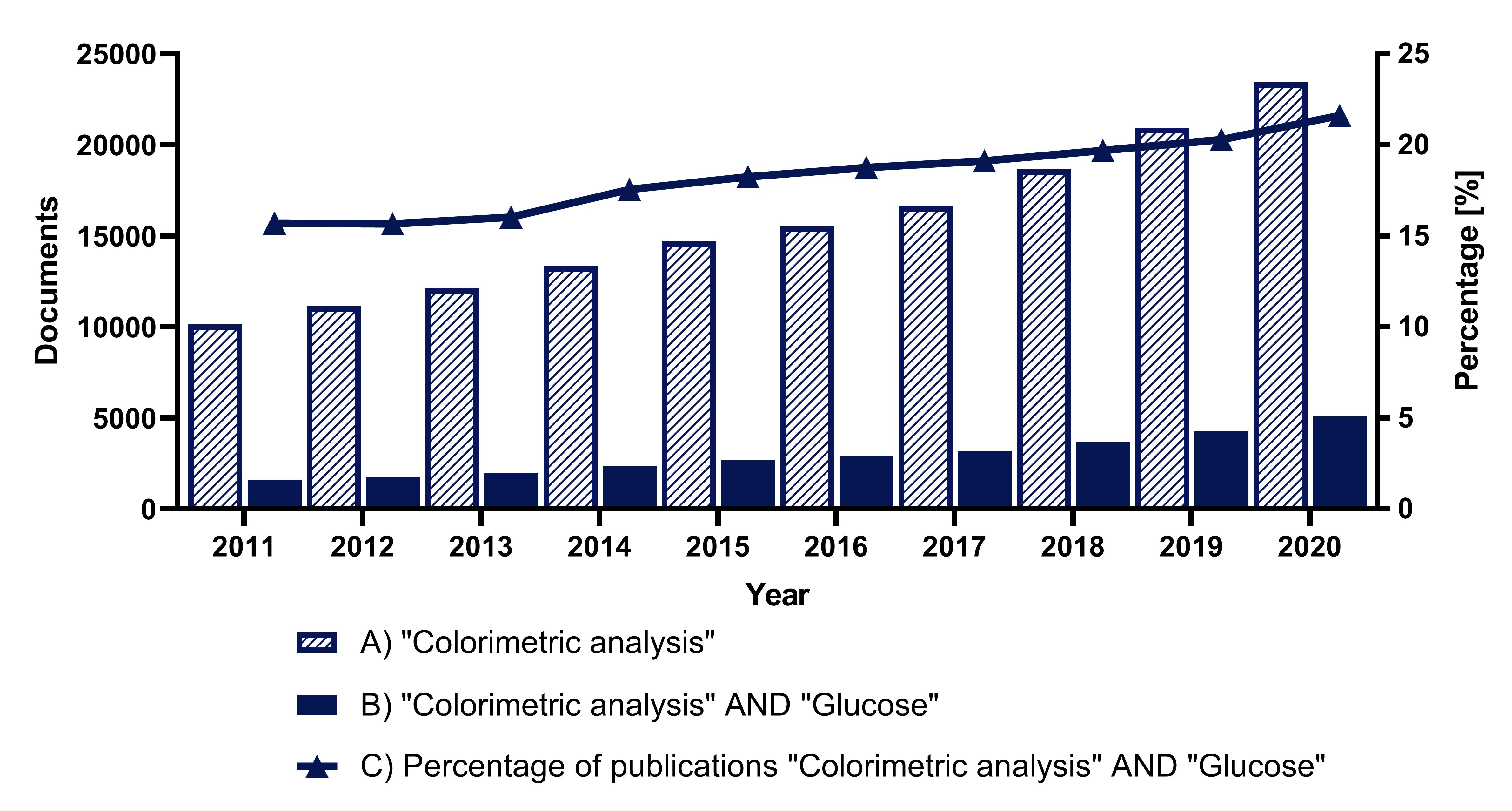 Figure 1. Analysis of the number of documents in Scopus over the last ten years: (A) using the search term “Colorimetric analysis”, (B) using the search terms “Colorimetric analysis” AND “Glucose”, (C) percentage of papers found using the term “Glucose” in the category “Colorimetric analysis”.
Figure 1. Analysis of the number of documents in Scopus over the last ten years: (A) using the search term “Colorimetric analysis”, (B) using the search terms “Colorimetric analysis” AND “Glucose”, (C) percentage of papers found using the term “Glucose” in the category “Colorimetric analysis”.This entry is adapted from the peer-reviewed paper 10.3390/bios11120482
References
- Suntornsuk, W.; Suntornsuk, L. Recent applications of paper-based point-of-care devices for biomarker detection. Electrophoresis 2020, 41, 287–305.
- Shrivastava, S.; Trung, T.Q.; Lee, N.E. Recent progress, challenges, and prospects of fully integrated mobile and wearable point-of-care testing systems for self-testing. Chem. Soc. Rev. 2020, 49, 1812–1866.
- Abdollahi-Aghdam, A.; Majidi, M.R.; Omidi, Y. Microfluidic paper-based analytical devices (µPADs) for fast and ultrasensitive sensing of biomarkers and monitoring of diseases. BioImpacts 2018, 8, 237–240.
- Kim, S.; Kim, D.; Kim, S. Simultaneous quantification of multiple biomarkers on a self-calibrating microfluidic paper-based analytic device. Anal. Chim. Acta 2020, 1097, 120–126.
- Da Silva, V.A.O.P.; de Freitas, R.C.; de Oliveira, P.R.; Moreira, R.C.; Marcolino-Júnior, L.H.; Bergamini, M.F.; Coltro, W.K.T.; Janegitz, B.C. Microfluidic paper-based device integrated with smartphone for point-of-use colorimetric monitoring of water quality index. Measurement 2020, 164, 108085.
- Cai, Y.; Niu, J.C.; Du, X.L.; Fang, F.; Wu, Z.Y. Novel field amplification for sensitive colorimetric detection of microalbuminuria on a paper-based analytical device. Anal. Chim. Acta 2019, 1080, 146–152.
- Ouyang, J.; Pu, S.; Chen, X.; Yang, C.; Zhang, X.; Li, D. A convenient and rapid method for detecting D-glucose in honey used smartphone. Food Chem. 2020, 331, 127348.
- Aksorn, J.; Teepoo, S. Development of the simultaneous colorimetric enzymatic detection of sucrose, fructose and glucose using a microfluidic paper-based analytical device. Talanta 2020, 207, 120302.
- Coetzee, A.; van de Vyver, M.; Hoffmann, M.; Hall, D.R.; Mason, D.; Conradie, M. A comparison between point-of-care testing and venous glucose determination for the diagnosis of diabetes mellitus 6–12 weeks after gestational diabetes. Diabet. Med. 2019, 36, 591–599.
- Lisi, F.; Peterson, J.R.; Gooding, J.J. The application of personal glucose meters as universal point-of-care diagnostic tools. Biosens. Bioelectron. 2020, 148, 111835.
- Chin, C.D.; Linder, V.; Sia, S.K. Commercialization of microfluidic point-of-care diagnostic devices. Lab Chip 2012, 12, 2118–2134.
- Kosack, C.S.; Page, A.L.; Klatser, P.R. A guide to aid the selection of diagnostic tests. Bull. World Health Organ. 2017, 95, 639–645.
- Martinez, A.W.; Phillips, S.T.; Whitesides, G.M.; Carrilho, E. Diagnostics for the Developing World: Microfluidic Paper-Based Analytical Devices. Anal. Chem. 2010, 82, 3–10.
- World Health Organization. From Bench to Bedside: Setting a Path for the Translation of Improved STI Diagnostics into Health Care Delivery in the Developing World an Informal Consultation Jointly Organised and Sponsored By; World Health Organization: Geneva, Switzerland, 2001.
- Instituto Nacional de Estadística y Geografía; Instituto Nacional de Salud Pública; Secretaría de Salud. Encuesta Nacional de Salud y Nutrición. Ensanut 2018, 1, 47.
- International Diabetes Federation. IDF Diabetes Atlas, 9th ed.; International Diabetes Federation: Brussels, Belgium, 2019; ISBN 9782930229874.
- DeFronzo, R.A.; Ferrannini, E.; Groop, L.; Henry, R.R.; Herman, W.H.; Holst, J.J.; Hu, F.B.; Kahn, C.R.; Raz, I.; Shulman, G.I.; et al. Type 2 diabetes mellitus. Nat. Rev. Dis. Prim. 2015, 1, 15019.
- Riddy, D.M.; Delerive, P.; Summers, R.J.; Sexton, P.M.; Langmead, C.J. G protein–coupled receptors targeting insulin resistance, obesity, and type 2 diabetes mellitus. Pharmacol. Rev. 2018, 70, 39–67.
- Kim, J.; Campbell, A.S.; de Ávila, B.E.F.; Wang, J. Wearable biosensors for healthcare monitoring. Nat. Biotechnol. 2019, 37, 389–406.
- Liu, S.; Su, W.; Ding, X. A review on microfluidic paper-based analytical devices for glucose detection. Sensors 2016, 16, 2086.
- Van Enter, B.J.; Von Hauff, E. Challenges and perspectives in continuous glucose monitoring. Chem. Commun. 2018, 54, 5032–5045.
- Nery, E.W.; Kundys, M.; Jeleń, P.S.; Jönsson-Niedziólka, M. Electrochemical glucose sensing: Is there still room for improvement? Anal. Chem. 2016, 88, 11271–11282.
- World Health Organization. The Global Health Observatory. Available online: https://www.who.int/data/gho/indicator-metadata-registry/imr-details/2380 (accessed on 28 July 2021).
- American Diabetes Association. 2. Classification and Diagnosis of Diabetes: Standards of Medical Care in Diabetes—2020. Diabetes Care 2020, 43, S14–S31.
- Tabák, A.G.; Herder, C.; Rathmann, W.; Brunner, E.J.; Kivimäki, M. Prediabetes: A high-risk state for diabetes development. Lancet 2012, 379, 2279–2290.
- Tang, L.; Chang, S.J.; Chen, C.J.; Liu, J.T. Non-invasive blood glucose monitoring technology: A review. Sensors 2020, 20, 6925.
- Yoo, E.H.; Lee, S.Y. Glucose biosensors: An overview of use in clinical practice. Sensors 2010, 10, 4558–4576.
- Wang, J. Electrochemical glucose biosensors. Chem. Rev. 2008, 108, 814–825.
- Juska, V.B.; Pemble, M.E. A Critical Review of Electrochemical Glucose Sensing: Evolution of Biosensor Platforms Based on Advanced Nanosystems. Sensors 2020, 20, 6013.
- Naikoo, G.A.; Awan, T.; Salim, H.; Arshad, F.; Hassan, I.U.; Pedram, M.Z.; Ahmed, W.; Faruck, H.L.; Aljabali, A.A.A.; Mishra, V.; et al. Fourth-generation glucose sensors composed of copper nanostructures for diabetes management: A critical review. Bioeng. Transl. Med. 2021, e10248.
- Zhang, H.; Zhang, W.; Zhou, A. Smartphone for glucose monitoring. In Smartphone Based Medical Diagnostics; Elsevier: Amsterdam, The Netherlands, 2020; pp. 45–65. ISBN 9780128170441.
- Teymourian, H.; Barfidokht, A.; Wang, J. Electrochemical glucose sensors in diabetes management: An updated review (2010–2020). Chem. Soc. Rev. 2020, 49, 7671–7709.
- De Tarso Garcia, P.; Garcia Cardoso, T.M.; Garcia, C.D.; Carrilho, E.; Tomazelli Coltro, W.K. A handheld stamping process to fabricate microfluidic paper-based analytical devices with chemically modified surface for clinical assays. RSC Adv. 2014, 4, 37637–37644.
- Luo, X.; Xia, J.; Jiang, X.; Yang, M.; Liu, S. Cellulose-Based Strips Designed Based on a Sensitive Enzyme Colorimetric Assay for the Low Concentration of Glucose Detection. Anal. Chem. 2019, 91, 15461–15468.
- Hou, P.; Deng, R.; Guo, J.; Chen, W.; Li, X.; Yu, H.-Z. A WiFi scanner in conjunction with disposable multiplex paper assay for the quantitation of disease markers in blood plasma. Anal. Bioanal. Chem. 2021, 413, 4625–4634.
- Kim, D.; Kim, S.; Kim, S. An innovative blood plasma separation method for a paper-based analytical device using chitosan functionalization. Analyst 2020, 145, 5491–5499.
- Ki, H.; Jang, H.; Oh, J.; Han, G.R.; Lee, H.; Kim, S.; Kim, M.G. Simultaneous Detection of Serum Glucose and Glycated Albumin on a Paper-Based Sensor for Acute Hyperglycemia and Diabetes Mellitus. Anal. Chem. 2020, 92, 11530–11534.
- Baek, S.H.; Park, C.; Jeon, J.; Park, S. Three-Dimensional Paper-Based Microfluidic Analysis Device for Simultaneous Detection of Multiple Biomarkers with a Smartphone. Biosensors 2020, 10, 187.
- Feng, L.X.; Tang, C.; Han, X.X.; Zhang, H.C.; Guo, F.N.; Yang, T.; Wang, J.H. Simultaneous and sensitive detection of multiple small biological molecules by microfluidic paper-based analytical device integrated with zinc oxide nanorods. Talanta 2021, 232, 122499.
- Kang, B.H.; Park, M.; Jeong, K.H. Colorimetric Schirmer strip for tear glucose detection. BioChip J. 2017, 11, 294–299.
- Soni, A.; Jha, S.K. Smartphone based non-invasive salivary glucose biosensor. Anal. Chim. Acta 2017, 996, 54–63.
- Santana-Jiménez, L.; Márquez-Lucero, A.; Osuna, V.; Estrada-Moreno, I.; Dominguez, R. Naked-Eye Detection of Glucose in Saliva with Bienzymatic Paper-Based Sensor. Sensors 2018, 18, 1071.
- Vaquer, A.; Barón, E.; de la Rica, R. Detection of low glucose levels in sweat with colorimetric wearable biosensors. Analyst 2021, 146, 3273–3279.
- Xiao, J.; Liu, Y.; Su, L.; Zhao, D.; Zhao, L.; Zhang, X. Microfluidic Chip-Based Wearable Colorimetric Sensor for Simple and Facile Detection of Sweat Glucose. Anal. Chem. 2019, 91, 14803–14807.
- Luo, J.-J.; Pan, S.-W.; Yang, J.-H.; Chang, T.-L.; Lin, P.-Y.; Wu, C.-L.; Liu, W.-F.; Huang, X.-R.; Koshevoy, I.; Chou, P.-T.; et al. Detecting Glucose Levels in Blood Plasma and Artificial Tear by Au(I) Complex on the Carbopol Polymer: A Microfluidic Paper-Based Method. Polymers 2018, 10, 1001.
- Martinkova, P.; Brtnicky, M.; Kynicky, J.; Pohanka, M. Fast and simple glucose assay based on filter paper as enzymes carrier using phone camera detection. Chem. Pap. 2018, 72, 2719–2728.
- Singh, A.K.; Jha, S.K. Fabrication and Validation of a Handheld Non-Invasive, Optical Biosensor for Self-Monitoring of Glucose Using Saliva. IEEE Sens. J. 2019, 19, 8332–8339.
- Choobbari, M.L.; Rad, M.B.; Jahanshahi, A.; Ghourchian, H. A sample volume independent paper microfluidic device for quantifying glucose in real human plasma. Microfluid. Nanofluidics 2020, 24, 74.
- Granica, M.; Tymecki, Ł. Prussian Blue (bio)sensing device for distance-based measurements. Anal. Chim. Acta 2020, 1136, 125–133.
- Zhang, H.; Chen, Z.; Dai, J.; Zhang, W.; Jiang, Y.; Zhou, A. A low-cost mobile platform for whole blood glucose monitoring using colorimetric method. Microchem. J. 2021, 162, 105814.
- Soni, A.; Jha, S.K. A paper strip based non-invasive glucose biosensor for salivary analysis. Biosens. Bioelectron. 2015, 67, 763–768.
- Gao, Z.F.; Sann, E.E.; Lou, X.; Liu, R.; Dai, J.; Zuo, X.; Xia, F.; Jiang, L. Naked-eye point-of-care testing platform based on a pH-responsive superwetting surface: Toward the non-invasive detection of glucose. NPG Asia Mater. 2018, 10, 177–189.
- Fernandes, G.M.; Silva, W.R.; Barreto, D.N.; Lamarca, R.S.; Lima Gomes, P.C.F.; da S Petruci, J.F.; Batista, A.D. Novel approaches for colorimetric measurements in analytical chemistry—A review. Anal. Chim. Acta 2020, 1135, 187–203.
- Nathan, D.M.; Balkau, B.; Bonora, E.; Borch-Johnsen, K.; Buse, J.B.; Colagiuri, S.; Davidson, M.B.; DeFronzo, R.; Genuth, S.; Holman, R.R.; et al. International Expert Committee Report on the Role of the A1C Assay in the Diagnosis of Diabetes. Diabetes Care 2009, 32, 1327–1334.
- American Diabetes Association. 2. Classification and Diagnosis of Diabetes: Standards of Medical Care in Diabetes—2019. Diabetes Care 2019, 42, S13–S28.
- Karnchanasorn, R.; Huang, J.; Ou, H.-Y.; Feng, W.; Chuang, L.-M.; Chiu, K.C.; Samoa, R. Comparison of the Current Diagnostic Criterion of HbA1c with Fasting and 2-Hour Plasma Glucose Concentration. J. Diabetes Res. 2016, 2016, 1–11.
- American Diabetes Association. 2. Classification and Diagnosis of Diabetes: Standards of Medical Care in Diabetes—2021. Diabetes Care 2021, 44, S15–S33.
- Nathan, D.M.; Griffin, A.; Perez, F.M.; Basque, E.; Do, L.; Steiner, B. Accuracy of a Point-of-Care Hemoglobin A1c Assay. J. Diabetes Sci. Technol. 2019, 13, 1149–1153.
- Dubach, I.L.; Christ, E.R.; Diem, P. HbA1c-testing: Evaluation of two point-of-care analysers. Prim. Care Diabetes 2019, 13, 583–587.
- Hirst, J.A.; McLellan, J.H.; Price, C.P.; English, E.; Feakins, B.G.; Stevens, R.J.; Farmer, A.J. Performance of point-of-care HbA1c test devices: Implications for use in clinical practice—a systematic review and meta-analysis. Clin. Chem. Lab. Med. 2017, 55, 167–180.
- Lenters-Westra, E.; English, E. Evaluation of Four HbA1c Point-of-Care Devices Using International Quality Targets: Are They Fit for the Purpose? J. Diabetes Sci. Technol. 2018, 12, 762–770.
- Dupuy, A.M.; Badiou, S.; Bargnoux, A.S.; Russelo, J.; Cristol, J.P. Evaluation of Point of Care Analyzer for Hemoglobin A1c. J. Diabetes Sci. Technol. 2019, 13, 150–151.
- Arnold, W.D.; Kupfer, K.; Little, R.R.; Amar, M.; Horowitz, B.; Godbole, N.; Swensen, M.H.; Li, Y.; San George, R.C. Accuracy and Precision of a Point-of-Care HbA1c Test. J. Diabetes Sci. Technol. 2020, 14, 883–889.
- Boonyasit, Y.; Chailapakul, O.; Laiwattanapaisal, W. A multiplexed three-dimensional paper-based electrochemical impedance device for simultaneous label-free affinity sensing of total and glycated haemoglobin: The potential of using a specific single-frequency value for analysis. Anal. Chim. Acta 2016, 936, 1–11.
- Hossain, M.D.M.; Moon, J.M.; Gurudatt, N.G.; Park, D.S.; Choi, C.S.; Shim, Y.B. Separation detection of hemoglobin and glycated hemoglobin fractions in blood using the electrochemical microfluidic channel with a conductive polymer composite sensor. Biosens. Bioelectron. 2019, 142, 111515.
- Shajaripour Jaberi, S.Y.; Ghaffarinejad, A.; Omidinia, E. An electrochemical paper based nano-genosensor modified with reduced graphene oxide-gold nanostructure for determination of glycated hemoglobin in blood. Anal. Chim. Acta 2019, 1078, 42–52.
- Koga, M.; Kasayama, S. Clinical impact of glycated albumin as another glycemic control marker. Endocr. J. 2010, 57, 751–762.
- Dorcely, B.; Katz, K.; Jagannathan, R.; Chiang, S.S.; Oluwadare, B.; Goldberg, I.J.; Bergman, M. Novel biomarkers for prediabetes, diabetes, and associated complications. Diabetes Metab. Syndr. Obes. Targets Ther. 2017, 10, 345–361.
- Paroni, R.; Ceriotti, F.; Galanello, R.; Leoni, G.B.; Panico, A.; Scurati, E.; Paleari, R.; Chemello, L.; Quaino, V.; Scaldaferri, L.; et al. Performance characteristics and clinical utility of an enzymatic method for the measurement of glycated albumin in plasma. Clin. Biochem. 2007, 40, 1398–1405.
- Gan, T.; Liu, X.; Xu, G. Glycated Albumin Versus HbA1c in the Evaluation of Glycemic Control in Patients With Diabetes and CKD. Kidney Int. Rep. 2018, 3, 542–554.
- Vernon Roohk, H.; Zaidi, A.R. A review of glycated albumin as an intermediate glycation index for controlling diabetes. J. Diabetes Sci. Technol. 2008, 2, 1114–1121.
- Hatada, M.; Wilson, E.; Khanwalker, M.; Probst, D.; Okuda-Shimazaki, J.; Sode, K. Current and future prospective of biosensing molecules for point-of-care sensors for diabetes biomarker. Sens. Actuators B Chem. 2022, 351, 130914.
- Belsare, S.; Coté, G. Development of a colorimetric paper fluidic dipstick assay for measurement of glycated albumin to monitor gestational diabetes at the point-of-care. Talanta 2021, 223, 121728.
- Hatada, M.; Tsugawa, W.; Kamio, E.; Loew, N.; Klonoff, D.C.; Sode, K. Development of a screen-printed carbon electrode based disposable enzyme sensor strip for the measurement of glycated albumin. Biosens. Bioelectron. 2017, 88, 167–173.
- Hatada, M.; Loew, N.; Okuda-Shimazaki, J.; Khanwalker, M.; Tsugawa, W.; Mulchandani, A.; Sode, K. Development of an Interdigitated Electrode-Based Disposable Enzyme Sensor Strip for Glycated Albumin Measurement. Molecules 2021, 26, 734.
- Schleicher, E.D.; Vogt, B.W. Standardization of serum fructosamine assays. Clin. Chem. 1990, 36, 136–139.
- Baker, J.R.; Johnson, R.N.; Scott, D.J. Serum fructosamine concentrations in patients with type II (non-insulin-dependent) diabetes mellitus during changes in management. BMJ 1984, 288, 1484–1486.
- Boonyasit, Y.; Laiwattanapaisal, W. A microfluidic paper-based analytical device for the assay of albumin-corrected fructosamine values from whole blood samples. Bioanalysis 2015, 7, 79–90.
- Selvin, E.; Rawlings, A.M.; Grams, M.; Klein, R.; Sharrett, A.R.; Steffes, M.; Coresh, J. Fructosamine and glycated albumin for risk stratification and prediction of incident diabetes and microvascular complications: A prospective cohort analysis of the Atherosclerosis Risk in Communities (ARIC) study. Lancet Diabetes Endocrinol. 2014, 2, 279–288.
- Khoury, Z.H.H.; Illesca, P.; Sultan, A.S.S. Salivary Fructosamine as a Noninvasive Glycemic Biomarker: A Systematic Review. JDR Clin. Transl. Res. 2021, 6, 382–389.
- Guo, J.; Ma, X. Simultaneous monitoring of glucose and uric acid on a single test strip with dual channels. Biosens. Bioelectron. 2017, 94, 415–419.
- Manbohi, A.; Ahmadi, S.H. Chitosan–Fe3O4 nanoparticle enzymatic electrodes on paper as an efficient assay for glucose and uric acid detection in biological fluids. Chem. Pap. 2020, 74, 2675–2687.
- Moreira, N.S.; Chagas, C.L.S.; Oliveira, K.A.; Duarte-Junior, G.F.; de Souza, F.R.; Santhiago, M.; Garcia, C.D.; Kubota, L.T.; Coltro, W.K.T. Fabrication of microwell plates and microfluidic devices in polyester films using a cutting printer. Anal. Chim. Acta 2020, 1119, 1–10.
- Chen, Y.-A.; Tsai, F.-J.; Zeng, Y.-T.; Wang, J.-C.; Hong, C.P.; Huang, P.-H.; Chuang, H.-L.; Lin, S.-Y.; Chan, C.-T.; Ko, Y.-C.; et al. Fast and Effective Turn-on Paper-based Phosphorescence Biosensor for Detection of Glucose in Serum. J. Chin. Chem. Soc. 2016, 63, 424–431.
- Rossini, E.L.; Milani, M.I.; Lima, L.S.; Pezza, H.R. Paper microfluidic device using carbon dots to detect glucose and lactate in saliva samples. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2021, 248, 119285.
- Yi, D.; Wei, Z.; Zheng, W.; Pan, Y.; Long, Y.; Zheng, H. Glucose detection based on the photothermal effect of OxTMB using a thermometer. Sens. Actuators B Chem. 2020, 323, 128691.
- Davaji, B.; Lee, C.H. A paper-based calorimetric microfluidics platform for bio-chemical sensing. Biosens. Bioelectron. 2014, 59, 120–126.
- Colletes, T.C.; Garcia, P.T.; Campanha, R.B.; Abdelnur, P.V.; Romão, W.; Coltro, W.K.T.; Vaz, B.G. A new insert sample approach to paper spray mass spectrometry: A paper substrate with paraffin barriers. Analyst 2016, 141, 1707–1713.
- De Freitas, S.V.; De Souza, F.R.; Rodrigues Neto, J.C.; Vasconcelos, G.A.; Abdelnur, P.V.; Vaz, B.G.; Henry, C.S.; Coltro, W.K.T. Uncovering the Formation of Color Gradients for Glucose Colorimetric Assays on Microfluidic Paper-Based Analytical Devices by Mass Spectrometry Imaging. Anal. Chem. 2018, 90, 11949–11954.
- De Castro, L.F.; de Freitas, S.V.; Duarte, L.C.; de Souza, J.A.C.; Paixão, T.R.L.C.; Coltro, W.K.T. Salivary diagnostics on paper microfluidic devices and their use as wearable sensors for glucose monitoring. Anal. Bioanal. Chem. 2019, 411, 4919–4928.
